Ingredients
 | A can |  | A cooker hob |
 | Some tongs |  | A tray |
Instructions
Warning: this experiment involves inverting a can of boiling water and you should use eye protection and be careful.
Take an empty drinks can (not one with a plastic widget) and pour in a small amount of water, only a centimetre or so depth is fine. Place the can on a cooker hob (being careful to make sure it's stable) and heat until the water is boiling and you can see plenty of steam coming out of the top.
Fill a shallow bowl or tray with cold water and put it in a convenient place next to your hob.
Pick up the can with a pair of tongs and carefully invert the can into the cold water, so the mouth of the can is at the bottom, under the surface of the water.
Result
The can collapses very soon after it hits the cold water
Explanation
As you heat the can to 100°C the water inside starts to boil, turning into steam and increasing in volume by a factor of about 600 times. This steam pushes out, or displaces, most of the air in the can.
 | 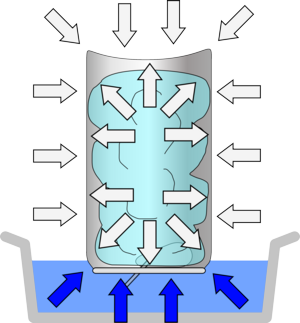 | 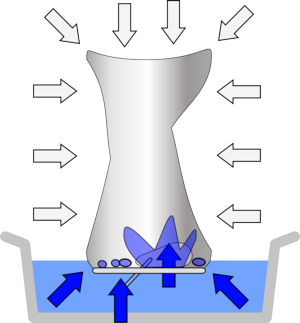 |
| When you heat the can the water turns to steam, displacing the air. | While there is steam in the can its pressure is equal to the air pressure outside, so there is no overall force on the can. | When it cools, the steam condenses very rapidly, leaving nothing to support the can from the inside - so the air pressure crushes it! |
When you put the can into the cold water the sides of the can cool down rapidly, which allows the steam to condense on the sides of the can. This happens much too rapidly for the water to rush in through the mouth of the can and fill the space, so the pressure decreases inside the can and the external air pressure crushes the can.
Is this effect useful?
It was the basis for one of the first designs of successful steam engines developed by Thomas Newcomen. This worked by injecting steam into the cylinder, then shutting the valve to seal it off. Cold water was then squirted in, which caused the steam to condense. This allowed the air pressure to push the piston down the cylinder. This force was then transmitted to a pump at the bottom of a mine using a long chain.
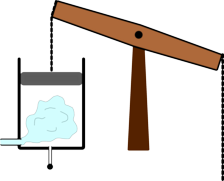 | 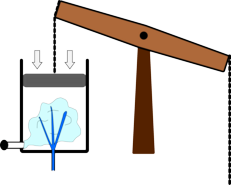 | 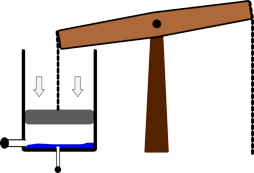 |
| Steam is injected into the Newcomen engine | Water is then injected into the cylinder | This reduces the pressure inside the cylinder and the air pressure pushes the piston down. |









Comments
Add a comment