Imaging Cancer
Interview with
Ben - The science surrounding cancer is multifaceted. It involves clinicians, chemists, geneticists, and even physicists. The essential work of developing new treatments and clinical approaches is backed up by developments in imaging technology that allow us to detect and observe tumours with ever increasing fidelity. Professor Martin Leach is Co-director of the Cancer Research UK and EPSRC Cancer Imaging Centre at the Institute of Cancer Research, and he joins us now. Martin, thank you very much for joining us. I was wondering first of all, what sorts of roles does imaging play in cancer and what sorts of imaging do you actually do?
Martin - Well, imaging ranges all the way through the cancer pathway. A very important aspect is whether we can detect disease early because the earlier we can detect it, the better we can treat it. Then we have to see what the status of the disease is, how extensive it is, and whether it's spreading through the body because that dictates different types of treatment. And then we need to guide new treatments using imaging, and imaging is very important in a whole range of guidance methods. We do a lot of magnetic resonance imaging (MRI), that's one of the important techniques that's come on the last 20 years or so, but we also do CT and x-ray imaging, and isotope imaging. And all of these have different roles to play and are often complimentary in understanding the disease and how it's behaving.
 Ben - So what are the challenges you're facing in imaging at the moment? What are the goals that you'd really like to reach?
Ben - So what are the challenges you're facing in imaging at the moment? What are the goals that you'd really like to reach?
Martin - Well, we really like to be able to now, not just see where disease is - and that's the historical perspective. If you look at x-rays, you see these spots on the screens, or broken bones, you can see where the problem is, but you don't know much about it. What we really want to know is how the tissue is behaving and then if we treat it, whether that treatment is doing what it's supposed to do. So those are the new challenges.
Ben - So that's almost a bit like comparing an MRI of the brain to an fMRI. The fMRI tells you where the activity is. The MRI just tells you what shape it is and then you're looking to take those steps in cancer.
Martin - That's right, yeah.
Ben - How can you do that?
Martin - Well one of the techniques we've been working on recently allows us to look at the diffusion of water molecules in tissues and this is quite a neat technique particularly from a physics point of view, but with MRI, we can actually measure how far water molecules have moved or diffused in tissues over a period of time. So if you've got a rapidly growing tumour, all of the cells are clustered very closely together and they effectively squeeze all the water out. If you then successfully kill a lot of those cells, you then get spaces in between the cells and a lot more water and therefore, the water molecules can move much further. We can measure that now and this gives us a way of picking up tumours, detecting them, but also, it helps us to see how far they've spread throughout the body. So we're working now on top of that to try and develop computer methods to measure how much disease there is in the body and then when we've got new treatments that are hoping to reverse the spread of disease, we should be able to detect whether that's working or not.
Ben - So you essentially take a baseline for a patient and then you'd know how the water travels. Either, I guess you could compare healthy tissue with unhealthy tissue or you just know how it started out and then months down the line, you can see how it responds.
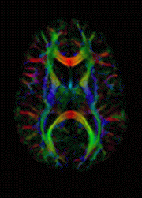 Martin - That's right, yeah. Usually, we do baseline scans and then do follow up scans so we can look in the same tumour.
Martin - That's right, yeah. Usually, we do baseline scans and then do follow up scans so we can look in the same tumour.
Ben - You've mentioned the very interesting physics about this but clearly, computer scientists also have a very large role to play in how we actually understand and interpret that information.
Martin - Yeah. A large part of our work is developing analysis methods to analyse and present the data in a simple way. So this is giving us new tools. In fact, to look at some of the heterogeneity in tumours so that's the fact that some of the tumour cells are driven by one pathway and then perhaps some of the cells have adapted to be driven by a different pathway, and we can begin to try and pick up those differences with a range of imaging techniques.
Ben - So this is really looking at the chemistry, not just looking at the structure, but actually seeing what chemical reactions are taking place. I guess that also will help you to lead and advise clinicians on what treatments might be appropriate.
Martin - Well that's what we're aiming for. In this year of personalised medicine that was discussed earlier, we want to be able to monitor individual treatments, identify what may be the most appropriate treatment and particularly identify if that treatment isn't working any longer and we need to switch. So that's an important area and chemical techniques are central to that. So we've had positron emission tomography for quite a long time and that allows us to probe some important pathways in the cells, but the problem with that is that we can't tell how the molecule we're looking at, and it's a probe molecule, has changed. Cells of course are all about interacting with molecules and making them do different things. So we're now working on a new technique called Dynamic Nuclear Polarisation (DNP) and this essentially is going to make MRI much more sensitive and allow us to look at very small levels of molecules. So, with MRI, we normally look at water and we see only about 7 molecules in every million. So if you thought about the UK population of 60 million, we would be seeing about 400 people. With DNP, we can take that up to about 10 million people. So, that gives us the possibility of looking at molecules that are only present in relatively small amounts and one of those that we're looking at is pyruvate. That's a very important intermediate in the energy supply to the cell, and is very much influenced by cancer. So we're already finding, looking at some of the new treatments that have been developed at the Institute of Cancer Research that we can see changes in that metabolism at a very early stage in the application of the new treatment. And we hope that is then going to inform how we can look at those patients in early stage trials to test whether these new treatments are really working.
Ben - How do you go about taking an idea from the incredible physics that we have and then - you at the ICR, make sure that these things work, and that we know that we can see it - how does it then get into being used in medicine? Surely, all of the radiologists or whoever's role it is in the hospitals need to understand the new techniques in order to interpret them.
Martin - Yeah, well we go through a range of tests first before we try and take these techniques through to the clinic, but then what we try and do is bolt them on to early stage studies, or to routine studies that the patients are having where we can just add this on. It depends on the type of measurement we're doing, but we've introduced a number of these approaches successfully into early stage trials of new treatments. And that gives us feedback on whether they are actually useful imaging techniques and if they are, we then try and move to generalise those to other centres and that helps more clinicians understand how useful they are and gives us more evidence. So, we go through a series of trials and then clinicians will learn about them from conferences and the papers that are published.
Ben - Well thank you very much. That's Professor Martin Leach from the CRUK and EPSRC Cancer Imaging Centre.









Comments
Add a comment