Post by: hamdani yusuf on 27/05/2020 14:02:48
Post by: Halc on 27/05/2020 14:17:04
If heat is generated somewhere or comes through the incompletely insulated container, then there will be heat transferred from the water to the ice, which allows the ice water to maintain its 0°C temperature. This is how your ice water maintains constant temperature despite the glass sitting there on a hot day.
Post by: hamdani yusuf on 29/05/2020 08:26:04
Material Emissivity Coefficient - ε -
Water 0.993 - 0.998
Ice 0.98
Snow 0.969 - 0.997
Sand 0.949 - 0.962
Granite 0.898
https://www.engineeringtoolbox.com/radiation-heat-emissivity-d_432.html
It means water radiates more energy than ice at the same temperature. Doesn't it mean that they are in a dynamic equilibrium? some water molecules will turn into ice while some ice molecules turn into water?
Post by: Bored chemist on 29/05/2020 10:48:54
But it's not to do with the emissivity.
Conductivity and random thermal fluctuations will do the job.
Post by: Petrochemicals on 29/05/2020 14:54:38
In a sealed container, put some ice and water at 0 degree C (in atmospheric pressure). Is there a net heat exchange between them?By definition in your question, at 0c everything is the same temperature. Unless one changes temperature no. No ice can form or melt without the exchange of energy in the form of kinetic energy of the molecules. Non nil nein nada negagive niet nix.
Post by: hamdani yusuf on 03/06/2020 08:57:01
They are in a dynamic equilibrium (you can prove this with isotope tracing).Do you mean they exchange phase? How is the isotope tracing done?
Quote
But it's not to do with the emissivity.Do you think that emissivity has no effect here?
Conductivity and random thermal fluctuations will do the job.
Post by: hamdani yusuf on 03/06/2020 09:06:55
Post by: evan_au on 03/06/2020 09:48:56
Quote from: hamdani yusuf
How is the isotope tracing done?One way is to make the ice cubes from "heavy water" and the liquid water from normal "light" water.
- Heavy Water is made with Deuterium instead of Hydrogen; the two are easily distinguished with a mass spectrometer
If you hold the temperature constant, the ice won't all melt into the water, and the water won't all freeze into ice.
- But if you lift out the ice cube after a few minutes in the water, you will find that some of the heavy water has migrated from the ice into the liquid water, and some of the light water has migrated from the liquid water into the ice.
You may want to rerun the experiment with the roles reversed, as heavy water has a freezing point of 3.8C, compared to normal water at 0C.
It looks odd, but heavy water ice cubes don't float...
https://en.wikipedia.org/wiki/Heavy_water
Due to the difference in melting points of heavy water and normal water, it may be better to use heavy water which has normal hydrogen, but a heavier isotope of oxygen...
Post by: Bored chemist on 03/06/2020 10:29:42
Do you think that emissivity has no effect here?Certainly not much effect.
Ice dosn't emit much IR, and nor does cold water.
The emissivity and absorption are identical at any wavelength.
https://en.wikipedia.org/wiki/Kirchhoff%27s_law_of_thermal_radiation
And imagine that I had some water, and some ice (Initially at 0 C) enclosed in a reflector so that any radiation they emitted was contained until it was absorbed again.
They must both stay at 0C.
If they didn't, I could run a heat engine from the difference and get free energy.
On the other hand, the water and ice are in contact, so thermal conductivity is a good pathway for heat transfer- if there was going to be any.
Do you mean they exchange phase? How is the isotope tracing done?Freeze some tritium labeled water.
Put the radiolabeled ice in normal water at 0C.
Wait, and then measure the activity of the water phase.
It slowly becomes active.
The same thing can be used to show that "insoluble" solids exchance ions with the solution they arein.
By definition in your question, at 0c everything is the same temperature. Unless one changes temperature no. No ice can form or melt without the exchange of energy in the form of kinetic energy of the molecules. Non nil nein nada negagive niet nix.Fluctuations.
The temperature is a property of the ensemble of molecules, but individual molecules are allowed to have different KE from the average.
So the exchange is possible.
Incidentally, after someone has said the exchange does, in fact, take place, it's not wise to categorically say that it can't.
Post by: hamdani yusuf on 18/03/2022 07:55:12
In a sealed container, put some ice and water at 0 degree C (in atmospheric pressure). Is there a net heat exchange between them?Here's an idea. The container has a thermally conductive separator which split the container into two chambers. Put the ice into the left chamber, and water into the right chamber. Will there be any thermal energy transfer through the separator?
Post by: Bored chemist on 18/03/2022 09:02:08
Not many have heard of the zeroth law of thermodynamics.
https://en.wikipedia.org/wiki/Zeroth_law_of_thermodynamics
Since the ice and water are both in equilibrium with the barrier, they are in equilibrium with eachother.
That means their temperature is the same and there's no temperature gradient for heat to pass"down".
Post by: hamdani yusuf on 18/03/2022 10:20:53
Fluctuations.
The temperature is a property of the ensemble of molecules, but individual molecules are allowed to have different KE from the average.
How thin the barrier can be until energy exchange from fluctuation can no longer take place?
Post by: Bored chemist on 18/03/2022 10:26:37
Energy will be loaned and returned by random molecular movements and vibrations regardless of whether there's a barrier there or not.
But temperature is only defined for an ensemble of molecules.
Post by: hamdani yusuf on 18/03/2022 11:54:36
At one point on the barrier, energy flows from water side to ice side. What prevents the ice molecules touching the barrier from transferring the energy it just received to another molecules further from the barrier?
Post by: Bored chemist on 18/03/2022 12:59:14
What prevents the ice molecules touching the barrier from transferring the energy it just received to another molecules further from the barrier?Nothing.
Nor will anything prevent energy transfer the other way.
So the net effect will be nothing.
Post by: hamdani yusuf on 19/03/2022 16:22:39
Ice melting is an endothermic process. The molecule undergoing this phase transition has local temperature lower than its surroundings where no phase transition is occurring. Since ice side has more ice than water side, there will be more endothermic process there. Energy will flow until equilibrium is achieved.What prevents the ice molecules touching the barrier from transferring the energy it just received to another molecules further from the barrier?Nothing.
Nor will anything prevent energy transfer the other way.
So the net effect will be nothing.
Post by: Origin on 19/03/2022 16:43:05
Ice melting is an endothermic process. The molecule undergoing this phase transition has local temperature lower than its surroundings where no phase transition is occurringLike you said melting is endothermic, but there is no temperature difference so there is no bulk heat flow and hence no bulk melting. This has been said multiple time in multiple ways, so I am not sure where your problem is.
Post by: Bored chemist on 19/03/2022 17:32:30
Energy will flow until equilibrium is achieved.It was already at equilibrium.
Post by: gem on 20/03/2022 01:21:44
Hamdani you raise a interesting scenario,
BC
Energy will flow until equilibrium is achieved.It was already at equilibrium.
I am not sure that statement is totally correct, if you consider the energy imbalance between the two sides as described,
for example the latent heat/energy is unbalanced as is the kinetic energy of the molecules, also the density's.
Post by: hamdani yusuf on 20/03/2022 04:07:55
I'd like to add that freezing is an exothermic process before I continue. In a mixture of water and ice in equilibrium, melting and freezing happen at the same rate.Ice melting is an endothermic process. The molecule undergoing this phase transition has local temperature lower than its surroundings where no phase transition is occurringLike you said melting is endothermic, but there is no temperature difference so there is no bulk heat flow and hence no bulk melting. This has been said multiple time in multiple ways, so I am not sure where your problem is.
The key concepts here are fluctuation and local temperature difference. In the side where there's more ice, melting occurs more often than freezing. On the other hand, in the side where there's more water, freezing occurs more often than melting.
Post by: Bored chemist on 20/03/2022 09:45:31
The key concepts here are fluctuationWhat fluctuations?
How long do they last?
Post by: hamdani yusuf on 20/03/2022 10:23:17
Kinetic energy of each molecule is supposed to fluctuate over time.The key concepts here are fluctuationWhat fluctuations?
How long do they last?
The fluctuation keeps going even without phase transition.
Post by: Bored chemist on 20/03/2022 22:19:21
How long do they last?
Post by: hamdani yusuf on 20/03/2022 22:26:17
as long as the system exists.How long do they last?
Post by: Bored chemist on 20/03/2022 22:30:08
Post by: hamdani yusuf on 20/03/2022 22:39:12
How long does each fluctuation last?does it really matter?
Post by: Bored chemist on 21/03/2022 09:08:23
If if didn't matter I wouldn't be asking you for a fourth time...How long does each fluctuation last?does it really matter?
Post by: hamdani yusuf on 21/03/2022 11:17:13
What difference would it make if it lasts for less than 1 nanosecond instead of more than 1 ns?If if didn't matter I wouldn't be asking you for a fourth time...How long does each fluctuation last?does it really matter?
Post by: chiralSPO on 22/03/2022 12:01:25
What difference would it make if it lasts for less than 1 nanosecond instead of more than 1 ns?If we were to view the system at such a fine resolution that we could observe extremely short fluctuations (ie 10–20 seconds), then the ice could be interpreted as being in equilibrium with superheated plasma (due to the Heisenberg Uncertainty relationship of time vs energy, which can be demonstrated as physically "real" and meaningful by the broad spectrum of ultrafast lasers).
As has been pointed out numerous times in this thread: temperature and equilibrium are emergent properties of large systems, and deal with things like averages. You will get into trouble by trying to extend these concepts to individual molecules. There isn't any meaningful definition of temperature for a single molecule. Likewise, there is no meaningful difference between solid or liquid for a single molecule.
We can look at the whole bulk system (many molecules, with macroscopic spatial and temporal resolution) and talk about temperature, equilibrium, phase, entropy, enthalpy etc., using classical physics/thermodynamics.
Or we can take a molecular/atomistic view, and look at small collections of molecules, in which case we will need to use quantum theories.
We can't try applying both models—this leads to nonsensical answers.
Post by: alancalverd on 22/03/2022 14:33:20
Post by: Eternal Student on 22/03/2022 23:22:18
If I recall, there was a situation described by someone (Hamdani?) earlier.
There was a container insulated to the outside world but with a barrier between two inner regions. The barrier allowed thermal contact between the two regions. Region I was filled with mainly ice (and let's say some liquid water to ensure good thermal contact) at 0 deg C. Meanwhile, Region II was filled with mainly liquid water at 0 deg C. So everything is held at 0 deg. C on a macroscopic scale.
If the barrier is a perfect insulator then the reasonable expectation of microscopic random interactions is that some melting and some freezing will occur in each region, it's just that overall there's no net change. However, the barrier is a problem because it can pass energy over to the other region if a phase change occurs close to the barrier.
There is more ice in region I so, just by the assumption of random phase shifts on a microscopic scale, melting happens more often in region I than in region II (and conversely freezing is more frequent in region II than region I). Over time, I would expect an equilibrium to be reached where there is an equal proportion of ice and liquid in both regions.
That does mean that a significant proportion of the originally liquid region has frozen while a significant amount of the icy region has melted: There has been a net transfer of energy (latent heat) from one region to the other.
(I've never actually done the experiment, just seems reasonable).
Also, on a minor note: Water changes density when freezing. I've been ignoring pressure and volume changes.
Best Wishes.
Post by: Bored chemist on 23/03/2022 12:56:40
Given that all the molecules in a crystal are moving, how long does it take before you know if it has melted?What difference would it make if it lasts for less than 1 nanosecond instead of more than 1 ns?If if didn't matter I wouldn't be asking you for a fourth time...How long does each fluctuation last?does it really matter?
Do you know that diffusion takes place in solids?
Post by: hamdani yusuf on 23/03/2022 14:07:32
There will always be molecular interchange at the interface between ice and water because the ensemble is not at 0K, but as you have defined them at the same temperature, there will be no NET exchange of energy.Yes, but there's different phase between left and right side of the separator, hence difference in internal energy.
A rotating magnet and an aluminum disk can have the same initial temperature. But when they are brought close to each other, some temperature increase is observed on the aluminum disk.
Post by: hamdani yusuf on 23/03/2022 14:10:07
We can look at the whole bulk system (many molecules, with macroscopic spatial and temporal resolution) and talk about temperature, equilibrium, phase, entropy, enthalpy etc., using classical physics/thermodynamics.But we can observe the changes in the system, if any.
Or we can take a molecular/atomistic view, and look at small collections of molecules, in which case we will need to use quantum theories.
We can't try applying both models—this leads to nonsensical answers.
It's either some ice in the left side melts or it doesn't.
It's either some water in the right side freezes or it doesn't.
Post by: hamdani yusuf on 23/03/2022 14:19:44
Given that all the molecules in a crystal are moving, how long does it take before you know if it has melted?If there is energy flow, then after an hour or so I should be able to see some water in the left side of the container, which is previously filled with ice. Some ice would be formed in the right side.
If there is no energy flow, then after a day I should only still find ice in the left side of the container. The right side should still contain water.
Post by: hamdani yusuf on 23/03/2022 14:27:35
There is more ice in region I so, just by the assumption of random phase shifts on a microscopic scale, melting happens more often in region I than in region II (and conversely freezing is more frequent in region II than region I). Over time, I would expect an equilibrium to be reached where there is an equal proportion of ice and liquid in both regions.It seems reasonable to me too.
That does mean that a significant proportion of the originally liquid region has frozen while a significant amount of the icy region has melted: There has been a net transfer of energy (latent heat) from one region to the other.
(I've never actually done the experiment, just seems reasonable).
Also, on a minor note: Water changes density when freezing. I've been ignoring pressure and volume changes.Consider the container is flexible enough to keep in touch with its contents, hence maintaining the pressure while the volume is changing.
Post by: Eternal Student on 23/03/2022 17:07:14
Consider the container is flexible enough to keep in touch with its contents, hence maintaining the pressure while the volume is changing.Sadly, that's still a minor issue. Work, pΔV, is done on a system if there's a volume change ΔV while maintaining constant pressure p.
If you want to avoid doing work on either region (and effectively changing the internal energy of the material in that region), then I think you must have a rigid container where the volume is held constant. Inevitably then the pressure of both regions will start to change and this will oppose the freezing in region II and the melting in region I. However, the effect probably won't be too important. Overall the proportion of ice-to-liquid won't end up being exactly the same in both regions BUT there will still have been some net freezing in region II and net melting in region I.
- - - - - - - - - - - - -
I think the point made by others ( @Bored chemist and @chiralSPO for example) is good and worth mentioning again. Temperature is an extremely difficult thing to define. It's a macroscopic property not a microscopic property. As such temperature and most of thermodynamics only applies to large scales and average actions of particles in large bodies.
In practice, a barrier that allows idealised thermal energy transmission is a practical impossibility. A real barrier allows energy flow based solely on the local regions that are in the vicinity of the barrier. It would allow a net flow of energy when there is some localised atypical region next to the barrier (causing what looks like a local temperature gradient from one side to the other). You can't physically build a suitable barrier that will sample the wide-scale temperature of everything in region I and compare that to the wide-scale temperature in region II and only allows energy to pass if those wide-scale temperatures show a difference.
So, yes, in practice you can get a net transfer of energy from an icy region to a liquid region when they are at the same temperature but only because the barrier is not idealised and a temperature is just a wide-scale average property, it does not prevent a localised atypical patch forming close to the barrier.
Best Wishes.
Post by: hamdani yusuf on 24/03/2022 03:03:07
Sadly, that's still a minor issue. Work, pΔV, is done on a system if there's a volume change ΔV while maintaining constant pressure p.Where does the energy come from, and where does it go to?
Here is what I found in Wikipedia. Does it also apply to liquid and solid?
Quote
https://en.wikipedia.org/wiki/Work_(physics)#Work_by_a_gas
(https://wikimedia.org/api/rest_v1/media/math/render/svg/ceac3c0def499fb2a60f691d1a964aa686186192)
Where P is pressure, V is volume, and a and b are initial and final volumes.
Post by: hamdani yusuf on 24/03/2022 04:24:01
Temperature is an extremely difficult thing to define. It's a macroscopic property not a microscopic property. As such temperature and most of thermodynamics only applies to large scales and average actions of particles in large bodies.If we want to be pedantic, many other physical parameters are also hard to define, such as pressure and electric current. Even fundamental parameters such as mass, length, and time are also hard to define.
But we can continue using current definitions, even some obsolete ones, if they are useful.
Post by: Bored chemist on 24/03/2022 12:07:03
If it takes an hour to notice any melting, but the fluctuations only last a nanosecond, will you notice any melting due to the fluctuations?Given that all the molecules in a crystal are moving, how long does it take before you know if it has melted?If there is energy flow, then after an hour or so I should be able to see some water in the left side of the container, which is previously filled with ice. Some ice would be formed in the right side.
If there is no energy flow, then after a day I should only still find ice in the left side of the container. The right side should still contain water.
What I really can't understand is how you didn't realise that this was important.
Post by: hamdani yusuf on 24/03/2022 12:12:21
Let's say that initially, the ice is -0.1°C while the water is 0.1°C. Temperature difference will cause energy transfer from water side to ice side until they have the same temperature, 0°C in this case. What will happen next? Will the energy transfer suddenly stop? Or it continues until reaching equilibrium, where both sides have the same water/ice ratio?
Post by: Bored chemist on 24/03/2022 12:35:15
In practice, it's hard to get 100% ice or 100% water at exactly 0°C.Not really. Get some ice, a little below freezing , put it in a closed container and put that in a slurry of crushed ice and water.
It will warm up to exactly 0C
Get some water, just above freezing, Seal it in a container and put it in a slurry of ice and water, it will cool down to exactly 0C.
Will the energy transfer suddenly stop?No, it will gradually stop.
After a while, the difference will be smaller than the random thermal variation.
Post by: hamdani yusuf on 24/03/2022 14:02:17
If it takes an hour to notice any melting, but the fluctuations only last a nanosecond, will you notice any melting due to the fluctuations?We will notice the melting and freezing after the imbalanced fluctuations accumulate in both sides. If each fluctuation lasts longer, then there will be fewer number of them in an hour, and vice versa.
What I really can't understand is how you didn't realise that this was important.
Post by: Eternal Student on 24/03/2022 15:02:06
ES Said: Sadly, that's still a minor issue. Work, pΔV, is done on a system if there's a volume change ΔV while maintaining constant pressure p.
Hamdani replied: Where does the energy come from, and where does it go to?
Where the container expands, work is done on the container by the system (the water region with net freezing). The container walls acquire the energy, the system loses that energy. If it's a container of the type you described (something elastic that tried to maintain a a constant pressure) most of the energy in the walls would be described as elastic potential. In the more general case, we don't care what form the energy takes in the walls of the container, it's usually heat which ultimately escapes to the outer environment. All that's important is that the system has done work on something external to the system.
Opposite idea where the container walls contract. Here the external environment has done work on the system.
Here is what I found in Wikipedia. Does it also apply to liquid and solid?Yes, for a container that maintains constant pressure, which is what you suggested, that integral reduces to p.ΔV.
Quote
(Integral formula appeared here)
This is some work done on the environment by the system (or vice versa if its negative). There can also be other ways of doing work on the system but that doesn't matter. Changing the volume of the container while the system was exerting pressure on the container walls is certainly one way. It doesn't matter if the system is a mixture of solid and liquid, an ideal gas, or something else.
Best Wishes.
Post by: Bored chemist on 24/03/2022 15:12:06
We will notice the melting and freezing after the imbalanced fluctuations accumulate in both sides.Why do you imagine that the fluctuations are not balanced?
Do you not understand what equilibrium means?
Post by: hamdani yusuf on 24/03/2022 15:49:04
Not really. Get some ice, a little below freezing , put it in a closed container and put that in a slurry of crushed ice and water.What's hard is getting the left compartment filled with pure ice while the right compartment contains pure water. We will need to maintain air temperature at 0°C, so is the tools we use to transport the water and ice. The lighting should also be taken into account, as well as body temperature of the experimenter.
It will warm up to exactly 0C
Get some water, just above freezing, Seal it in a container and put it in a slurry of ice and water, it will cool down to exactly 0C.
Post by: Bored chemist on 24/03/2022 16:01:00
It is easy.Not really. Get some ice, a little below freezing , put it in a closed container and put that in a slurry of crushed ice and water.What's hard is getting the left compartment filled with pure ice while the right compartment contains pure water. We will need to maintain air temperature at 0°C, so is the tools we use to transport the water and ice. The lighting should also be taken into account, as well as body temperature of the experimenter.
It will warm up to exactly 0C
Get some water, just above freezing, Seal it in a container and put it in a slurry of ice and water, it will cool down to exactly 0C.
You do everything in a tank of ice cold water.
Post by: hamdani yusuf on 25/03/2022 04:40:26
It is easy.Easy to say, but hard to do. Otherwise, someone must have done it already, and we can just watch the video.
You do everything in a tank of ice cold water.
Post by: alancalverd on 25/03/2022 15:52:52
Post by: Bored chemist on 25/03/2022 17:22:11
Otherwise, someone must have done it alreadyHow the **** do you think we know what the melting point of water is?
Messieurs Laplace and Lavoisier didn't video it, because they did the experiment in 1782.
https://www.nature.com/scitable/content/ice-calorimeter-developed-by-lavoisier-and-laplace-14898943/
Post by: hamdani yusuf on 27/03/2022 13:13:01
This sort of experiment has been done hundreds of times, to establish the triple point and phase diagram of water.How are the results? Are they consistent with each other?
Does the ice side still free from water, and the water side free from ice?
Post by: Bored chemist on 27/03/2022 13:17:03
How are the results?Extremely good.
Are they consistent with each other?Obviously, yes.
Does the ice side still free from water, and the water side free from ice?Yes.
Do you realise that you are asking "Does basic science work?"?
Post by: Bored chemist on 27/03/2022 13:21:34
https://isotech.co.uk/wp-content/uploads/2020/09/The-Establishment-of-ITS-90-Water-Triple-Point-References-to-%C2%B12uK-and-the-Assessment-of-100-Water-Triple-Point-Cells-Made-Between-2001-and-2006.pdf
Post by: hamdani yusuf on 27/03/2022 13:52:50
Did the experiment initially separate the ice from water?Otherwise, someone must have done it alreadyHow the **** do you think we know what the melting point of water is?
Messieurs Laplace and Lavoisier didn't video it, because they did the experiment in 1782.
https://www.nature.com/scitable/content/ice-calorimeter-developed-by-lavoisier-and-laplace-14898943/
Post by: alancalverd on 27/03/2022 15:05:15
A rotating magnet and an aluminum disk can have the same initial temperature. But when they are brought close to each other, some temperature increase is observed on the aluminum disk.because you are supplying energy to rotate the magnet. If you don't, no heat is generated.
Post by: hamdani yusuf on 27/03/2022 22:44:50
What if it's the aluminum disk which was initially rotating, while the magnet was stationary? I think that energy transfer and conversion still happen. Similar case would happen if the aluminum disk was attached to a spring and vibrate at resonance frequency.A rotating magnet and an aluminum disk can have the same initial temperature. But when they are brought close to each other, some temperature increase is observed on the aluminum disk.because you are supplying energy to rotate the magnet. If you don't, no heat is generated.
My point is that equality in temperature doesn't guarantee the absent of energy transfer, especially when some other forms of energy are involved.
Post by: Bored chemist on 27/03/2022 22:50:49
What if it's the aluminum disk which was initially rotating,then you would be supplying energy.
My point is that equality in temperature doesn't guarantee the absent of energy transfer, especially when some other forms of energy are involved.It guarantees that you don't get heat transfer.
Post by: alancalverd on 28/03/2022 07:41:08
What if it's the aluminum disk which was initially rotating, while the magnet was stationary?Makes no difference - all the disc "sees" is relative rotation.
Post by: hamdani yusuf on 28/03/2022 13:06:57
Similar case would happen if the aluminum disk was attached to a spring and vibrate at resonance frequency.What if the magnet is replaced by an electret?
Post by: Bored chemist on 28/03/2022 13:08:16
You still would not be talking about exchange of heat, so it still wouldn't be anything to do with the title of the thread.Similar case would happen if the aluminum disk was attached to a spring and vibrate at resonance frequency.What if the magnet is replaced by an electret?
Post by: hamdani yusuf on 28/03/2022 15:56:20
You still would not be talking about exchange of heat, so it still wouldn't be anything to do with the title of the thread.What does it take to say that there is heat exchange in a process?
Post by: alancalverd on 28/03/2022 17:46:05
Post by: Eternal Student on 29/03/2022 02:30:02
What does it take to say that there is heat exchange in a process?
What level of answer did you want?
General principles:
Heat isn't a substance or a property that some matter can have. A system can have a temperature or some internal energy but it does not have anything you can call "heat". It should be possible to identify two systems if there is a heat flow, energy flows out of one system and into the other system. While the energy is in transfer, that is "heat". This exchange of energy is commonly called a heat flow or heat exchange.
Medium level answer:
Heat or "heat flow" describes the natural and automatic transfer of energy from a body at a higher temperature to one of lower temperature when they come into thermal contact.
Two bodies are in thermal contact if there is atleast one path along which heat can flow.
You will then focus in on a precise definition of heat by either: excluding certain energy transfers OR you'll take a constructive approach - start with an empty set of permitted energy transfers and directly specify the mechanisms of energy transfer that will be included in the definition of heat flow.
The Exclusion method to define heat exchange:
Certain forms of energy transfer are specifically excluded. Heat should not be thermodynamic work done on a system. Also there should not be a direct transfer of matter (which might have some internal energy) from one system to the other.
The constructive method (start from nothing and inlcude things):
The mechanisms of energy transfer that define heat include conduction, through direct contact of immobile bodies, or through a wall or barrier that is impermeable to matter; or radiation between separated bodies; or friction due to isochoric mechanical or electrical or magnetic or gravitational work done by the surroundings on the system of interest....
... (Convection might be included but Wikipedia choose to exclude it and argue that although it's an intermediate process, the ultimate flow of energy is achieved by conduction or radiation once the fluid has has been carried to a particular location).
[Both these approaches are based on the Wikipedia entry for heat and some text is a direct quotation].
Best Wishes.
Post by: hamdani yusuf on 29/03/2022 04:44:37
Certain forms of energy transfer are specifically excluded. Heat should not be thermodynamic work done on a system. Also there should not be a direct transfer of matter (which might have some internal energy) from one system to the other.Can you give some examples?
Post by: hamdani yusuf on 29/03/2022 04:48:42
The mechanisms of energy transfer that define heat include conduction, through direct contact of immobile bodies, or through a wall or barrier that is impermeable to matter; or radiation between separated bodies; or friction due to isochoric mechanical or electrical or magnetic or gravitational work done by the surroundings on the system of interest....Heat conduction can be seen as near field radiation. It's extremely rare case for two ordinary objects to collide at atomic level.
... (Convection might be included but Wikipedia choose to exclude it and argue that although it's an intermediate process, the ultimate flow of energy is achieved by conduction or radiation once the fluid has has been carried to a particular location).
Post by: Eternal Student on 29/03/2022 07:45:05
Can you give some examples?There's not much point. There are text books and other sources of information if you're really interested. We've also already spent some time discussing work done by (or on) a system which shows a volume change.
What seems more relevant is just to jump straight to a higher level answer:
"Temperature", "thermal contact" and "heat" are all very difficult things to define. At least, they are difficult to define or explain at any microscopic scale (where you're trying to take a reductionist approach and break things down to the smallest indivisible units like particles with well defined properties). However, they can be easily defined (or really just decalred to exist) on a macroscopic scale. This is the usual development of the subject matter that is called "thermodynamics".
For example, if you try to take a microscopic definition for "heat" then it is not supposed to include a transfer of matter from one system to another BUT a transfer of photons such as infra-red radiation is something we would very much like to include in the definition of heat. What makes a photon different to some other particle with energy? If an electron and positron cross a barrier then that would seem to be a transfer of matter across the barrier and would not be considered as a heat transfer. However, if they annhilate on one side of the barrier, only some photons cross the barrier and then the photons interact with some nucleus on the other side of the barrier to reform particles and anti-particles - that would seem to be just fine.
Best Wishes.
Post by: alancalverd on 29/03/2022 10:56:54
Heat conduction can be seen as near field radiation. It's extremely rare case for two ordinary objects to collide at atomic level.No. "Radiation" excludes energy transfer by collision conduction or convection. Words have very precise meanings in physics, which is why some of us despise priests, politicians and philosophers. And sloppy journalists.
Post by: hamdani yusuf on 29/03/2022 11:42:52
Water and ice have very precise meanings in physics. It doesn't mean that they can't have the same substance. What I wrote there is comparable to saying that ice is just frozen water.Heat conduction can be seen as near field radiation. It's extremely rare case for two ordinary objects to collide at atomic level.No. "Radiation" excludes energy transfer by collision conduction or convection. Words have very precise meanings in physics, which is why some of us despise priests, politicians and philosophers. And sloppy journalists.
Radio wave, microwave, visible light, and X ray are also manifestations of the same phenomenon.
Post by: Bored chemist on 29/03/2022 11:47:43
Water and ice have very precise meanings in physics.Not if you are talking about a single molecule.
That's part of the problem.
Post by: Bored chemist on 29/03/2022 11:51:42
It's extremely rare case for two ordinary objects to collide at atomic level.In the condensed phase (liquid, gas or dense SCF) they are already in contact.
Even in the gas phase, collision frequencies are very high- GHz for ordinary air.
Post by: hamdani yusuf on 29/03/2022 11:54:53
Is conduction have a precise meaning for a single molecule?Water and ice have very precise meanings in physics.Not if you are talking about a single molecule.
That's part of the problem.
Post by: hamdani yusuf on 29/03/2022 12:01:48
What's the maximum distance between two molecules where they are said to be still in contact?It's extremely rare case for two ordinary objects to collide at atomic level.In the condensed phase (liquid, gas or dense SCF) they are already in contact.
Even in the gas phase, collision frequencies are very high- GHz for ordinary air.
I was referring to collisions between atoms from two adjacent macroscopic solid objects.
Post by: Bored chemist on 29/03/2022 12:54:31
No.Is conduction have a precise meaning for a single molecule?Water and ice have very precise meanings in physics.Not if you are talking about a single molecule.
That's part of the problem.
But energy transfer from one molecule to a second by collision is different from energy transfer by radiation.
Post by: Bored chemist on 29/03/2022 12:56:05
What's the maximum distance between two molecules where they are said to be still in contact?It's hard to say, but if one mechanically transfers energy to the other they can't be far apart.
Post by: Eternal Student on 29/03/2022 15:01:51
"What is temperature"
https://www.thenakedscientists.com/forum/index.php?topic=80604.80
Post by: alancalverd on 29/03/2022 19:44:00
What I wrote there is comparable to saying that ice is just frozen water.No, it's very different. Ice is indeed one (actually several) macroscopic phase of an ensemble of H2O molecules, but radiation and conduction are entirely different phenomena.
The rate of heat exchange between two bodies by conduction depends on the temperature difference Th - Tl. The rate of heat exchange by radiation depends on Th4 - Tl4.
Post by: hamdani yusuf on 30/03/2022 12:33:35
Imagine two parallel square metal plates, 1 m² each. First plate has temperature of 200K, while second plate is 400K. Initial distance is 10 meters. Measure heat exchange rate, and record as P0.What I wrote there is comparable to saying that ice is just frozen water.No, it's very different. Ice is indeed one (actually several) macroscopic phase of an ensemble of H2O molecules, but radiation and conduction are entirely different phenomena.
The rate of heat exchange between two bodies by conduction depends on the temperature difference Th - Tl. The rate of heat exchange by radiation depends on Th4 - Tl4.
Reduce the distance to 1 meter, or 10% of previous distance. Measure and record the heat exchange as P1. Repeat the process until those plates are considered touching.
Will we find abrupt change or discontinuity in the record? At what point?
Post by: Bored chemist on 30/03/2022 12:40:19
Will we find abrupt change or discontinuity in the record?Yes
At what point?
When they touch.
There will be a very sharp increase just before they actually touch- at the distance when electron tunnelling becomes significant.
Post by: hamdani yusuf on 30/03/2022 13:49:04
Is there any experimental evidence for that?Will we find abrupt change or discontinuity in the record?YesAt what point?
When they touch.
There will be a very sharp increase just before they actually touch- at the distance when electron tunnelling becomes significant.
Post by: alancalverd on 30/03/2022 17:24:48
If 0 < s < d we approach a "near field" radiative exchange that is almost independent of s.
If s = 0 the exchange is conductive
It's all pretty standard stuff in radiation physics and thermal engineering.
Post by: hamdani yusuf on 31/03/2022 04:30:46
If the separation s exceeds twice the diameter d, we are in "far field" where the rate of exchange by radiation tends towards a 1/s2 law.It looks like rough estimation to me.
If 0 < s < d we approach a "near field" radiative exchange that is almost independent of s.
If s = 0 the exchange is conductive
It's all pretty standard stuff in radiation physics and thermal engineering.
What's the tolerance for s = 0? Even when two objects are in contact, there will still be some gap between them in microscopic level.
How much is the discontinuity around the transition between radiative and conductive heat transfer?
Quote
https://en.wikipedia.org/wiki/Thermal_contact_conductance
(https://upload.wikimedia.org/wikipedia/commons/thumb/a/af/Contact_conductance.svg/443px-Contact_conductance.svg.png)
Post by: Bored chemist on 31/03/2022 08:39:02
Is there any experimental evidence for that?Put your finger near- and then on- a block of ice.
Post by: alancalverd on 31/03/2022 10:04:40
What's the tolerance for s = 0?For the present context, it's the point at which their molecules can exchange vibrational kinetic energy without an intermediate photon. Hence the change from T4 to T law.
Post by: hamdani yusuf on 31/03/2022 10:40:19
Even in a crystal lattice, exchange of kinetic energy between one atom to the other occurs as electromagnetic interaction. Nuclear interaction is rare in a standard ambient environment.What's the tolerance for s = 0?For the present context, it's the point at which their molecules can exchange vibrational kinetic energy without an intermediate photon. Hence the change from T4 to T law.
Post by: Bored chemist on 31/03/2022 10:48:21
Are you talking about the exchange of VIRTUAL photons as the mechanism for electromagnetic forces?Even in a crystal lattice, exchange of kinetic energy between one atom to the other occurs as electromagnetic interaction. Nuclear interaction is rare in a standard ambient environment.What's the tolerance for s = 0?For the present context, it's the point at which their molecules can exchange vibrational kinetic energy without an intermediate photon. Hence the change from T4 to T law.
That's not going to help here.
Post by: alancalverd on 31/03/2022 12:41:18
Even in a crystal lattice, exchange of kinetic energy between one atom to the other occurs as electromagnetic interaction.No, electrostatic.
Post by: hamdani yusuf on 01/04/2022 10:34:20
Those electrons are moving, at least in microscopic scale, hence not static.Even in a crystal lattice, exchange of kinetic energy between one atom to the other occurs as electromagnetic interaction.No, electrostatic.
AFAIK, even a single electron has magnetic properties. What prevents them to interact electromagnetically?
Post by: alancalverd on 01/04/2022 10:39:39
Post by: hamdani yusuf on 01/04/2022 10:41:36
When the finger barely touches the ice, the contact area is infinitesimally close to 0. But if we push forward, the contact area will become larger.Is there any experimental evidence for that?Put your finger near- and then on- a block of ice.
(https://upload.wikimedia.org/wikipedia/commons/thumb/a/af/Contact_conductance.svg/443px-Contact_conductance.svg.png)
T2A gets closer to T2B when the gap between A and B is shrinking. How would the discontinuity of energy transfer appear in the graph?
Post by: hamdani yusuf on 01/04/2022 10:43:09
so why make it complicatedTo make it more accurate for wider range of circumstances.
Post by: Bored chemist on 01/04/2022 11:23:53
As far as I can tell, that picture is wrong.If the separation s exceeds twice the diameter d, we are in "far field" where the rate of exchange by radiation tends towards a 1/s2 law.It looks like rough estimation to me.
If 0 < s < d we approach a "near field" radiative exchange that is almost independent of s.
If s = 0 the exchange is conductive
It's all pretty standard stuff in radiation physics and thermal engineering.
What's the tolerance for s = 0? Even when two objects are in contact, there will still be some gap between them in microscopic level.
How much is the discontinuity around the transition between radiative and conductive heat transfer?Quotehttps://en.wikipedia.org/wiki/Thermal_contact_conductance
(https://upload.wikimedia.org/wikipedia/commons/thumb/a/af/Contact_conductance.svg/443px-Contact_conductance.svg.png)
Post by: alancalverd on 01/04/2022 11:49:48
Adding nonsense doesn't improve accuracy. This is physics, not politics, philosophy or religion.so why make it complicatedTo make it more accurate for wider range of circumstances.
Post by: hamdani yusuf on 02/04/2022 08:27:22
As far as I can tell, that picture is wrong.How can we make it right? Which parts should we change, remove, or add?
Post by: hamdani yusuf on 02/04/2022 08:31:43
Adding nonsense doesn't improve accuracy. This is physics, not politics, philosophy or religion.What's the nonsense that I should remove without impairing accuracy?
Post by: alancalverd on 02/04/2022 12:02:26
Post by: hamdani yusuf on 03/04/2022 04:19:55
The notion of electromagnetic interactions determining heat transfer by conduction.What are the fundamental interactions/forces involved in heat transfer by conduction?
If it's not an electromagnetic interaction, you can choose the others: weak force, strong force, gravitation.
Post by: hamdani yusuf on 03/04/2022 04:22:12
Here's the picture, just in case you have trouble finding it.As far as I can tell, that picture is wrong.How can we make it right? Which parts should we change, remove, or add?
(https://upload.wikimedia.org/wikipedia/commons/thumb/a/af/Contact_conductance.svg/443px-Contact_conductance.svg.png)
Post by: Bored chemist on 03/04/2022 10:34:58
Things are in contact or they are not.
The picture shows what you get if there's a gap between the two objects.
Post by: Bored chemist on 03/04/2022 10:36:33
The notion of electromagnetic interactions determining heat transfer by conduction.What are the fundamental interactions/forces involved in heat transfer by conduction?
If it's not an electromagnetic interaction, you can choose the others: weak force, strong force, gravitation.
Are you talking about the exchange of VIRTUAL photons as the mechanism for electromagnetic forces?
That's not going to help here.
Post by: hamdani yusuf on 03/04/2022 12:18:10
What would help?The notion of electromagnetic interactions determining heat transfer by conduction.What are the fundamental interactions/forces involved in heat transfer by conduction?
If it's not an electromagnetic interaction, you can choose the others: weak force, strong force, gravitation.Are you talking about the exchange of VIRTUAL photons as the mechanism for electromagnetic forces?
That's not going to help here.
Post by: alancalverd on 03/04/2022 17:08:55
Post by: hamdani yusuf on 04/04/2022 02:34:05
Acceptance of a ball-and-spring electrostatic model of intermolecular energy exchange.The model might work for solid objects. How does it work for liquid, gas, or plasma?
Post by: alancalverd on 04/04/2022 10:35:03
Quote
However, because of the large difference in mass between electrons and ions, their temperatures may be different, sometimes significantly so. This is especially common in weakly ionized technological plasmas, where the ions are often near the ambient temperature while electrons reach thousands of kelvin. The opposite case is the z-pinch plasma where the ion temperature may exceed that of electrons.(Wikipedia).
A good friend measures the temperature of plasmas in tokamaks by studying the black body emission spectrum.
Post by: Spamessays on 05/04/2022 01:51:00
Post by: hamdani yusuf on 05/04/2022 09:03:06
A good friend measures the temperature of plasmas in tokamaks by studying the black body emission spectrum.Did the plasma produce black body radiation spectra?
Post by: hamdani yusuf on 05/04/2022 09:08:52
Very loose springs for liquids, billiard balls for gases.Ok. So the ball and spring model only works for solid. I don't think it is useful to answer the question in the title of this thread.
Post by: Bored chemist on 05/04/2022 17:00:46
Ok. So the ball and spring model only works for solid.Not really.
It works OK for liquids too. It's also useful for gases.
Post by: alancalverd on 06/04/2022 17:07:42
It's ideal. Ice floats because the springs are rigid and tetrahedrally disposed, water is denser because the springs are slack and the liquid is long-range disordered. The absorption spectrum of water vapor is enormous and complicated because H2O forms all sorts of temporary polymers that work as various-sized sticky billiard balls with weak springs joining the strongly-bonded and not-very-symmetric individual molecules.Very loose springs for liquids, billiard balls for gases.Ok. So the ball and spring model only works for solid. I don't think it is useful to answer the question in the title of this thread.
Post by: luplay on 05/05/2022 10:40:47
I have learned a lot.
Post by: hamdani yusuf on 06/05/2022 11:04:11
useful threadInteresting. What have you learned?
I have learned a lot.
Do you think there's a net heat exchange between water and ice at 0 degree C?
Post by: alancalverd on 07/05/2022 10:11:22
Post by: hamdani yusuf on 08/05/2022 09:19:58
I hope not - that would destroy the whole of thermodynamics.Or perhaps less dramatically, your understanding of thermodynamics.
Has anyone ever actually did the experiment? What's the step by step procedure to minimize sources of errors, and get reliable result to answer the question?
Post by: Bored chemist on 08/05/2022 10:24:34
Has anyone ever actually did the experiment?Which experiment?
Do you realise that every experiment in thermodynamics requires that the zeroth law works.
If your idea was right then no experiment would work
Post by: alancalverd on 08/05/2022 15:14:58
Temperature is the mean internal kinetic energy of a body.
Heat flows from a hotter body (one with a higher temperature) to a cooler one.
If the opposite were true, we could extract an infinite amount of energy from any two bodies since the heat could flow from A to B then back to A. There is no evidence that this can happen and it would contradict our definition of energy as a conserved quantity.
Therefore we must conclude that no heat can flow between bodies at the same temperature.
Post by: hamdani yusuf on 10/05/2022 05:01:00
Which experiment?The experiment described in the first post.
Here's my idea to minimize noise over signal:
- Prepare 50/50 ice-water mixture at around 0°C in a large plastic bowl. Let it in refrigerator for an hour to reach equilibrium.
- Fill a metal cup with 90% water and 10% ice from the mixture.
- Fill another metal cup with 10% water and 90% ice from the mixture.
- Put both metal cups into the bowl containing the remaining of the mixture.
- Let them in refrigerator for an hour to reach equilibrium.
- See the result, if the ratio of ice-water in the cups change.
What do we expect if there is a net heat exchange?
What do we expect if there is no net heat exchange?
Do you think this experiment can provide the answer?
Is there something need to be done to avoid erroneous conclusion?
Post by: hamdani yusuf on 10/05/2022 05:18:17
Let's go back to square one.
Temperature is the mean internal kinetic energy of a body.
Heat flows from a hotter body (one with a higher temperature) to a cooler one.
If the opposite were true, we could extract an infinite amount of energy from any two bodies since the heat could flow from A to B then back to A. There is no evidence that this can happen and it would contradict our definition of energy as a conserved quantity.
Therefore we must conclude that no heat can flow between bodies at the same temperature.
Eternal Student has answered the question which is different than yours. Where do you think he got it wrong?
Hi.
If I recall, there was a situation described by someone (Hamdani?) earlier.
There was a container insulated to the outside world but with a barrier between two inner regions. The barrier allowed thermal contact between the two regions. Region I was filled with mainly ice (and let's say some liquid water to ensure good thermal contact) at 0 deg C. Meanwhile, Region II was filled with mainly liquid water at 0 deg C. So everything is held at 0 deg. C on a macroscopic scale.
If the barrier is a perfect insulator then the reasonable expectation of microscopic random interactions is that some melting and some freezing will occur in each region, it's just that overall there's no net change. However, the barrier is a problem because it can pass energy over to the other region if a phase change occurs close to the barrier.
There is more ice in region I so, just by the assumption of random phase shifts on a microscopic scale, melting happens more often in region I than in region II (and conversely freezing is more frequent in region II than region I). Over time, I would expect an equilibrium to be reached where there is an equal proportion of ice and liquid in both regions.
That does mean that a significant proportion of the originally liquid region has frozen while a significant amount of the icy region has melted: There has been a net transfer of energy (latent heat) from one region to the other.
(I've never actually done the experiment, just seems reasonable).
Also, on a minor note: Water changes density when freezing. I've been ignoring pressure and volume changes.
Best Wishes.
And Here's mine.
I'd like to add that freezing is an exothermic process before I continue. In a mixture of water and ice in equilibrium, melting and freezing happen at the same rate.Ice melting is an endothermic process. The molecule undergoing this phase transition has local temperature lower than its surroundings where no phase transition is occurringLike you said melting is endothermic, but there is no temperature difference so there is no bulk heat flow and hence no bulk melting. This has been said multiple time in multiple ways, so I am not sure where your problem is.
The key concepts here are fluctuation and local temperature difference. In the side where there's more ice, melting occurs more often than freezing. On the other hand, in the side where there's more water, freezing occurs more often than melting.
Post by: alancalverd on 10/05/2022 09:00:37
Eternal Student has answered the question which is different than yours. Where do you think he got it wrong?
Quote from: Eternal Student on 22/03/2022 23:22:18
Hi.
If I recall, there was a situation described by someone (Hamdani?) earlier.
There was a container insulated to the outside world but with a barrier between two inner regions. The barrier allowed thermal contact between the two regions. Region I was filled with mainly ice (and let's say some liquid water to ensure good thermal contact) at 0 deg C. Meanwhile, Region II was filled with mainly liquid water at 0 deg C. So everything is held at 0 deg. C on a macroscopic scale.
If the barrier is a perfect insulator then the reasonable expectation of microscopic random interactions is that some melting and some freezing will occur in each region, it's just that overall there's no net change. However, the barrier is a problem because it can pass energy over to the other region if a phase change occurs close to the barrier.
Alas, no. Melting and freezing require an exchange of heat because the potential energy of the two states is different. If you are in the ice block, there is no adjacent area at a higher temperature therefore no heat input. If you are in the water puddle, there is no adjacent area at a lower temperature therefore no heat loss.
Post by: hamdani yusuf on 10/05/2022 12:27:43
Alas, no. Melting and freezing require an exchange of heat because the potential energy of the two states is different. If you are in the ice block, there is no adjacent area at a higher temperature therefore no heat input. If you are in the water puddle, there is no adjacent area at a lower temperature therefore no heat loss.What if you are at the interface between water and ice?
Or at the metal surface between ice-rich side and water-rich side?
What do you think about my planned experiment?
Here's my idea to minimize noise over signal:
- Prepare 50/50 ice-water mixture at around 0°C in a large plastic bowl. Let it in refrigerator for an hour to reach equilibrium.
- Fill a metal cup with 90% water and 10% ice from the mixture.
- Fill another metal cup with 10% water and 90% ice from the mixture.
- Put both metal cups into the bowl containing the remaining of the mixture.
- Let them in refrigerator for an hour to reach equilibrium.
- See the result, if the ratio of ice-water in the cups change.
What do we expect if there is a net heat exchange?
What do we expect if there is no net heat exchange?
Do you think this experiment can provide the answer?
Is there something need to be done to avoid erroneous conclusion?
Post by: Bored chemist on 10/05/2022 13:05:56
What if you are at the interface between water and ice?Then there is no temperature difference to drive the transfer of energy.
See the result, if the ratio of ice-water in the cups change.It won't.
Because, that would require the transfer of heat to or from teh ice and, because everything is at the same temperature, there is no impetus to drive the energy from one place to another.
Why do you not understand and accept this?
Which bit does not make sense to you?
Post by: alancalverd on 10/05/2022 13:06:10
What if you are at the interface between water and ice?Wherever you are in the entire universe, heat can only flow from a hot body (one at a higher temperature) to a cold one.
Or at the metal surface between ice-rich side and water-rich side?
BC and ES will leap on this by saying "what happens when a cold planet emits a thermal photon that travels towards the sun?" but the magic word in the OP is "net" and the definition of temperature: a statistic of large numbers, not the descriptor of a single event.
The problem with your experiment (I've done similar things with ice calorimeters to measure radiation dose) is the anomalous convection of water. Recorded in detail by Darwin but nowadays known as the M'pemba effect (though M'pemba could not possibly have conducted the experiment as reported), bulk water starts moving as it freezes.
Post by: Eternal Student on 11/05/2022 00:00:23
I've only just seen that I was mentioned recently....
Eternal Student has answered the question which is different than yours. Where do you think he got it wrong?Probably spending time on the forum instead of doing my housework, I would have thought.
However @alancalverd highlighted something like this:
the reasonable expectation of microscopic random interactions is that some melting and some freezing will occur in each region, it's just that overall there's no net change...With the remark.... "Alas, No..."
Radioisotope tracers can be used. I think @Bored chemist mentioned it some long time ago. Put tritium enriched water ice cubes into some liquid water all kept at 0 deg C (as best as you can). Some of the radio-isotope moves out of the ice and into the liquid. Some melting and also some freezing does seem to be happening at a microscopic level, it's just that overall or in terms of "net" changes there aren't any. However, the ice particles are not the same ice particles you started with.
Temperature tells you very little about the action and interaction of individual particles, you know they have a range of energies and sometimes interactions occur. If only you could talk about very local temperatures - then ice will melt if the local liquid is just above 0 deg C. Meanwhile, on average, some equivalent amount of liquid is locally below 0 deg C and soon joins to the existing solid ice and freezes. So there's no net change. However, it's best not to even try and consider "temperature" to be something that is sensibly defined at such small and local levels. It's preferable to say the liquid-ice mixture retained a global temperature of 0 deg C but individual groups of particles had a range of energies and some interactions occurred where particles left the ice phase and entered the liquid phase (and vice versa).
You (@alancalverd) were technically right to say no heat flowed. Heat is usually defined as the flow of energy from one place to another (by natural processes like conduction etc.) due to a temperature difference. The energy didn't flow from a thing with a higher temperature to one of lower temperature, it wasn't really possible to give that microscopic element a temperature at all. Energy (not heat) certainly was taken from some particles and given to others though. Moreover, thermodynamics doesn't say anything about energy never being exchanged between two bodies at the same temperature, they can exchange, of course they can, just so long as the input equals the output and there is no net exchange of energy.
Best Wishes.
Post by: alancalverd on 11/05/2022 00:16:26
However, it's best not to even try and consider "temperature" to be something that is sensibly defined at such small and local levels...precisely because, like life, it is not a property of individual particles but of a very large ensemble of particles, i.e. a classical mesoscopic "body".
Post by: Eternal Student on 11/05/2022 00:59:43
There's an odd overlap going on here between two related threads.
..precisely because, like life, it is not a property of individual particles but of a very large ensemble of particles, i.e. a classical mesoscopic "body"...
I think that is going in the right direction for this thread**. I don't know but I can only imagine the OP is trying to identify the deeper meaning of temperature and, as is often done in science, they are trying for a reductionist approach. E.g. What is temperature at the most fundamental level? It's probably nothing at all, or just an abstract property as suggested in the approach taken by theoretical temperature scales. It is usefully left as a macroscopic or emergent property that a body can have.
LATE EDITING: ** Possibly for the other thread called "What is temperature" and I forgot I was writing on this one. Let's say it's going in the right direction for some of the assumed purpose for some of the related threads.
Best Wishes.
Post by: alancalverd on 11/05/2022 10:39:37
Diffusion and isotope exchange don't imply energy flow, at least to the first order. If we have two gases with identical molecular masses at the same temperature and bring them into contact, they will diffuse together. If there was a significant difference in their molecular masses we might be able to separate them with a centrifuge but we would be adding energy to the system and there's no a priori equipartition so the concept of temperature would not apply to the whole.
Now given that your ice/water mixture is in thermal equilibrium, for every T2O molecule that moves in one direction we expect on average to see an H2O moving in the opposite direction at a slightly greater speed but conveying the same amount of kinetic energy, so that the internal k.e. of both ice and water remains constant: that's the definition of thermal equilibrium.
Post by: Eternal Student on 11/05/2022 11:38:20
Not sure where this diffusion conversation is going but it seems ok.
If it's relevant, I think my much earlier comment ( N pages back but recently re-quoted by @hamdani yusuf ) about more ice melting in a region where there is more ice may have been an unreasonable simplification. At about 0 deg. C, ice only tends to form where there is already a nucleation site. So where there is more ice we might expect there to be more localised melting but also there may be a similar increase in localised freezing at precisely the same surfaces and locations.
There may also be practical stuff and limits on reality, there usually is.
Best Wishes.
Post by: hamdani yusuf on 11/05/2022 14:48:27
It won't.Here's the list of possible results.
Because, that would require the transfer of heat to or from teh ice and, because everything is at the same temperature, there is no impetus to drive the energy from one place to another.
1. both cups retain their ice/water ratio.
2. both cups increase ice/water ratio
3. both cups decrease ice/water ratio
4. first cup retains while second cup increases ice/water ratio
5. first cup retains while second cup decreases ice/water ratio
6. first cup increases while second cup retains ice/water ratio
7. first cup decreases while second cup retains ice/water ratio
8. first cup increases while second cup decreases ice/water ratio
9. first cup decreases while second cup increases ice/water ratio
Which one is the most likely result?
Do you think that a real life experiment can settle our disagreement?
Post by: Origin on 11/05/2022 15:45:28
Which one is the most likely result?After 7 pages you still don't understand that there will not be a net heat exchange between water and ice at 0 degrees?
Do you think that a real life experiment can settle our disagreement?
Post by: alancalverd on 11/05/2022 17:04:59
There may also be practical stuff and limits on reality, there usually is.Especially with water. Our existence depends on its anomalous properties, and the need for nucleation in boiling and freezing just adds to the fun.
Post by: alancalverd on 11/05/2022 17:14:03
Do you think that a real life experiment can settle our disagreement?No. It will most likely demonstrate all sorts of anomalous behavior peculiar to water, and a whole lot of problems with your method. Sir Lawrence Bragg was a bit of an amateur, having only two Nobel Prizes to his name, but he used to say in his Royal Institution annual lecture demonstrations that if you get 20% repeatability in a heat experiment you are doing very well. Having mucked about with ice calorimetry at the UK National Physical Laboratory (long after Bragg retired as Director) I can only concur.
Why not just accept the obvious definitions of elementary thermodynamics that everyone else uses?
Post by: hamdani yusuf on 12/05/2022 05:18:07
No. It will most likely demonstrate all sorts of anomalous behavior peculiar to water, and a whole lot of problems with your method. Sir Lawrence Bragg was a bit of an amateur, having only two Nobel Prizes to his name, but he used to say in his Royal Institution annual lecture demonstrations that if you get 20% repeatability in a heat experiment you are doing very well. Having mucked about with ice calorimetry at the UK National Physical Laboratory (long after Bragg retired as Director) I can only concur.
Why not just accept the obvious definitions of elementary thermodynamics that everyone else uses?Because I want to avoid making false assumptions which can lead to unexpected results. I'm not sure with everyone else. But your suggestion would make science more dogmatic and harder to make corrections from previous errors.
Post by: hamdani yusuf on 12/05/2022 05:21:41
It shouldn't be hard for you to pick an answer as someone who already understand it? I guess you would pick number 1, but let me know if it's not the case, and why.Which one is the most likely result?After 7 pages you still don't understand that there will not be a net heat exchange between water and ice at 0 degrees?
Do you think that a real life experiment can settle our disagreement?
Post by: Bored chemist on 12/05/2022 08:51:03
Because I want to avoid making false assumptions which can lead to unexpected results.Thermodynamics has been working for about 200 years.
If there were any "unexpected results" we would have seen them by now.
But we don't.
Which is one reason why we know that the real world obeys the laws of thermodynamics.
The other reason, is that the conservation laws are mathematically proven to be true.
https://en.wikipedia.org/wiki/Noether%27s_theorem
So why do you continue to question something which has been seen to be true and proven can't be false?|
Are you trolling?
The other day you asked me to multiply 0.1 by 3 for you.
You also asked for video evidence that holding a lit match near a piece of charcoal won't set it on fire (and, by implication, said that it would if the ambient temperature was above 40C)
It's becoming increasingly clear that you are either trolling, or you just don't have the background to do science.
Post by: alancalverd on 12/05/2022 08:51:37
Because I want to avoid making false assumptionsA definition is not an assumption. Two objects are said to be at the same temperature if no heat can flow from one to the other.
Post by: Bored chemist on 12/05/2022 08:57:07
It shouldn't be hard for you to pick an answer as someone who already understand it?I did pick an answer.
The problem is that you can not , or will not understand what I wrote.
Quote from: hamdani yusuf on 10/05/2022 05:01:00
See the result, if the ratio of ice-water in the cups change.
It won't.
Only 1 of your outcomes is consistent with my reply.
This seems to be part of the problem. When people answer you, you don't understand that they have answered.
That's probably because you refuse to learn science.
Why do you do this?
Why do you make a fool of yourself here, rather than going to something like teh Khan academy and finding out?
Post by: hamdani yusuf on 12/05/2022 15:28:41
Astronomy has been working for centuries before Copernicus questioned it.
Thermodynamics has been working for about 200 years.
Post by: hamdani yusuf on 12/05/2022 15:45:34
Definitions involve assumptions.Because I want to avoid making false assumptionsA definition is not an assumption. Two objects are said to be at the same temperature if no heat can flow from one to the other.
Heat is a form of energy, instead of fluid.
Other forms of energy can be transferred in the absence of temperature difference.
Other forms of energy can be converted to heat and vice versa inside an object.
From the operation of chillers, we know that evaporation reduces temperature while condensation increase temperature of working fluid. By analogy, similar things may happen in melting and freezing.
The point of doing the real experiment is to see if local fluctuation can produce observable effects. It's okay if you are not willing to spend your time and resources to do the experiment, but don't discourage those who are. If you are right, you can use the result to support your position in future debates. On the other hand, if you're wrong, you can stop spreading false information. You have nothing to lose, and everything to gain.
Post by: hamdani yusuf on 12/05/2022 15:48:25
So why do you continue to question something which has been seen to be true and proven can't be false?|My response here is similar to my previous response to Alan.
Are you trolling?
The other day you asked me to multiply 0.1 by 3 for you.
You also asked for video evidence that holding a lit match near a piece of charcoal won't set it on fire (and, by implication, said that it would if the ambient temperature was above 40C)
It's becoming increasingly clear that you are either trolling, or you just don't have the background to do science.
Post by: hamdani yusuf on 12/05/2022 15:51:36
I did pick an answer.You quoted my response to Origin.
The problem is that you can not , or will not understand what I wrote.
Perhaps you can learn how to write better.
Post by: hamdani yusuf on 12/05/2022 16:09:15
Only 1 of your outcomes is consistent with my reply.Do you pick the first option, where the ice/water ratio don't change in both cups? Implicit answer may be used as an excuse to evade responsibility for giving a false but definitive answer.
This seems to be part of the problem. When people answer you, you don't understand that they have answered.
That's probably because you refuse to learn science.
Why do you do this?
Why do you make a fool of yourself here, rather than going to something like teh Khan academy and finding out?
Post by: hamdani yusuf on 12/05/2022 16:20:05
Let me make the first move. I'm on Veritasium's side. I'll explain my reasoning later.It crossed my mind to do similar thing here, although perhaps with lower and affordable stake. Since the thanks seems to be valuable here, perhaps we can bet so members in the losing side give thanks to the posts of members in the winning sides (up to n posts). I think it's an interesting idea.
Will someone give it a try to show their prowess in physics?
But then my curiosity creeped and I start doing the experiment I described previously. So it won't be fair to other members.
Post by: Origin on 12/05/2022 16:51:08
You quoted my response to Origin.Why did you even ask the question? I said there is no net heat transfer!
Perhaps you can learn how to write better.
The way you go on and on over the same ground is so annoying, that is why I put several of your threads on ignore. It is a ridiculous waste of time.
Post by: Bored chemist on 12/05/2022 18:03:02
No, it hadn't. the predictions were wrong.Astronomy has been working for centuries before Copernicus questioned it.
Thermodynamics has been working for about 200 years.
Not very wrong, but still wrong.
Post by: Bored chemist on 12/05/2022 18:03:48
Did you consider that the problem was your failure to read?I did pick an answer.You quoted my response to Origin.
The problem is that you can not , or will not understand what I wrote.
Perhaps you can learn how to write better.
Post by: Bored chemist on 12/05/2022 18:05:59
In another thread, a scientific disagreement was finally resolved by making a bet.In the UK it is illegal to bet on events where the outcome is already known.
We have repeatedly pointed out that the outcome of your experiment is already known.
That's a pity in a way; it might be fun to take your money.
Post by: Bored chemist on 12/05/2022 18:08:16
Implicit answer may be used as an excuse to evade responsibility for giving a false but definitive answer.Or it might be used to try to shoehorn some information into you.
But since you seem to be too dim to recognise reality anyway, I will make it simple.
Yes. your first option is still the only one that can actually happen.
Post by: hamdani yusuf on 12/05/2022 23:09:45
Because you didn't pick an answer explicitly. You just repeated a statement from a scientific theory, which may or may not accurately represent real life experiment. You can learn something from Alan's answer.You quoted my response to Origin.Why did you even ask the question? I said there is no net heat transfer!
Perhaps you can learn how to write better.
The way you go on and on over the same ground is so annoying, that is why I put several of your threads on ignore. It is a ridiculous waste of time.
But not the last suggestion.Do you think that a real life experiment can settle our disagreement?No. It will most likely demonstrate all sorts of anomalous behavior peculiar to water, and a whole lot of problems with your method. Sir Lawrence Bragg was a bit of an amateur, having only two Nobel Prizes to his name, but he used to say in his Royal Institution annual lecture demonstrations that if you get 20% repeatability in a heat experiment you are doing very well. Having mucked about with ice calorimetry at the UK National Physical Laboratory (long after Bragg retired as Director) I can only concur.
Why not just accept the obvious definitions of elementary thermodynamics that everyone else uses?
Post by: Bored chemist on 13/05/2022 13:08:04
You would learn a lot if you followed his last suggestion.Because you didn't pick an answer explicitly. You just repeated a statement from a scientific theory, which may or may not accurately represent real life experiment. You can learn something from Alan's answer.You quoted my response to Origin.Why did you even ask the question? I said there is no net heat transfer!
Perhaps you can learn how to write better.
The way you go on and on over the same ground is so annoying, that is why I put several of your threads on ignore. It is a ridiculous waste of time.But not the last suggestion.Do you think that a real life experiment can settle our disagreement?No. It will most likely demonstrate all sorts of anomalous behavior peculiar to water, and a whole lot of problems with your method. Sir Lawrence Bragg was a bit of an amateur, having only two Nobel Prizes to his name, but he used to say in his Royal Institution annual lecture demonstrations that if you get 20% repeatability in a heat experiment you are doing very well. Having mucked about with ice calorimetry at the UK National Physical Laboratory (long after Bragg retired as Director) I can only concur.
Why not just accept the obvious definitions of elementary thermodynamics that everyone else uses?
Post by: hamdani yusuf on 13/05/2022 13:54:48
Let me share an advice from a successful person. If you think that you are the smartest person in a room, it's most likely that you are in a wrong room.Implicit answer may be used as an excuse to evade responsibility for giving a false but definitive answer.Or it might be used to try to shoehorn some information into you.
But since you seem to be too dim to recognise reality anyway, I will make it simple.
Yes. your first option is still the only one that can actually happen.
Your attitude towards someone that you think is less intelligent than you needs to be improved. If you think that they made a mistake, just tell them what the mistake is, and suggest how to make it right. Telling that they are inferior than you is unnecessary. It can make them think that scientists are generally arrogant, which drives them away from learning proper science.
Post by: hamdani yusuf on 13/05/2022 13:58:37
You would learn a lot if you followed his last suggestion.I think I can learn a lot more if I know how thermodynamics was conceptualized from experimental results, and how currently accepted theory has beaten its competitors and alternatives.
Post by: Origin on 13/05/2022 14:01:55
Quote
Your attitude towards someone that you think is less intelligent than you needs to be improved. If you think that they made a mistake, just tell them what the mistake is, and suggest how to make it right. Telling that they are inferior than you is unnecessary.The negative comments are probably due to your posting style. You ask a question and people take the time to answer you and then you ignore the answer. That is going to lead to people ignoring you or getting frustrated with you. I suggest you change your attitude or expect negative comments.
Post by: hamdani yusuf on 13/05/2022 14:28:49
Here's my idea to minimize noise over signal:OK. I've finished my first round of experiment as described above. But I can't get the metal cups, so I just used ordinary drinking glasses. I guess the heat conductance is enough for this experiment since they are quite thin.
- Prepare 50/50 ice-water mixture at around 0°C in a large plastic bowl. Let it in refrigerator for an hour to reach equilibrium.
- Fill a metal cup with 90% water and 10% ice from the mixture.
- Fill another metal cup with 10% water and 90% ice from the mixture.
- Put both metal cups into the bowl containing the remaining of the mixture.
- Let them in refrigerator for an hour to reach equilibrium.
- See the result, if the ratio of ice-water in the cups change.
The temperature of the refrigerator is 4C, as shown by a thermometer left there for an hour. The result is the ice in both glasses decreased from initial ratio.
So I moved the system to the freezer, which is kept at -4C, as measured by a thermometer left there for an hour. The result is the ice in both glasses increased from initial ratio.
These results show that energy transfer between the system and the environment overwhelmed the energy transfered through the glasses. It means that the noise over signal ratio is too high to get reliable conclusion. Hence the experimental setups need to be improved.
I also learned that gravity and buoyancy play significant role in determining the results, which need to be addressed in upcoming experiments.
Post by: hamdani yusuf on 13/05/2022 14:45:25
The negative comments are probably due to your posting style. You ask a question and people take the time to answer you and then you ignore the answer. That is going to lead to people ignoring you or getting frustrated with you. I suggest you change your attitude or expect negative comments.I didn't ask what thermodynamics theory says. I can google it and quickly get an answer.
I asked what would happen if I do an experiment as described previously. What currently existing theories predict, and what factors can significantly affect the results.
Post by: Origin on 13/05/2022 14:49:40
I didn't ask what thermodynamics theory says. I can google it and quickly get an answer.Which you were told and you ignored.
I asked what would happen if I do an experiment as described previously. What currently existing theories predict, and what factors can significantly affect the results.
Post by: Origin on 13/05/2022 15:22:36
I also learned that gravity and buoyancy play significant role in determining the results, which need to be addressed in upcoming experiments.What you should have learned is that heat transfer is driven by the delta T.
Post by: hamdani yusuf on 13/05/2022 15:39:22
Did you make correct prediction?I didn't ask what thermodynamics theory says. I can google it and quickly get an answer.Which you were told and you ignored.
I asked what would happen if I do an experiment as described previously. What currently existing theories predict, and what factors can significantly affect the results.
Post by: hamdani yusuf on 13/05/2022 15:40:40
I already knew that. What I want to know is, what else drives heat transfer?I also learned that gravity and buoyancy play significant role in determining the results, which need to be addressed in upcoming experiments.What you should have learned is that heat transfer is driven by the delta T.
Post by: Origin on 13/05/2022 16:14:40
Did you make correct prediction?Yes, I predicted your experiment would fail because there is going to be a delta T between the environment and your ice bath since you do not have the equipment to accurately hold the temp at exactly 0C.
I already knew that. What I want to know is, what else drives heat transfer?If there is no delta T, then nothing else matters since there will be no heat transfer.
Post by: hamdani yusuf on 13/05/2022 16:21:09
I didn't propose to involve money.In another thread, a scientific disagreement was finally resolved by making a bet.In the UK it is illegal to bet on events where the outcome is already known.
We have repeatedly pointed out that the outcome of your experiment is already known.
That's a pity in a way; it might be fun to take your money.
Post by: hamdani yusuf on 13/05/2022 16:27:01
Your picked answer is different than what the actual results are.Did you make correct prediction?Yes, I predicted your experiment would fail because there is going to be a delta T between the environment and your ice bath since you do not have the equipment to accurately hold the temp at exactly 0C.I already knew that. What I want to know is, what else drives heat transfer?If there is no delta T, then nothing else matters since there will be no heat transfer.
Do you have an idea how to get the first possible result, i.e. both cups retain their ice/water ratio?
Post by: Origin on 13/05/2022 17:21:27
Your picked answer is different than what the actual results are.Of course! Why are you asking, haven't you read your own thread?
Do you have an idea how to get the first possible result, i.e. both cups retain their ice/water ratio?
Post by: Bored chemist on 13/05/2022 17:55:51
Your picked answer is different than what the actual results are.Where are the details of the experiment that gave you these "actual answers"?
Post by: alancalverd on 13/05/2022 19:02:44
Definitions involve assumptions.Wrong. A definition is an absolute statement that creates truth.
A cow is defined as a mature female bovine quadruped. No assumptions involved.
Post by: hamdani yusuf on 14/05/2022 12:15:37
Of course! Why are you asking, haven't you read your own thread?
Do you have an idea how to get the first possible result, i.e. both cups retain their ice/water ratio?
Post by: hamdani yusuf on 14/05/2022 12:17:54
Your picked answer is different than what the actual results are.Where are the details of the experiment that gave you these "actual answers"?
Here's my idea to minimize noise over signal:OK. I've finished my first round of experiment as described above. But I can't get the metal cups, so I just used ordinary drinking glasses. I guess the heat conductance is enough for this experiment since they are quite thin.
- Prepare 50/50 ice-water mixture at around 0°C in a large plastic bowl. Let it in refrigerator for an hour to reach equilibrium.
- Fill a metal cup with 90% water and 10% ice from the mixture.
- Fill another metal cup with 10% water and 90% ice from the mixture.
- Put both metal cups into the bowl containing the remaining of the mixture.
- Let them in refrigerator for an hour to reach equilibrium.
- See the result, if the ratio of ice-water in the cups change.
The temperature of the refrigerator is 4C, as shown by a thermometer left there for an hour. The result is the ice in both glasses decreased from initial ratio.
So I moved the system to the freezer, which is kept at -4C, as measured by a thermometer left there for an hour. The result is the ice in both glasses increased from initial ratio.
These results show that energy transfer between the system and the environment overwhelmed the energy transfered through the glasses. It means that the noise over signal ratio is too high to get reliable conclusion. Hence the experimental setups need to be improved.
I also learned that gravity and buoyancy play significant role in determining the results, which need to be addressed in upcoming experiments.
Post by: hamdani yusuf on 14/05/2022 12:22:37
You assume that someone who read your definition knows and agrees with the meaning of mature, female, bovine, and quadruped.Definitions involve assumptions.Wrong. A definition is an absolute statement that creates truth.
A cow is defined as a mature female bovine quadruped. No assumptions involved.
Post by: Bored chemist on 14/05/2022 12:41:19
Nope; the definition of "cow" is still correct, regardless of whether it is understood.You assume that someone who read your definition knows and agrees with the meaning of mature, female, bovine, and quadruped.Definitions involve assumptions.Wrong. A definition is an absolute statement that creates truth.
A cow is defined as a mature female bovine quadruped. No assumptions involved.
The same is true of "flash point".
The ignorance or knowledge of the reader isn't relevant to the definition.
Post by: hamdani yusuf on 14/05/2022 13:10:39
I also learned that gravity and buoyancy play significant role in determining the results, which need to be addressed in upcoming experiments.It turns out that ice-water bath is not adequate to keep the outer sides of the glasses at 0C. The ice tends to float, which makes lower part of the bath warmer, even when it's only a few centimeters deep. This temperature difference seems to cause the heat transfer through the glass.
So, obviously the next step to improve the experiment setup is to keep the ice at the bottom of the bath. Convection will make temperature of the bath more uniform around 0C.
Post by: hamdani yusuf on 14/05/2022 13:12:57
Nope; the definition of "cow" is still correct, regardless of whether it is understood.Have you heard that someone defined pi = 3?
The same is true of "flash point".
The ignorance or knowledge of the reader isn't relevant to the definition.
Post by: Origin on 14/05/2022 14:52:20
Have you heard that someone defined pi = 3?I'm beginning to think you enjoy the feeling of being confused. You now seem to not even know what a definition is. I don't get you at all.
Post by: Bored chemist on 14/05/2022 15:18:57
No.Nope; the definition of "cow" is still correct, regardless of whether it is understood.Have you heard that someone defined pi = 3?
The same is true of "flash point".
The ignorance or knowledge of the reader isn't relevant to the definition.
I'm aware that someone tried to.
https://en.wikipedia.org/wiki/Indiana_Pi_Bill
But reality stopped them.
Why did you think that was relevant?
Post by: alancalverd on 14/05/2022 16:11:15
I'm beginning to think you enjoy the feeling of being confused. You now seem to not even know what a definition is. I don't get you at all.The object of philosophy is to tell people that they don't (or even can't) understand the obvious. I think HY is an undercover philosopher.
Post by: alancalverd on 14/05/2022 16:12:07
Convection will make temperature of the bath more uniform around 0C.No. Convection requires a temperature gradient.
Post by: hamdani yusuf on 19/05/2022 12:47:50
It seems like you missed to read this claim.Have you heard that someone defined pi = 3?I'm beginning to think you enjoy the feeling of being confused. You now seem to not even know what a definition is. I don't get you at all.
Wrong. A definition is an absolute statement that creates truth.That's why you didn't get it.
Post by: Bored chemist on 19/05/2022 12:59:47
Did you notice that nobody actually defined pi as 3?
Post by: hamdani yusuf on 19/05/2022 13:01:21
The object of philosophy is to tell people that they don't (or even can't) understand the obvious. I think HY is an undercover philosopher.It looks like you need to read the introduction to philosophy. Here's a book recommendation I got in my twitter feed.
T H E GR E AT
CONVERSATION
A Historical Introduction to Philosophy
EIGHTH EDITION
NORMAN MELCHERT
Professor Emeritus, Lehigh University
DAVID R. MORROW
Visiting Fellow, George Mason University
Here's a part of the foreword.
Quote
One of the authors of this book had a teacher—
a short, white-haired, elderly gentleman with a thick German accent—who used to say, “Whether
you will philosophize or won’t philosophize, you
must philosophize.” By this, he meant that we can’t
help making decisions about these crucial matters.
We make them either well or badly, conscious
of what we are doing or just stumbling along.
Post by: hamdani yusuf on 19/05/2022 13:15:57
That's what I found as experimental results. Ice-water mixture don't maintain homogeneous temperature. Difference in density tends to make bottom part of the bath warmer than the surface.Convection will make temperature of the bath more uniform around 0C.No. Convection requires a temperature gradient.
Post by: Bored chemist on 20/05/2022 10:48:03
Difference in density tends to make bottom part of the bath warmer than the surface.You can only make stuff warmer by adding energy, so that finding can not be relevant to the discussion.
Post by: alancalverd on 20/05/2022 13:05:55
That's what I found as experimental results.Told you so.
Quote
Ice-water mixture don't maintain homogeneous temperature. Difference in density tends to make bottom part of the bath warmer than the surface.No, difference in temperature makes one part warmer than the other, and as water is denser than ice, the warmer bit sinks to the bottom.
It is interesting to speculate whether life would have evolved, or what it would look like, if ice was denser than water.
Post by: hamdani yusuf on 20/05/2022 15:02:45
May be it's not relevant to the title or theoretical consideration. But it's relevant if we want to get experimental evidence.Difference in density tends to make bottom part of the bath warmer than the surface.You can only make stuff warmer by adding energy, so that finding can not be relevant to the discussion.
Post by: Bored chemist on 21/05/2022 12:20:04
You will not get experimental evidence of what happens at 0C (as per the thread title) unless your equipment is at 0C.May be it's not relevant to the title or theoretical consideration. But it's relevant if we want to get experimental evidence.Difference in density tends to make bottom part of the bath warmer than the surface.You can only make stuff warmer by adding energy, so that finding can not be relevant to the discussion.
Post by: Origin on 21/05/2022 14:29:43
May be it's not relevant to the title or theoretical consideration. But it's relevant if we want to get experimental evidence.A poorly designed and executed experiment gives meaningless results.
Post by: hamdani yusuf on 23/05/2022 03:28:57
You will not get experimental evidence of what happens at 0C (as per the thread title) unless your equipment is at 0C.It shows that the experimental evidence is not that easy to get, contrary to your previous claim.
It is easy.Not really. Get some ice, a little below freezing , put it in a closed container and put that in a slurry of crushed ice and water.What's hard is getting the left compartment filled with pure ice while the right compartment contains pure water. We will need to maintain air temperature at 0°C, so is the tools we use to transport the water and ice. The lighting should also be taken into account, as well as body temperature of the experimenter.
It will warm up to exactly 0C
Get some water, just above freezing, Seal it in a container and put it in a slurry of ice and water, it will cool down to exactly 0C.
You do everything in a tank of ice cold water.
Post by: hamdani yusuf on 23/05/2022 03:30:14
It's inconclusive, but not meaningless. It tells us that something must be improved.May be it's not relevant to the title or theoretical consideration. But it's relevant if we want to get experimental evidence.A poorly designed and executed experiment gives meaningless results.
You never fail until you stop trying.
Post by: alancalverd on 23/05/2022 18:35:16
Post by: hamdani yusuf on 24/05/2022 03:45:40
Sadly, many people have attempted to disprove the laws of thermodynamics, with increasingly sophisticated apparatus. The goal is unlimited wealth and omnipotence - you could make a whole new universe! But AFAIK nobody has succeeded.I don't think that my experiment would disprove the laws of thermodynamics. It only shows that average temperature of an object (ice-water mixture) can be different than local temperature at specific location in the object. In this case, it was caused mostly by buoyancy of ice in water.
Post by: Bored chemist on 24/05/2022 09:03:54
true, but your proposed system is not at equilibrium.Sadly, many people have attempted to disprove the laws of thermodynamics, with increasingly sophisticated apparatus. The goal is unlimited wealth and omnipotence - you could make a whole new universe! But AFAIK nobody has succeeded.I don't think that my experiment would disprove the laws of thermodynamics. It only shows that average temperature of an object (ice-water mixture) can be different than local temperature at specific location in the object. In this case, it was caused mostly by buoyancy of ice in water.
Post by: Bored chemist on 24/05/2022 09:04:52
If you keep trying something which has been proven to be impossible, then you are failing.It's inconclusive, but not meaningless. It tells us that something must be improved.May be it's not relevant to the title or theoretical consideration. But it's relevant if we want to get experimental evidence.A poorly designed and executed experiment gives meaningless results.
You never fail until you stop trying.
Post by: alancalverd on 24/05/2022 10:10:59
It only shows that average temperature of an object (ice-water mixture) can be different than local temperature at specific locationAn average being different from a sample? My god, he's invented statistics! There's a Nobel physics prize awaiting if you can demonstrate it, and Fields Medal if you can present a mathematical proof. Or maybe not.
Post by: hamdani yusuf on 25/05/2022 04:48:11
If you keep trying something which has been proven to be impossible, then you are failing.Do you mean mathematical proof? What is it?
Post by: Bored chemist on 25/05/2022 09:13:20
Do you mean mathematical proof? What is it?
Yes.
I mean the same mathematical proof that I already pointed out and which you ignored because you rfefuse to do science.
The other reason, is that the conservation laws are mathematically proven to be true.
https://en.wikipedia.org/wiki/Noether%27s_theorem
Post by: hamdani yusuf on 25/05/2022 12:47:00
What's the impossible thing do you think I tried to prove using my experimental setup described previously?Do you mean mathematical proof? What is it?
Yes.
I mean the same mathematical proof that I already pointed out and which you ignored because you rfefuse to do science.The other reason, is that the conservation laws are mathematically proven to be true.
https://en.wikipedia.org/wiki/Noether%27s_theorem
Post by: Bored chemist on 25/05/2022 13:14:33
They do.
Post by: hamdani yusuf on 25/05/2022 14:34:02
You seem to think that the laws of energy conservation don't work.What makes you think that way?
They do.
Here are my main points in this thread :
1. Heat flows from higher temperature object to lower temperature object.
2. Ice and water at melting point has the same average temperature, 0C.
3. At melting point, water has higher internal energy compared to ice.
4. Local temperature can be different from average temperature.
Is there any point you don't agree with?
Post by: Bored chemist on 25/05/2022 16:38:25
Here's an idea. The container has a thermally conductive separator which split the container into two chambers. Put the ice into the left chamber, and water into the right chamber. Will there be any thermal energy transfer through the separator?Saying stuff like that suggests that you don't understand the laws of thermodynamics.
Post by: alancalverd on 25/05/2022 20:31:28
If you can make water flow uphill, or indeed put the sh1t back in the donkey by any means, you are doing better than God.
Post by: hamdani yusuf on 26/05/2022 04:36:53
Or classical thermodynamics doesn't apply in some circumstances, just like Maxwell's electromagnetic theory. Which one is true can be resolved by experiments.Here's an idea. The container has a thermally conductive separator which split the container into two chambers. Put the ice into the left chamber, and water into the right chamber. Will there be any thermal energy transfer through the separator?Saying stuff like that suggests that you don't understand the laws of thermodynamics.
You can have an internally consistent mathematical model, but doesn't represent physical reality.
Post by: hamdani yusuf on 26/05/2022 04:51:06
Heat (i.e.thermal energy) can only flow from a higher to a lower temperature.That's true by definition. When energy flows from a lower temperature object to higher temperature object, we can simply call it something other than heat flow or just unnatural. Laser cutter heating a metal target or induction stove heating a steel pan are some examples.
Post by: alancalverd on 26/05/2022 06:38:16
Quotefrom: alancalverd on Yesterday at 20:31:28That's true by definition.
Heat (i.e.thermal energy) can only flow from a higher to a lower temperature.
So you agree that if ice and water are in contact at 0°C, no net heat will flow between them?
Post by: Bored chemist on 26/05/2022 09:07:36
You can have an internally consistent mathematical model, but doesn't represent physical reality.You could.
But nobody uses them to model reality, so they aren't relevant.
Post by: hamdani yusuf on 27/05/2022 13:07:08
So you agree that if ice and water are in contact at 0°C, no net heat will flow between them?Yes, at that specific point.
But a water body with 0°C average temperature has various local temperature. So does the ice. Moreover,
0°C ice can receive heat energy without changing its temperature, by melting.
0°C water can give away heat energy without changing its temperature, by freezing.
When I alternated the ambient temperature between -4°C and 4°C (by moving between refrigerator and freezer), the ice/water ratio of both containers tend to get closer to each other.
I used normal tap water to avoid complication with supercooling things. The freezing always starts from the water surface in the container. Warmer water (up to 4°C) tends to go to the bottom, due to its density.
Hence when temperature difference and phase separation due to buoyancy are not eliminated, both containers tend to end up with more similar ice/water ratio than when they were started.
The question is how can we eliminate them? How far can we eliminate them? Are there other factors that can generate local temperature difference?
Post by: Bored chemist on 27/05/2022 13:32:56
But a water body with 0°C average temperature has various local temperature. So does the ice. Moreover,And, on average, these cancel out which is why the answer to your question
0°C ice can receive heat energy without changing its temperature, by melting.
0°C water can give away heat energy without changing its temperature, by freezing.
"Is there a net heat exchange between water and ice at 0 degree C?"
Is still no- just like everyone has said all along.
Post by: alancalverd on 27/05/2022 13:39:32
But a water body with 0°C average temperature has various local temperature.Yet again, you have discovered the meaning of "average".
The freezing always starts from the water surface in the container. Warmer water (up to 4°C) tends to go to the bottom, due to its density.And the anomalous convection of cold water, without which life could not exist on this planet.
Are there other factors that can generate local temperature difference?The only thing that can change the temperature of a body is adding or removing energy. Local temperature fluctuations are inevitable in a body that is being heated or cooled anisotropically. In most cases the temperature gradient can be calculated from knowledge of the thermal diffusivity or convective behavior of the material but in the case of water and a few other liquids, you also need to know the starting temperature so you can anticipate anomalous convection or solid phase change.
To repeat Sir Lawrence Bragg's dictum: if you get better than ±20% in a heat experiment, you are doing very well. Been there, done that, spent 20 person-years getting to ± 0.1% in an amorphous, homogeneous solid.
Post by: hamdani yusuf on 27/05/2022 22:44:41
Yet again, you have discovered the meaning of "average".What's your interpretation?
Post by: hamdani yusuf on 27/05/2022 22:46:43
The only thing that can change the temperature of a body is adding or removing energy.Or changing from other forms of energy, such as chemical, mechanical, or electrical energy.
Post by: Bored chemist on 28/05/2022 00:48:12
Yet again, you have discovered the meaning of "average".That's the whole story here, neatly summarised by Alan.
Post by: hamdani yusuf on 28/05/2022 17:02:45
What do you think that average means? How can it be used to predict or explain my experimental results?Yet again, you have discovered the meaning of "average".That's the whole story here, neatly summarised by Alan.
Post by: Bored chemist on 28/05/2022 17:12:54
What do you think that average means?1.
a number expressing the central or typical value in a set of data, in particular the mode, median, or (most commonly) the mean, which is calculated by dividing the sum of the values in the set by their number.
https://en.wikipedia.org/wiki/Arithmetic_mean
How can it be used to predict or explain my experimental results?As far as I can tell, your experiment did not involve a system that was at 0oC.
Post by: alancalverd on 28/05/2022 17:39:22
I will concede that. Chemical reactions are well known.A spectacular mechanical example was BC's mention of the fire at the Windscale nuclear reactor, where the accumulated microscopic mechanical stresses due to neutrons displacing carbon atoms from their lattice, were all resolved in a short time when the temperature rose to the annealing point and then "took off" in a chain reaction. And the classic case of the electrical energy of a body being converted to heat is the spontaneous self-destruction of lithium batteries.The only thing that can change the temperature of a body is adding or removing energy.Or changing from other forms of energy, such as chemical, mechanical, or electrical energy.
Post by: hamdani yusuf on 31/05/2022 15:51:52
And the classic case of the electrical energy of a body being converted to heat is the spontaneous self-destruction of lithium batteries.I think it involves chemical energy, in the form of chemical bonds. Discharge of a capacitor is a more exclusive example of electrical energy turning into heat which changes temperature.
Post by: hamdani yusuf on 31/05/2022 16:08:00
Which kind of average is the most relevant to temperature? Mol weighted average, mass weighted average, volume weighted average, time weighted average?What do you think that average means?1.
a number expressing the central or typical value in a set of data, in particular the mode, median, or (most commonly) the mean, which is calculated by dividing the sum of the values in the set by their number.
https://en.wikipedia.org/wiki/Arithmetic_meanHow can it be used to predict or explain my experimental results?As far as I can tell, your experiment did not involve a system that was at 0oC.
What is the average temperature of ice water mixture in atmospheric pressure after being left to reach equilibrium?
Post by: Origin on 31/05/2022 16:27:10
What is the average temperature of ice water mixture in atmospheric pressure after being left to reach equilibrium?How can you possibly be asking this question after 11 pages?!? Are seriously saying you don't know the temperature will be 0C??? I'm beginning to think there is something wrong with you....
Post by: Bored chemist on 31/05/2022 18:46:39
Which kind of average is the most relevant to temperature? Mol weighted average, mass weighted average, volume weighted average, time weighted average?Since the ice and water are at exactly the same temperature, it doesn't matter.
Post by: alancalverd on 02/06/2022 18:09:27
I think it involves chemical energy, in the form of chemical bonds.No.Lithium batteries can develop an internal short circuit. Used to cause the occasional laptop computer to burst into flames so laptop use was were banned from passenger aircraft for a while, then a few early Dreamliners caught fire thanks to the aircraft's own hi-tech lightweight starter batteries!
Post by: hamdani yusuf on 04/06/2022 14:54:28
What is the average temperature of ice water mixture in atmospheric pressure after being left to reach equilibrium?How can you possibly be asking this question after 11 pages?!? Are seriously saying you don't know the temperature will be 0C??? I'm beginning to think there is something wrong with you....
Maybe you can understand if you've read the post as a whole, including which statement I was responding to. Otherwise, you'll keep wondering.
As far as I can tell, your experiment did not involve a system that was at 0oC.
Post by: hamdani yusuf on 04/06/2022 15:10:31
Since the ice and water are at exactly the same temperature, it doesn't matter.But that's only an idealized and unrealistic condition, which is hard to achieve in real life experiment.
Post by: Bored chemist on 04/06/2022 15:15:28
Here's how they did it a few hundred years ago.Since the ice and water are at exactly the same temperature, it doesn't matter.But that's only an idealized and unrealistic condition, which is hard to achieve in real life experiment.
https://www.nature.com/scitable/content/ice-calorimeter-developed-by-lavoisier-and-laplace-14898943/
Post by: hamdani yusuf on 04/06/2022 15:16:18
What makes you think that the energy is not stored in the form of chemical bonds?I think it involves chemical energy, in the form of chemical bonds.No.Lithium batteries can develop an internal short circuit. Used to cause the occasional laptop computer to burst into flames so laptop use was were banned from passenger aircraft for a while, then a few early Dreamliners caught fire thanks to the aircraft's own hi-tech lightweight starter batteries!
Post by: hamdani yusuf on 04/06/2022 15:21:20
Here's how they did it a few hundred years ago.It doesn't show that the ice and water are at exactly the same temperature at every point.
https://www.nature.com/scitable/content/ice-calorimeter-developed-by-lavoisier-and-laplace-14898943/
Post by: Origin on 04/06/2022 15:39:26
Maybe you can understand if you've read the post as a whole, including which statement I was responding to. Otherwise, you'll keep wondering.No that doesn't help because you have been told multiple time that the temperature would be 0C. For some bizarre reason no matter how many times you are given the correct answer you continue to ask the question.
Post by: Bored chemist on 04/06/2022 15:46:30
It does; it's just that you don't understand it.Here's how they did it a few hundred years ago.It doesn't show that the ice and water are at exactly the same temperature at every point.
https://www.nature.com/scitable/content/ice-calorimeter-developed-by-lavoisier-and-laplace-14898943/
If you have ice and water at equilibrium, in a closed vessel surrounded by ice and water, what other temperature can it be apart from 0C?
Consider the following before you answer.
(1) That if you add heat to the system, you won't change the temperature, you will just met some ice.
(2) That if you remove heat from the system, you won't reduce the temperature, you will just freeze some water
(3) Since the ice and water inside the container is at the same temperature as the ice and water outside it, there is no temperature gradient across the container wall, and therefore no heat transfer.
So, if there was a transfer of heat, the temperature wouldn't change, and there's no mechanism for a transfer of heat anyway.
Post by: Bored chemist on 04/06/2022 15:47:45
Energy can be stored in chemical bonds, and it is absorbed or released when chemical reactions take place.What makes you think that the energy is not stored in the form of chemical bonds?I think it involves chemical energy, in the form of chemical bonds.No.Lithium batteries can develop an internal short circuit. Used to cause the occasional laptop computer to burst into flames so laptop use was were banned from passenger aircraft for a while, then a few early Dreamliners caught fire thanks to the aircraft's own hi-tech lightweight starter batteries!
But in the case of ice and water, there are no reactions, so there is no change in energy.
Post by: hamdani yusuf on 04/06/2022 17:22:20
Those were not what I've observed in my experiment. The ice-water mixture in the large container couldn't prevent the ice-water mixture in smaller container from changing its ratio. Melting or freezing still happened depending on the ambient temperature.It does; it's just that you don't understand it.Here's how they did it a few hundred years ago.It doesn't show that the ice and water are at exactly the same temperature at every point.
https://www.nature.com/scitable/content/ice-calorimeter-developed-by-lavoisier-and-laplace-14898943/
If you have ice and water at equilibrium, in a closed vessel surrounded by ice and water, what other temperature can it be apart from 0C?
Consider the following before you answer.
(1) That if you add heat to the system, you won't change the temperature, you will just met some ice.
(2) That if you remove heat from the system, you won't reduce the temperature, you will just freeze some water
(3) Since the ice and water inside the container is at the same temperature as the ice and water outside it, there is no temperature gradient across the container wall, and therefore no heat transfer.
So, if there was a transfer of heat, the temperature wouldn't change, and there's no mechanism for a transfer of heat anyway.
Post by: Bored chemist on 04/06/2022 17:25:15
Those were not I've observed in my experiment.There are two possible reasons for that.
Post by: hamdani yusuf on 04/06/2022 17:27:40
You haven't even tried.Maybe you can understand if you've read the post as a whole, including which statement I was responding to. Otherwise, you'll keep wondering.No that doesn't help because you have been told multiple time that the temperature would be 0C. For some bizarre reason no matter how many times you are given the correct answer you continue to ask the question.
Post by: Bored chemist on 04/06/2022 17:35:12
Tried what?You haven't even tried.Maybe you can understand if you've read the post as a whole, including which statement I was responding to. Otherwise, you'll keep wondering.No that doesn't help because you have been told multiple time that the temperature would be 0C. For some bizarre reason no matter how many times you are given the correct answer you continue to ask the question.
Tried doing an experiment badly?
Do you remember posting about this?
Galileo did a thought experiment and overturned the views of Aristotle.It's not a fallacy if they are an authority.It seems like you haven't learned about Galileo.
And then, because he was trying to explain it to people who were unaccustomed to actually thinking, he did the practical experiment.
So what?
Where I pointed out that Galileo already knew (from a thought experiment) what the outcome of the real experiment would be. He only had to do the practical experiment because the people he was talking to didn't understand what he was telling them.
Post by: Origin on 04/06/2022 18:38:17
You haven't even tried.When I was at university I did labs that proved to me that heat transfer is driven by the delta T.
Post by: alancalverd on 04/06/2022 21:48:26
Melting or freezing still happened depending on the ambient temperature.Wonders will never cease.
Post by: alancalverd on 04/06/2022 21:55:21
Between 0 and 100°C, H2O is a liquid
Below 0°C it is a solid
So what happens at 0°C?
Either the solid and liquid phases coexist in equilibrium, or they spontaneously vanish.
What do we observe?
They don't vanish.
Post by: hamdani yusuf on 05/06/2022 06:44:07
Try looking at it a different way. A sort of Galilean thought experiment.If ambient temperature is slightly above 0 degrees C, then water in contact with the container increases its temperature, increases its density, and sinks to the bottom.
Between 0 and 100°C, H2O is a liquid
Below 0°C it is a solid
So what happens at 0°C?
Either the solid and liquid phases coexist in equilibrium, or they spontaneously vanish.
What do we observe?
They don't vanish.
On the other hand, If ambient temperature is slightly below 0 degrees C, then water in contact with the container decreases its temperature, decreases its density, and floats to the top before it freezes. The temperature gradient will form naturally through buoyancy.
Post by: alancalverd on 05/06/2022 10:14:11
Post by: Bored chemist on 05/06/2022 13:42:39
The temperature gradient will form naturally through buoyancy.No it will not.
Because, if all the water is at 0 C then it all has the same density and there can not be a gradient.
Why don't you understand that?
Post by: hamdani yusuf on 06/06/2022 12:39:06
In my experiment, outside of the large plastic container was either -4°C or 4°C. The inner side of the container will be somewhere between 0°C and -4°C or 4°C.The temperature gradient will form naturally through buoyancy.No it will not.
Because, if all the water is at 0 C then it all has the same density and there can not be a gradient.
Why don't you understand that?
Post by: Origin on 06/06/2022 13:43:44
In my experiment, outside of the large plastic container was either -4°C or 4°C. The inner side of the container will be somewhere between 0°C and -4°C or 4°C.Then there is a delta T, so there will be heat transfer. So your experiment will not address the question posed in the OP. So you get to go in circles and never accept an answer to the OP. Is this how you have fun, confusing yourself?
Post by: hamdani yusuf on 06/06/2022 13:58:03
It shows the difference between theory and practice. What you think as obviously simple might be hard to demonstrate in real life. You can simply have faith to your theory without bothering to check with reality. Or you can try to improve the experiment to improve signal over noise ratio.In my experiment, outside of the large plastic container was either -4°C or 4°C. The inner side of the container will be somewhere between 0°C and -4°C or 4°C.Then there is a delta T, so there will be heat transfer. So your experiment will not address the question posed in the OP. So you get to go in circles and never accept an answer to the OP. Is this how you have fun, confusing yourself?
Post by: Origin on 06/06/2022 16:25:13
You can simply have faith to your theory without bothering to check with reality.Absolutely wrong. We are smart enough to understand the underlying principals and can apply them to different situations without having to run an experiment.
If I hit my thumb with a hammer it will hurt like hell. I do not have to run an experiment to see if hitting my thumb with a crescent wrench will also hurt. I can use my physic knowledge to realize it would also hurt. You on the other hand would have the whole contents of the toolbox lined up and just keep selecting each different tool and smashing your thumbs just to make sure, I guess.
Post by: Bored chemist on 06/06/2022 16:50:25
In my experiment, outside of the large plastic container was either -4°C or 4°C.Then you did a poor experiment.
Post by: alancalverd on 06/06/2022 20:26:05
What you think as obviously simple might be hard to demonstrate in real life.I repeat, yet again, the wisdom of Sir Lawrence Bragg and the experience of one who spent many years measuring small temperature rises: if you get better than 20% accuracy in a heat experiment, you are doing very well. And believe me, getting from 20% to 0.1% requires a lot of engineering.
Post by: hamdani yusuf on 07/06/2022 07:23:57
The main experiment is in the small containers. The larger container was meant to provide 0°C environment, which was shown to be inadequate.In my experiment, outside of the large plastic container was either -4°C or 4°C.Then you did a poor experiment.
It's possible to provide multiple layers of containers.
Post by: hamdani yusuf on 07/06/2022 07:37:48
We are smart enough to understand the underlying principals and can apply them to different situations without having to run an experiment.Let's wait and see if your confidence is supported by experimental results. This kind of confidence reminds me of ancient natural philosophers and classical scientist in the end of 19th century.
Post by: hamdani yusuf on 07/06/2022 07:58:14
Here are my main points in this thread :In my other thread, I concluded that temperature of an object represents its internal kinetic energy, although some refinements may be done to the definitions of internal and kinetic, in contrast to external and potential energies, respectively. The water and ice at 0°C have the same internal kinetic energy. Hence the water must have higher potential energy, assuming that no external energy is observed.
1. Heat flows from higher temperature object to lower temperature object.
2. Ice and water at melting point has the same average temperature, 0C.
3. At melting point, water has higher internal energy compared to ice.
4. Local temperature can be different from average temperature.
Is there any point you don't agree with?
My hypothesis which I want to test using the experiment is that the water-ice mixture has dynamic equilibrium, which means that ,conversions occur in both direction at the same rate, instead of static equilibrium, which means that no conversion occurs at all.
Post by: Bored chemist on 07/06/2022 08:54:14
In my other thread, I concluded that temperature of an object represents its internal kinetic energySo you made a mistake.
Post by: alancalverd on 07/06/2022 09:02:56
My hypothesis which I want to test using the experiment is that the water-ice mixture has dynamic equilibrium, which means that ,conversions occur in both direction at the same rate, instead of static equilibrium, which means that no conversion occurs at all.That is entirely obvious, since neither body has zero internal kinetic energy. On another thread we discussed the possibility of a block of ice changing shape with no net exchange of mass.
Post by: hamdani yusuf on 07/06/2022 09:09:30
What's the correct conclusion?In my other thread, I concluded that temperature of an object represents its internal kinetic energySo you made a mistake.
Post by: hamdani yusuf on 07/06/2022 09:14:07
The fact that there's still disagreements here shows that it's not that obvious yet.My hypothesis which I want to test using the experiment is that the water-ice mixture has dynamic equilibrium, which means that ,conversions occur in both direction at the same rate, instead of static equilibrium, which means that no conversion occurs at all.That is entirely obvious, since neither body has zero internal kinetic energy. On another thread we discussed the possibility of a block of ice changing shape with no net exchange of mass.
Post by: alancalverd on 07/06/2022 09:48:00
Post by: Bored chemist on 07/06/2022 13:12:28
What's the correct conclusion?temperature of an object does not represent just its internal kinetic energy
Post by: hamdani yusuf on 08/06/2022 07:22:57
temperature of an object does not represent just its internal kinetic energyWhat else are there?
Post by: hamdani yusuf on 08/06/2022 07:24:51
The only person expressing any disagreement is yourself, apparently based on a meaningless and irrelevant experiment and a failure to comprehend "average".Do you have any idea to make the experiment meaningful?
How do you interpret average?
Post by: Bored chemist on 08/06/2022 08:42:41
Do you have any idea to make the experiment meaningful?Yes.
That's why I posted it.
If you have ice and water at equilibrium, in a closed vessel surrounded by ice and water, what other temperature can it be apart from 0C?
Consider the following before you answer.
(1) That if you add heat to the system, you won't change the temperature, you will just met some ice.
(2) That if you remove heat from the system, you won't reduce the temperature, you will just freeze some water
(3) Since the ice and water inside the container is at the same temperature as the ice and water outside it, there is no temperature gradient across the container wall, and therefore no heat transfer.
So, if there was a transfer of heat, the temperature wouldn't change, and there's no mechanism for a transfer of heat anyway.
Post by: alancalverd on 08/06/2022 18:01:01
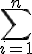 xi)/n. At least it was when I was alive, but this thread seems to be some kind of scientific purgatory.
xi)/n. At least it was when I was alive, but this thread seems to be some kind of scientific purgatory.
Post by: Origin on 08/06/2022 18:46:16
The average of n samples of x is ( xi)/n. At least it was when I was alive, but this thread seems to be some kind of scientific purgatory.Agreed. I think I saw a Twilight Zone where this thread the basic premise of the show.
Post by: Bored chemist on 08/06/2022 19:14:25
The average of n samples of x is (It's important, in some cases to remember that there are several sorts of averages- mode, median and at least 3 sorts of mean.xi)/n. At least it was when I was alive, but this thread seems to be some kind of scientific purgatory.
And, even if you are talking about an arithmetic mean (as Alan highlighted) you can still run into problems.
If I mix the meat from one horse and one chicken, is it a 50:50 mixture?
And it's also important to distinguish between averaging over, for example, time, or position.
But when absolutely ****ing well everything under consideration is at 0oC, the average is zero, regardless of what definition you use.
Any non-stupid definition will give the same answer.
Post by: alancalverd on 08/06/2022 19:16:24
But when absolutely ****ing well everything under consideration is at 0oC, the average is zero, regardless of what definition you use.No, the average is 273 x kB. It tends to zero at 0K, not 0°C.
But what matters here is that the average is the same for ice and water, or indeed any number of components at the same temperature.
Quote
If I mix the meat from one horse and one chicken, is it a 50:50 mixture?no, but if you mix them well then any sample will be close to the average!
Post by: Bored chemist on 08/06/2022 19:32:12
But when absolutely ****ing well everything under consideration is at 0oC, the average is zero, regardless of what definition you use.To clarify...
But when absolutely ****ing well everything under consideration is at 0oC, the average is zero oC.
Post by: paul cotter on 08/06/2022 21:02:35
Post by: Bored chemist on 08/06/2022 21:39:20
Again, it's Galileo dropping things off a tower.
He did not need to do the experiment.
He knew that the light thing and the heavy thing had to fall at the same speed- because he had considered what would happen if you tied a heavy ball to a light ball and dropped both.
The combined "thing" consisting of the two balls would obviously weigh more than the constituents.
So, if Aristotle had been right, the combination would fall faster than the heavy ball.
And the light ball would fall more slowly.
But how could the combined thing fall faster then the heavy ball when the light ball was trying to fall slower?
It's impossible.
Galileo knew that.
He only did the experiment for the benefit of the local dignitaries who were not clever enough to understand the logic.
Now we are looking at the laws of thermodynamics, rather than falling objects.
But, like Galileo, we have the advantage of a deep understanding.
We have known for a hundred years or so that momentum, angular momentum and energy are conserved.
We don't rely on experiments to know this.
We have a mathematical proof.
https://en.wikipedia.org/wiki/Noether%27s_theorem
And yet, we still have hamdani yusuf saying that, because he can't do a proper experiment, he doesn't believe it.
He refuses to learn, or even accept the science.
You can lead a horse to water, but you can't make it drink.
He is even less well informed that those local busybodies for whom Galilei had to climb the tower and drop stuff.
Post by: alancalverd on 09/06/2022 15:33:18
He knew that the light thing and the heavy thing had to fall at the same speed- because he had considered what would happen if you tied a heavy ball to a light ball and dropped both.He should have known better, living half a century after da Vinci invented the parachute. Didn't anyone do a literature search?
Post by: hamdani yusuf on 10/06/2022 05:10:09
The average of n samples of x is (What if the particles have different masses?xi)/n. At least it was when I was alive, but this thread seems to be some kind of scientific purgatory.
Post by: hamdani yusuf on 10/06/2022 05:11:59
That's what I did, and the result was different than your prediction.Do you have any idea to make the experiment meaningful?Yes.
That's why I posted it.If you have ice and water at equilibrium, in a closed vessel surrounded by ice and water, what other temperature can it be apart from 0C?
Consider the following before you answer.
(1) That if you add heat to the system, you won't change the temperature, you will just met some ice.
(2) That if you remove heat from the system, you won't reduce the temperature, you will just freeze some water
(3) Since the ice and water inside the container is at the same temperature as the ice and water outside it, there is no temperature gradient across the container wall, and therefore no heat transfer.
So, if there was a transfer of heat, the temperature wouldn't change, and there's no mechanism for a transfer of heat anyway.
Post by: hamdani yusuf on 10/06/2022 05:18:45
All you need are the laws of thermodynamics and the phase diagram for water to predict the outcome. The op, in my opinion, thinks the laws of thermodynamics are some sort of scientific 'dogma'. The laws of thermodynamics are probably the most solid and incontrovertible of all the laws of science. They have been tested and tested to the n+1 degree again and again and again(longer than this discussion).Just for fun,
(https://pbs.twimg.com/media/FUqedw8akAA2cEq?format=jpg&name=900x900)
Post by: hamdani yusuf on 10/06/2022 05:33:59
Between first and second compartment is a thermal conductor, such as aluminum plate. While second compartment is separated from third compartment by thermal insulator, such as styrofoam plate.
Second compartment will be filled by 90% ice & 10% water, while first and third compartment will be filled by 10% ice & 90% water. Heat transfer with the environment should be equal between first and third compartment. If there are different result, it must come from heat transfer with second compartment.
Is there any objection to this plan?
Post by: Bored chemist on 10/06/2022 08:45:40
That's what I did,I thought you said you did it at the wrong temperature
In my experiment, outside of the large plastic container was either -4°C or 4°C.
Post by: alancalverd on 10/06/2022 12:49:12
What if the particles have different masses?The average remains the average. If all the members were known to be identical there would be no point in calculating the average since every member is completely representative of the population.
Post by: alancalverd on 10/06/2022 12:52:20
Is there any objection to this plan?Only that it would be a pointless waste of the promising life of an enthusiastic experimental physicist.
If you get no difference, what would you deduce?
Post by: hamdani yusuf on 10/06/2022 12:55:36
Water-ice mixture in the larger container, which is outside smaller containers, is supposed to keep its temperature at 0°C.That's what I did,I thought you said you did it at the wrong temperatureIn my experiment, outside of the large plastic container was either -4°C or 4°C.
Post by: hamdani yusuf on 10/06/2022 13:11:56
This experiment only needs to be done once. I can share it to Youtube for free so everyone can save their resources for something else deemed more important.Is there any objection to this plan?Only that it would be a pointless waste of the promising life of an enthusiastic experimental physicist.
If you get no difference, what would you deduce?
If the first and third compartments have the same ice-water ratio at the end of the experiment, it means that microscopic fluctuation and local temperature variation doesn't affect overall macroscopic heat flow.
Otherwise, microscopic fluctuation and local temperature variation does affect overall macroscopic heat flow.
Post by: Bored chemist on 10/06/2022 13:13:51
Water-ice mixture in the larger container, which is outside smaller containers, is supposed to keep its temperature at 0°C.And, if it had, the inner container would not have been able to lose or gain heat.
So, the million dollar question is "did you use enough ice and water?"
Post by: Bored chemist on 10/06/2022 13:15:30
This experiment only needs to be done once.No
It does not need to be done at all.
Did you see what I wrote here?
It's not how well tested the laws of thermodynamics are that matters here.
Again, it's Galileo dropping things off a tower.
He did not need to do the experiment.
He knew that the light thing and the heavy thing had to fall at the same speed- because he had considered what would happen if you tied a heavy ball to a light ball and dropped both.
The combined "thing" consisting of the two balls would obviously weigh more than the constituents.
So, if Aristotle had been right, the combination would fall faster than the heavy ball.
And the light ball would fall more slowly.
But how could the combined thing fall faster then the heavy ball when the light ball was trying to fall slower?
It's impossible.
Galileo knew that.
He only did the experiment for the benefit of the local dignitaries who were not clever enough to understand the logic.
Now we are looking at the laws of thermodynamics, rather than falling objects.
But, like Galileo, we have the advantage of a deep understanding.
We have known for a hundred years or so that momentum, angular momentum and energy are conserved.
We don't rely on experiments to know this.
We have a mathematical proof.
https://en.wikipedia.org/wiki/Noether%27s_theorem
And yet, we still have hamdani yusuf saying that, because he can't do a proper experiment, he doesn't believe it.
He refuses to learn, or even accept the science.
You can lead a horse to water, but you can't make it drink.
He is even less well informed that those local busybodies for whom Galilei had to climb the tower and drop stuff.
and did you understand it?
Since the answer is known, what is the point of the experiment?
Post by: alancalverd on 10/06/2022 14:36:12
This experiment only needs to be done once.I repeat: if you detect no difference, what would you deduce?
Post by: hamdani yusuf on 11/06/2022 04:17:52
I found this video quite informative and related to this thread.
Post by: hamdani yusuf on 11/06/2022 04:20:57
How much is enough?Water-ice mixture in the larger container, which is outside smaller containers, is supposed to keep its temperature at 0°C.And, if it had, the inner container would not have been able to lose or gain heat.
So, the million dollar question is "did you use enough ice and water?"
I used water-ice mixture in the larger container more than ten times the volume of the smaller containers.
Post by: hamdani yusuf on 11/06/2022 04:23:15
This experiment only needs to be done once.I repeat: if you detect no difference, what would you deduce?
If the first and third compartments have the same ice-water ratio at the end of the experiment, it means that microscopic fluctuation and local temperature variation doesn't affect overall macroscopic heat flow.
Otherwise, microscopic fluctuation and local temperature variation does affect overall macroscopic heat flow.
Post by: hamdani yusuf on 11/06/2022 04:41:14
The point is, to identify internal or external factors which can affect the experimental result. In free falling case, people may observe that heavy rain falls faster than drizzle. Stone falls faster than sand or dust. Hammer falls faster than feather. In those cases, the influential factor is air friction. Your idealized thought experiment says nothing about it.This experiment only needs to be done once.No
It does not need to be done at all.
Did you see what I wrote here?It's not how well tested the laws of thermodynamics are that matters here.
Again, it's Galileo dropping things off a tower.
He did not need to do the experiment.
He knew that the light thing and the heavy thing had to fall at the same speed- because he had considered what would happen if you tied a heavy ball to a light ball and dropped both.
The combined "thing" consisting of the two balls would obviously weigh more than the constituents.
So, if Aristotle had been right, the combination would fall faster than the heavy ball.
And the light ball would fall more slowly.
But how could the combined thing fall faster then the heavy ball when the light ball was trying to fall slower?
It's impossible.
Galileo knew that.
He only did the experiment for the benefit of the local dignitaries who were not clever enough to understand the logic.
Now we are looking at the laws of thermodynamics, rather than falling objects.
But, like Galileo, we have the advantage of a deep understanding.
We have known for a hundred years or so that momentum, angular momentum and energy are conserved.
We don't rely on experiments to know this.
We have a mathematical proof.
https://en.wikipedia.org/wiki/Noether%27s_theorem
And yet, we still have hamdani yusuf saying that, because he can't do a proper experiment, he doesn't believe it.
He refuses to learn, or even accept the science.
You can lead a horse to water, but you can't make it drink.
He is even less well informed that those local busybodies for whom Galilei had to climb the tower and drop stuff.
and did you understand it?
Since the answer is known, what is the point of the experiment?
My experiment will determine if microscopic fluctuation and local temperature variation are significant. The follow up question would be are there other influential factors? Such as surface color of the separator, its roughness, thickness, heat conductance, enlarged contact area by pleats, etc.
Post by: hamdani yusuf on 11/06/2022 09:28:57
Let's say that at the beginning of the experiment, the system is in equilibrium, where average temperature is 0°C.
Temperature of the metal surface in contact with water is also 0°C. Microscopic fluctuation causes a spot to be slightly hotter, say 0.002°C, while another spot is slightly colder, say -0.002°C, but the average temperature is still 0°C.
The hotter spot will give some of its thermal energy to the water, increasing the water temperature at that spot from 0°C to 0.001°C while reducing its own temperature from 0.002°C to 0.001°C.
The colder spot will receive some of thermal energy from the water, freezing the water while increasing the temperature at that spot from -0.002°C to 0°C.
Those pair of interactions will increase the average temperature of the metal surface in water side to slightly above 0°C.
Meanwhile, on the other side of the metal plate, temperature of the metal surface in contact with ice is also 0°C. Microscopic fluctuation causes a spot to be slightly hotter, say 0.002°C, while another spot is slightly colder, say -0.002°C, but the average temperature is still 0°C.
The hotter spot will give some of its thermal energy to the ice, melting the ice while decreasing the temperature at that spot from 0.002°C to 0°C.
The colder spot will receive some of thermal energy from the ice, decreasing the ice temperature at that spot from 0°C to -0.001°C while increasing its own temperature from -0.002°C to -0.001°C.
Those pair of interactions will decrease the average temperature of the metal surface in ice side to slightly below 0°C.
After the reaction, the average temperature at water side of the metal will be different than the ice side. This temperature difference may cause heat flow from water side to ice side of the metal plate.
Of course, there is a possibility that the water and ice in each sides react faster to the temperature difference caused by that fluctuation, hence maintaining the average temperature of each sides and preventing heat flow. But I couldn't find any reference about this. That's why I thought some experiments were needed to answer it.
Post by: hamdani yusuf on 11/06/2022 10:19:27
And yet, we still have hamdani yusuf saying that, because he can't do a proper experiment, he doesn't believe it.Let me share what I learned about science in a fun way.
He refuses to learn, or even accept the science.
Post by: alancalverd on 11/06/2022 12:21:48
The hotter spot will give some of its thermal energy to the water, increasing the water temperature at that spot from 0°C to 0.001°C while reducing its own temperature from 0.002°C to 0.001°C.Only if 1 = 2.
The colder spot will receive some of thermal energy from the water, freezing the water while increasing the temperature at that spot from -0.002°C to 0°C.
Post by: hamdani yusuf on 11/06/2022 13:56:43
You can simply say that thermal fluctuation doesn't exist.The hotter spot will give some of its thermal energy to the water, increasing the water temperature at that spot from 0°C to 0.001°C while reducing its own temperature from 0.002°C to 0.001°C.Only if 1 = 2.
The colder spot will receive some of thermal energy from the water, freezing the water while increasing the temperature at that spot from -0.002°C to 0°C.
Post by: alancalverd on 12/06/2022 00:34:24
Your post #273 simply does not make sense.
Post by: hamdani yusuf on 12/06/2022 09:43:48
Why would I say that? The kinetic energy of the constituent atoms and molecules of any body can be assumed to be something like a boltzmann distribution, so at any instant some will have more and some less than the mean. But temperature is a statement of the mean, so if two bodies are at the same temperature there can be no net heat exchange between them.
Your post #273 simply does not make sense.
So when a metal particle is about to interact with a water molecule next to it, it must know the average energy of its neighbors before giving or receiving thermal energy.
Post by: alancalverd on 12/06/2022 11:22:40
And there is no reason why a single molecular interaction should depend (at least to the first order) on the kinetic energy of any other molecules.
Post by: hamdani yusuf on 12/06/2022 23:04:21
And there is no reason why a single molecular interaction should depend (at least to the first order) on the kinetic energy of any other molecules.That's precisely why I said that perhaps the variation can produce observable effects, which was the reason I made the experiment in the first place. I wouldn't waste my time if I was sure that there would be no variation, nor if I thought that it's impossible to observe the effects.
Post by: alancalverd on 12/06/2022 23:17:13
Post by: hamdani yusuf on 13/06/2022 07:27:21
It would be impossible to observe any effect because for every interaction A -> B there will be, somewhere, an equal and opposite B->A, due to the definition of temperature. So how do you choose which molecule to observe?There are at least 4 processes may accompany thermal fluctuation in the experiment:
increase of local temperature.
decrease of local temperature.
melting.
freezing.
The first two involve changing of kinetic energy. The last two involve changing of potential energy.
I choose to observe the molecules which undergo freezing. Ice is less dense than water. It tends to float and accumulates on the water surface.
Post by: alancalverd on 13/06/2022 09:37:20
Either you are going to alter the average internal kinetic energy of something (i.e. change its temperature) or not.
If you alter any temperature your result will not be an answer to the question.
If you don't, you have simply confirmed that you understand the meaning of "temperature".
Post by: hamdani yusuf on 13/06/2022 23:47:36
"Thermal fluctuation" implies a change in temperature.One part of an object can increase its temperature while another part decrease its temperature, thus the average temperature doesn't change.
Either you are going to alter the average internal kinetic energy of something (i.e. change its temperature) or not.
Post by: alancalverd on 14/06/2022 12:40:38
Except for the anthropic "can increase its temperature". Not spontaneously or idiogenically, unless you are talking about yogic physiology or hibernating mammals. Or maybe dragonflies, who warm up their engines before takeoff.
Post by: hamdani yusuf on 14/06/2022 16:04:30
Post by: alancalverd on 14/06/2022 18:15:23
Post by: Bored chemist on 15/06/2022 08:48:32
One part of an object can increase its temperature while another part decrease its temperature, thus the average temperature doesn't change.The problem with sating that (well one problem) is the temperature of something is always an average.
So what you are saying is that the average changes, but the average doesn't change.
Post by: hamdani yusuf on 15/06/2022 13:51:36
You forget to distinguish between local and global temperature.One part of an object can increase its temperature while another part decrease its temperature, thus the average temperature doesn't change.The problem with sating that (well one problem) is the temperature of something is always an average.
So what you are saying is that the average changes, but the average doesn't change.
Post by: alancalverd on 15/06/2022 14:00:11
Post by: Eternal Student on 15/06/2022 20:44:40
I don't suppose I've managed to read every post since I was last here but I think I've got the gist.
I have to strongly agree with what @alancalverd and @Bored chemist have just tried to say:
It is dangerous and difficult to try and consider "local temperature" when you're considering a volume so small that you have only a few molecules. It makes very little sense to model that volume as one homogeneous body with many particles that have an average kinetic energy corresponding to the given temperature (because it just does not have many particles - so by assuming it has you're almost bound to get nonsense results and consequences).
Yes, the phrase "local temperature" is used frequently but not on those small scales. The weather presenter will tell you that the temperature of the air in Spain is different to the temperature in Canada. However that's still millions of particles of Nitrogen that exist in a given region. It is reasonable to model that volume and number of particles as one homogeneous body with a well defined temperature.
Best Wishes.
Post by: Bored chemist on 15/06/2022 22:40:38
You invented a distinction that does not actually exist.You forget to distinguish between local and global temperature.One part of an object can increase its temperature while another part decrease its temperature, thus the average temperature doesn't change.The problem with sating that (well one problem) is the temperature of something is always an average.
So what you are saying is that the average changes, but the average doesn't change.
Post by: hamdani yusuf on 16/06/2022 14:02:28
Hi.Can we measure the temperature of water in a 1 cubic micron?
I don't suppose I've managed to read every post since I was last here but I think I've got the gist.
I have to strongly agree with what @alancalverd and @Bored chemist have just tried to say:
It is dangerous and difficult to try and consider "local temperature" when you're considering a volume so small that you have only a few molecules. It makes very little sense to model that volume as one homogeneous body with many particles that have an average kinetic energy corresponding to the given temperature (because it just does not have many particles - so by assuming it has you're almost bound to get nonsense results and consequences).
Yes, the phrase "local temperature" is used frequently but not on those small scales. The weather presenter will tell you that the temperature of the air in Spain is different to the temperature in Canada. However that's still millions of particles of Nitrogen that exist in a given region. It is reasonable to model that volume and number of particles as one homogeneous body with a well defined temperature.
Best Wishes.
What's the guarantee that its temperature is exactly the same as its neighboring water body?
Post by: alancalverd on 16/06/2022 14:24:34
(b) negligible
But the answer to the OP remains "no".
Post by: Bored chemist on 16/06/2022 17:19:10
Can we measure the temperature of water in a 1 cubic micron?Over what timescale?
What's the guarantee that its temperature is exactly the same as its neighboring water body?
Post by: Eternal Student on 16/06/2022 20:27:42
Can we measure the temperature of water in a 1 cubic micron?I'm going to say no, not reliably or meaningfully.
There's no set scale at which everyone declares that it's unreasonable to assign a temperature, instead the approximation (treating the region as a homogeneous body with many particles, having average energy corresponding to the temperature) just becomes progressively less useful.
Best Wishes.
Post by: Bored chemist on 16/06/2022 21:06:14
I'm going to say no, not reliably or meaningfully.Others may differ
http://users.mrl.illinois.edu/cahill/intel08.pdf
But if you asked about a nanometre cubed, it would be tricky.
Post by: hamdani yusuf on 20/06/2022 06:16:42
Whatever needed by a measuring device / method to produce conclusive result.Can we measure the temperature of water in a 1 cubic micron?Over what timescale?
What's the guarantee that its temperature is exactly the same as its neighboring water body?
Post by: Bored chemist on 20/06/2022 18:56:03
Good.Whatever needed by a measuring device / method to produce conclusive result.Can we measure the temperature of water in a 1 cubic micron?Over what timescale?
What's the guarantee that its temperature is exactly the same as its neighboring water body?
That means that the answer is simple. If the water you are seeking to measure the temperature of is in equilibrium with ice then the temperature is (on average) 0C.
Post by: alancalverd on 20/06/2022 23:06:53
Whatever needed by a measuring device / method to produce conclusive result.A really tiny thermistor, or literally, a gnat's whisker. Mosquitoes and bed bugs have unbelievably good temperature sensors built on micron scales.
Post by: Deecart on 21/06/2022 20:50:34
What happens if you put on each side in a perfect isolated box half ice and half water at 0 C ? (With no air i suppose ?)
Then my answer would be : You will end up with some sort of icebergs at 0C with liquid water cavity inside, in a water at 0C... but you will have less ice than water (you loose some of the ice).
Because : The dynamic system (ice become liquid and reversa) try to maximise the heat exchange, so the surface will be maximum.
So could think, huu, so we end up with isolaed icebergs.
But there is not enought room for that.
And furthermore... immediatly when water can mix with the ice (initialy the ice is stucked to the walls) the Archimed force will add some energy at the system.
So perhaps ice could gain some temperature (0 C at the border but a little bit more inside the ice, heat conduction lowers when the ice is thick (with no proportionality to the tickness of ice).
I dont know exactly how we have to take entropy in account here, but the mixed ice and water block could represent some gain in entropy (disorder) : Therefore this energy is taken from the whole other energys stored in the system (so temperature lowers a bit)
Therefore i have no real clue what will happens finally (not sure if we can really do the calculation in such a complex system).
Post by: alancalverd on 22/06/2022 17:28:53
The dynamic system (ice become liquid and reversa) try to maximise the heat exchange,What heat exchange? Heat flow only occurs if there is a temperarture difference. If not, how would the heat know which way to flow, and when to stop?
Post by: Bored chemist on 22/06/2022 18:05:05
Therefore i have no real clue what will happens finallyNothing- as Alan pointed out.
(not sure if we can really do the calculation in such a complex system).We can.
We did.
Post by: Deecart on 23/06/2022 21:12:15
Quote from: alancalverd
What heat exchange? Heat flow only occurs if there is a temperarture difference. If not, how would the heat know which way to flow, and when to stop?
Sure there is heat exchange, or kinetic energy flow if you prefer.
If you had two molecules that could not interact, i would agree with you : You could in this case say that there is some heat exchange only if you take some very small volume, due to the variability of vibration (very small so very small heat exchange, even with small volume).
But here, we have something else and it is very interesting in term of interaction.
You have only water but in 2 states.
And the two states are at the limit where (statisticaly...) they can become liquid or solid.
You surely know the fact that if you let some liquid water in a pot at room temperature, it evaporate.
There is no need to reach the 100°C boiling temperature to have the liquid water become vapor water.
Same with ice, wich can become liquid or even vapor at 0°C.
It is due to the fact that the speed of the molecules match statisticaly to the average temperature, but individualy they can be slower or faster (i take here the simple representation) and therefore interact to become ice, liquid or vapor.
And moreover, every time such a molecule change its state, it will also add or remove some kinetic energy !
Quote from: chem.libretext
Phase changes are always accompanied by a change in the energy of a system. For example, converting a liquid, in which the molecules are close together, to a gas, in which the molecules are, on average, far apart, requires an input of energy (heat) to give the molecules enough kinetic energy to allow them to overcome the intermolecular attractive forces. The stronger the attractive forces, the more energy is needed to overcome them. Solids, which are highly ordered, have the strongest intermolecular interactions, whereas gases, which are very disordered, have the weakest.https://chem.libretexts.org/Bookshelves/General_Chemistry/Map%3A_Chemistry_-_The_Central_Science_(Brown_et_al.)/11%3A_Liquids_and_Intermolecular_Forces/11.04%3A_Phase_Changes
Thus any transition from a more ordered to a less ordered state (solid to liquid, liquid to gas, or solid to gas) requires an input of energy; it is endothermic.
Conversely, any transition from a less ordered to a more ordered state (liquid to solid, gas to liquid, or gas to solid) releases energy; it is exothermic.
So solid to liquid phase transformation is endothermic (it lower the kinetic energy available to the system).
And liquid to solid phase transformation is exothermic (it upgrade the kinetic energy available to the system).
When ice form, it add heat and when ice melt it lower heat.
Therefore, these transformations (you can not avoid) will produce the flow of the heat we are talking about, even if the average temperature of both phases are equal.
Post by: alancalverd on 23/06/2022 22:37:00
It is due to the fact that the speed of the molecules match statisticaly to the average temperature,If you start with an incorrect statement, you can confuse yourself.
Temperature is a measure of the average kinetic energy of the molecules in a sample, not the other way around.
Post by: Deecart on 24/06/2022 12:16:30
Quote
If you start with an incorrect statement, you can confuse yourself.
Temperature is a measure of the average kinetic energy of the molecules in a sample, not the other way around.
I am not confused : "match" is equivalent to "be linked".
But this was only to say (i sbould not have talked about temperature in my sentence) : The kinetic energy of every individual molecule differ randomly, thats all.
Post by: hamdani yusuf on 19/08/2022 11:11:19
My plan to improve the signal over noise ratio of the experiment is by improving heat insulation between the system and its surrounding. I'll build a multilayer styrofoam box, closed in all six sides. The box will be divided into 3 compartments.
Between first and second compartment is a thermal conductor, such as aluminum plate. While second compartment is separated from third compartment by thermal insulator, such as Styrofoam plate.
Second compartment will be filled by 90% ice & 10% water, while first and third compartment will be filled by 10% ice & 90% water. Heat transfer with the environment should be equal between first and third compartment. If there are different result, it must come from heat transfer with second compartment.
Is there any objection to this plan?
I finally finished the experiment, after some unexpected challenges. The Styrofoam board I used turned out to be leaky. I covered the internal sides of the Styrofoam box using the hot melt glue used to stick the boards to form the box. But it was still leaky, and the Styrofoam shrunk when overheated. Finally the problem was solved by applying wax layer on top of the glue layer, using candles and hot air gun.
The experiment itself brought its own problems. But they can finally be solved. I'll upload the video when I'm done editing it.
Post by: Origin on 19/08/2022 15:08:33
I finally finished the experiment, after some unexpected challenges. The Styrofoam board I used turned out to be leaky. I covered the internal sides of the Styrofoam box using the hot melt glue used to stick the boards to form the box. But it was still leaky, and the Styrofoam shrunk when overheated. Finally the problem was solved by applying wax layer on top of the glue layer, using candles and hot air gun.What a silly waste of time and energy. Your time would be much better spent learning some physics.
The experiment itself brought its own problems. But they can finally be solved. I'll upload the video when I'm done editing it.
Post by: hamdani yusuf on 19/08/2022 23:44:02
What a silly waste of time and energy. Your time would be much better spent learning some physics.You can't learn new things if you think you already know the answer.
Post by: Origin on 20/08/2022 01:47:29
You can't learn new things if you think you already know the answer.Yes, we do know the answer. You're not learning anything new, you are just wasting time.
The answer was given to you in the first reply and you have wasted your time and others in this never ending merry-go-round thread.
Post by: alancalverd on 20/08/2022 20:37:19
Post by: hamdani yusuf on 21/08/2022 02:48:03
You can't learn new things if you think you already know the answer.Yes, we do know the answer. You're not learning anything new, you are just wasting time.
The answer was given to you in the first reply and you have wasted your time and others in this never ending merry-go-round thread.
You won't know that your beliefs are false until experimental evidence contradicts them.You can't learn new things if you think you already know the answer.Yes, we do know the answer. You're not learning anything new, you are just wasting time.
The answer was given to you in the first reply and you have wasted your time and others in this never ending merry-go-round thread.
You won't know what factors can affect experimental results which will make you have inaccurate predictions.
Post by: hamdani yusuf on 21/08/2022 02:57:57
Let's go back to everyone's first physics lesson, or thereabouts. We learned that "temperature is the measure of hotness or coldness" (definition), and "heat flows from a hotter body to a colder body" (observation). Therefore if two bodies are at the same temperature, there can be no heat flow between them.My experiment tests the prediction made with following assumptions :
1. No heat flow without temperature difference.
2. Water-ice mixture at equilibrium in atmospheric pressure has temperature around 0 degree Celsius, irrespective of the ratio.
My experimental result indicates that at least one of them must be false.
Post by: alancalverd on 21/08/2022 10:05:32
If heat flows from A to B then by definition A is at a higher temperature than B.
At equilibrium, by definition of "equilibrium", there is no heat flow.
Post by: hamdani yusuf on 21/08/2022 12:03:46
It is not a prediction, it is a definition.Do you think that the second assumption is false?
If heat flows from A to B then by definition A is at a higher temperature than B.
At equilibrium, by definition of "equilibrium", there is no heat flow.
Post by: Bored chemist on 21/08/2022 18:39:32
What "assumption"?It is not a prediction, it is a definition.Do you think that the second assumption is false?
If heat flows from A to B then by definition A is at a higher temperature than B.
At equilibrium, by definition of "equilibrium", there is no heat flow.
Incidentally:
https://en.wikipedia.org/wiki/Zeroth_law_of_thermodynamics
Post by: alancalverd on 21/08/2022 18:53:59
Post by: Origin on 21/08/2022 21:41:13
My experiment tests the prediction made with following assumptions :So then you have your answer to this thread, which is, "Is there a net heat exchange between water and ice at 0 degree C?"
1. No heat flow without temperature difference.
Post by: hamdani yusuf on 22/08/2022 05:32:51
What "assumption"?
My experiment tests the prediction made with following assumptions :
1. No heat flow without temperature difference.
2. Water-ice mixture at equilibrium in atmospheric pressure has temperature around 0 degree Celsius, irrespective of the ratio.
My experimental result indicates that at least one of them must be false.
Post by: hamdani yusuf on 22/08/2022 05:40:59
A definition is not an assumption.Yes, it is. Here's an example. How do you define the color red?
Post by: hamdani yusuf on 22/08/2022 05:42:20
Post by: hamdani yusuf on 22/08/2022 05:45:59
Are you satisfied with making assumptions without trying to confirm or refute them with evidence?My experiment tests the prediction made with following assumptions :So then you have your answer to this thread, which is, "Is there a net heat exchange between water and ice at 0 degree C?"
1. No heat flow without temperature difference.
Post by: Bored chemist on 22/08/2022 10:39:31
If you were asking about your assumption(s) why did you say it immediately after quoting someone else?What "assumption"?My experiment tests the prediction made with following assumptions :
1. No heat flow without temperature difference.
2. Water-ice mixture at equilibrium in atmospheric pressure has temperature around 0 degree Celsius, irrespective of the ratio.
My experimental result indicates that at least one of them must be false.
Anyway, your error is in assuming that your experiment indicates that one of the assumptions is false.
The result of your experiment indicates that you have a heat leak somewhere.
Post by: alancalverd on 22/08/2022 11:38:42
How do you define the color red?A spectrum of photons predominantly in the region of 620 - 780 nm wavelength.
Post by: hamdani yusuf on 22/08/2022 11:39:29
If you were asking about your assumption(s) why did you say it immediately after quoting someone else?I answered your question.
Post by: hamdani yusuf on 22/08/2022 11:42:32
How did you get those numbers?How do you define the color red?A spectrum of photons predominantly in the region of 620 - 780 nm wavelength.
How do you define predominantly? Is 50.1% of intensity enough?
Post by: hamdani yusuf on 22/08/2022 11:46:54
The result of your experiment indicates that you have a heat leak somewhere.Anyone can easily try to replicate my experiment. I've shown step by step process clearly.
I checked the outer surface of the container, and found it dry. No indication of water leakage nor condensation due to thermal leakage.
Post by: alancalverd on 22/08/2022 11:47:18
Here's the experiment. What do you think?The invalid extrapolation occurs at 1:48 or thereabouts.
A domestic freezer operates at -5°C or lower, so the center of any remaining block of ice is below 0°C and the subsequent mixture cannot be considered homogeneous, nor can any chunk of ice be considered necessarily typical.
As I have pointed out several times before, heat experiments are not for the lazy or fainthearted.
Post by: hamdani yusuf on 22/08/2022 11:52:17
Anyway, your error is in assuming that your experiment indicates that one of the assumptions is false.My experiment indicates that there is heat transfer. Otherwise the ice blocks would completely melt in around the same time.
Hence, it's impossible if both assumptions above are true.
Post by: hamdani yusuf on 22/08/2022 11:56:28
But the aluminum sheet doesn't contact the center of the ice chunk. It touches the water.Here's the experiment. What do you think?The invalid extrapolation occurs at 1:48 or thereabouts.
A domestic freezer operates at -5°C or lower, so the center of any remaining block of ice is below 0°C and the subsequent mixture cannot be considered homogeneous, nor can any chunk of ice be considered necessarily typical.
As I have pointed out several times before, heat experiments are not for the lazy or fainthearted.
Post by: alancalverd on 22/08/2022 12:02:08
How did you get those numbers?It's the physical definition of "red" as you asked for. Many common adjectives are defined by numbers, e.g. 30 mph may determine "fast" in a particular context. For complex visual spectra there is an international "Pantone" scale and subsets such as BS4800 for specifying paint.
Note that "dominant" merely assumes an average human eye. Some folk can't distinguish physical or standard red signals from yellow or even blue, and the sensitivity of the "normal" eye varies with wavelength, peaking strongly in the yellow-green range, hence (a) the development of orthochromatic and panchromatic photographic films to map an image into consistently interpretable shades of grey and (b) color blindness tests for professional drivers and air crew. Interestingly, though, some forms of color blindness actually improve the detection of camouflaged structures and are prized by the military.
Post by: Bored chemist on 22/08/2022 12:02:22
Anyone can easily try to replicate my experiment.What would be the point?
Post by: alancalverd on 22/08/2022 12:05:16
But the aluminum sheet doesn't contact the center of the ice chunk. It touches the water.which touches the surface of the ice, which touches the middle of the ice.
Post by: hamdani yusuf on 22/08/2022 15:55:37
What is the temperature of the water? Does it ever go below 0 degree Celsius?But the aluminum sheet doesn't contact the center of the ice chunk. It touches the water.which touches the surface of the ice, which touches the middle of the ice.
Post by: alancalverd on 22/08/2022 16:03:46
Post by: hamdani yusuf on 22/08/2022 16:07:03
So anyone who are sceptical about my experimental result can try to falsify it or prove it themselves. If your convictions are not affected by contradicting evidences, they are not scientific.Anyone can easily try to replicate my experiment.What would be the point?
Post by: Bored chemist on 22/08/2022 20:03:19
If your convictions are not affected by contradicting evidences, they are not scientific.This
If heat flows from A to B then by definition A is at a higher temperature than B.is logic.
At equilibrium, by definition of "equilibrium", there is no heat flow.
If you are not accepting it, you are not doing science.
It's not my job to find the errors in your experiment; that's your responsibility.
Post by: Bored chemist on 22/08/2022 20:17:32
Here's the experiment. What do you think?At 5:04 you say "the difference can only be explained by..."
and that's wrong.
You are assuming that the water in the two outside compartments is all kept at 0C.
But it won't be.
The water at the bottom will warm up to 4C or so (the temperature of maximum density.)
How thick the layer of relatively warm water is will depend on how well lagged the container is.
But, in one case, the water is not only being cooled by the ice cube, it is being cooled by all the ice in the centre container.
That's why the ice which is cooled by heat transfer through the aluminium stays cooler than the ice which is insulated from that big supply of ice.
Imagine that you just put the ice and water in the middle container and left the two outer ones empty.
And then imagine that you put a thermometer in each of the two outer containers (and put the lid on).
Would you not expect the temperature in the left side to be lower than that in the right- because it is "connected" to the ice by the aluminium whereas the chamber on the right is insulated from the ice?
That's why the ice on the better lagged side melts quicker.
It's not being cooled by all that ice in the middle section.
Post by: alancalverd on 22/08/2022 22:17:25
Heat experiments are very difficult and it takes engineering genius, cunning and patience to improve on 1% accuracy. Domestic freezers, tap water and styrofoam won't challenge the answer in the textbook. Failure with school-level or even undergraduate equipment is no disgrace, and I admire HY's enthusiasm and audacity with very simple kit, but it sometimes looks like obsession rather than science.
Post by: hamdani yusuf on 22/08/2022 22:40:49
the ambient temperature is above melting point.I said in the video, the ambient is at room temperature.
Post by: hamdani yusuf on 22/08/2022 23:23:58
Is there any heat flow in melting ice? Is there any temperature difference between water and ice?If your convictions are not affected by contradicting evidences, they are not scientific.ThisIf heat flows from A to B then by definition A is at a higher temperature than B.is logic.
At equilibrium, by definition of "equilibrium", there is no heat flow.
If you are not accepting it, you are not doing science.
It's not my job to find the errors in your experiment; that's your responsibility.
Post by: Origin on 22/08/2022 23:40:56
Is there any heat flow in melting ice?Of course! Why do you think it is melting?
Is there any temperature difference between water and ice?There can be a difference or the temperature can be the same.
Post by: hamdani yusuf on 23/08/2022 09:46:31
So, heat flow can occur without temperature difference?Is there any heat flow in melting ice?Of course! Why do you think it is melting?Is there any temperature difference between water and ice?There can be a difference or the temperature can be the same.
Post by: alancalverd on 23/08/2022 11:15:43
Post by: hamdani yusuf on 23/08/2022 11:27:49
For the last time, no. By definition, not experiment.Then there's temperature difference between water and ice while melting.
Post by: alancalverd on 23/08/2022 13:35:27
HOWEVER water below 0°C will indeed melt ice if the water contains almost any soluble impurity, which is how we clear snow and ice by spraying with salt, and why icebergs melt in the arctic sea (about -2°C). Domestic tap water contains all sorts of solutes.
Yet again: heat experiments are very difficult. Particularly if they involve liquids, and most particularly if the liquids display anomalous convection - water being the most obvious. Gases and solids are relatively well-behaved but still tricky.
Post by: Origin on 23/08/2022 13:39:21
So, heat flow can occur without temperature difference?No. Why would you ask such an absurd question? If the ice is melting the there is obviously a temperature difference between the water and the ice.
Post by: Origin on 23/08/2022 13:41:23
Then there's temperature difference between water and ice while melting.See that wasn't difficult, it only took 345 posts.
Post by: hamdani yusuf on 23/08/2022 23:31:06
At 5:04 you say "the difference can only be explained by..."Of course it won't. A tripple dot won't explain anything.
and that's wrong.
You are assuming that the water in the two outside compartments is all kept at 0C.
But it won't be.
Post by: hamdani yusuf on 23/08/2022 23:33:16
So, heat flow can occur without temperature difference?No. Why would you ask such an absurd question? If the ice is melting the there is obviously a temperature difference between the water and the ice.
Then why did you write this?
There can be a difference or the temperature can be the same.
Post by: Origin on 24/08/2022 03:12:49
Is there any temperature difference between water and ice?I didn't realize this was in relation to melting ice. The answer is that the water temperature is higher than the ice. That is why the ice melts, it is really not that complicated.
Post by: hamdani yusuf on 24/08/2022 06:42:13
If I didn't publish my experimental result, you would still be thinking that there's no significant difference between compartment 1 and 3, because you think that there would be no heat flow to compartment 2 from them.Then there's temperature difference between water and ice while melting.See that wasn't difficult, it only took 345 posts.
Post by: hamdani yusuf on 24/08/2022 07:01:38
Is there any temperature difference between water and ice?I didn't realize this was in relation to melting ice. The answer is that the water temperature is higher than the ice. That is why the ice melts, it is really not that complicated.
Now you are contradicting BC, instead of me.
It does; it's just that you don't understand it.Here's how they did it a few hundred years ago.It doesn't show that the ice and water are at exactly the same temperature at every point.
https://www.nature.com/scitable/content/ice-calorimeter-developed-by-lavoisier-and-laplace-14898943/
If you have ice and water at equilibrium, in a closed vessel surrounded by ice and water, what other temperature can it be apart from 0C?
Consider the following before you answer.
(1) That if you add heat to the system, you won't change the temperature, you will just met some ice.
(2) That if you remove heat from the system, you won't reduce the temperature, you will just freeze some water
(3) Since the ice and water inside the container is at the same temperature as the ice and water outside it, there is no temperature gradient across the container wall, and therefore no heat transfer.
So, if there was a transfer of heat, the temperature wouldn't change, and there's no mechanism for a transfer of heat anyway.
Post by: Bored chemist on 24/08/2022 08:42:56
Now you are contradicting BC, instead of me.Not really.
There's a difference between (mixed) ice and water - which will maintain 0C and water with a single ice cube floating in it.
But, in any event, I explained your experimental result" - it arose from poor experimental design.
Do you accept that?
Post by: hamdani yusuf on 24/08/2022 09:22:18
You are assuming that the water in the two outside compartments is all kept at 0C.
But it won't be.
The water at the bottom will warm up to 4C or so (the temperature of maximum density.)
How thick the layer of relatively warm water is will depend on how well lagged the container is.
But, in one case, the water is not only being cooled by the ice cube, it is being cooled by all the ice in the centre container.
That's why the ice which is cooled by heat transfer through the aluminium stays cooler than the ice which is insulated from that big supply of ice.
I've raised the conern previously, which you confidently dismissed.
That's what I found as experimental results. Ice-water mixture don't maintain homogeneous temperature. Difference in density tends to make bottom part of the bath warmer than the surface.Convection will make temperature of the bath more uniform around 0C.No. Convection requires a temperature gradient.
The temperature gradient will form naturally through buoyancy.No it will not.
Because, if all the water is at 0 C then it all has the same density and there can not be a gradient.
Why don't you understand that?
Post by: Bored chemist on 24/08/2022 10:49:19
if all the water is at 0 CThe point remains; if you have a temperature gradient, you don't have ice and water in equilibrium (and it's also not the conditions you specify in the title of the thread).
Post by: alancalverd on 24/08/2022 12:44:03
As I mentioned in a previous post, doing precise heat experiments with liquids is always difficult and among the "simple" liquids, water is particularly so because of its anomalous convection near the freezing point.
When we do near-equilibrium measurements with liquids, we like to use a very large volume so that heat transfer across the surfaces is minimised in comparison with the heat capacity of the experimental fluid, then make our measurements near the centre. Then you can either leave it alone for a very long time to settle any swirling current (I recently detected swirl currents in a 40 liter sphere, an hour after we moved it just 1 meter into an MRI unit, and we always allowed 50-liter water calorimeters to settle for at least 12 hours before using them to measure radiation dose) OR use a paddle stirrer to induce sufficiently random turbulence that an average measurement has some validity (but remember to allow for the energy introduced by the paddle).
Getting an ice-water mixture to anything like equilibrium in practice requires constant stirring in three dimensions as you slowly cool the sample into a homogeneous slush. It is not for the faint-hearted or anyone with less than a year, a decent workshop, and a few thousand dollars to spend on getting it right.
Which is why we have textbooks written by well-funded national laboratories, to tell us the answer.
Post by: Origin on 24/08/2022 13:08:57
Now you are contradicting BC, instead of me.Nope, there is no contradiction. Water and ice in equilibrium will both be at 0C. A container with ice melting in water is not in equilibrium and so the temperature of the ice and water are not both exactly 0C, the water will be slightly warmer. If there was no difference in temperature there would be no heat transfer.
If I didn't publish my experimental result, you would still be thinking that there's no significant differenceDo you actually believe that your experiment yielded any useful information? Let me assure you it did not.
Post by: hamdani yusuf on 25/08/2022 09:14:02
So, you don't know what "if" means.Initially, they are at 0° C.if all the water is at 0 CThe point remains; if you have a temperature gradient, you don't have ice and water in equilibrium (and it's also not the conditions you specify in the title of the thread).
My experiment gives evidence that water-ice mixture can't maintain the temperature equilibrium, even when heat transfer with the environment has been significantly reduced.
It's not as easy as you thought it would be.
It is easy.Not really. Get some ice, a little below freezing , put it in a closed container and put that in a slurry of crushed ice and water.What's hard is getting the left compartment filled with pure ice while the right compartment contains pure water. We will need to maintain air temperature at 0°C, so is the tools we use to transport the water and ice. The lighting should also be taken into account, as well as body temperature of the experimenter.
It will warm up to exactly 0C
Get some water, just above freezing, Seal it in a container and put it in a slurry of ice and water, it will cool down to exactly 0C.
You do everything in a tank of ice cold water.
Post by: Bored chemist on 25/08/2022 10:45:00
It's not as easy as you thought it would be.Have you tried?
when heat transfer with the environment has been significantly reduced.The point is that "reduced" isn't good enough, is it?
If you have heat flowing in, you don't have an equilibrium so this
can't maintain the temperature equilibriumis meaningless.
It's now pretty clear that this thread has no point.
Post by: hamdani yusuf on 25/08/2022 23:23:08
Do you actually believe that your experiment yielded any useful information? Let me assure you it did not.If you already have a conclusion which is unfalsifiable and unaffected by experimental results, then my experiments would be useless to you. But they might by useful for anyone else trying to stabilize temperature of a system in a real world environment.
Post by: hamdani yusuf on 26/08/2022 05:12:24
Have you tried?Yes. Read again my posts here.
If I can isolate the system completely by any way, I'd like to test it.
Post by: Bored chemist on 26/08/2022 10:40:20
But they might by useful for anyone else trying to stabilize temperature of a system in a real world environment.If that person hadn't previously been aware of an ice bath as a way to get some degree of temperature regulation.
Yes. Read again my posts here.I did not notice the post in which you adopted my suggestion and enclosed the whole apparatus in a tank containing a slurry of ice and water.
Please point it out.
If I can isolate the system completely by any way, I'd like to test it.You can...enclose the whole apparatus in a tank containing a slurry of ice and water.
Post by: alancalverd on 26/08/2022 14:23:26
f I can isolate the system completely by any way, I'd like to test it.It cannot be done, which is why heat experiments are very difficult.
BC's slurry tank will get you some of the way, maybe with in 0.1°C of constant temperature, but you need to keep stirring the slurry to stop the bottom of the tank getting warmer. If you add more ice, you will cool the top below 0°C, so you have to choose the right moment to do your measurements. And you need to seal the top of your experimental rig to stop ambient air getting in. Problem is that with ±0.1°C uncertainly, you won't be working anywhere near close to the textbook statement of "equilibrium". And you still have the problem of not knowing the temperature in the middle of your experimental ice chunks, even if the exterior slurry is well mixed.
So you would probably do better to use a thermostat tank of glycol antifreeze and a chiller coil to bring it down to zero. Keep stirring but at least you know it won't contain any chunks of sub-zero ice or anomalous convection. Then prepare your experimental ice by some magical method that doesn't involve a sub-zero chiller.
Post by: Deecart on 26/08/2022 15:14:01
So, heat flow can occur without temperature difference?
By definition, no.
Heat is a macroscopical physical quantity.
It is the energy of some mass at some velocity.
But if you go further and further with less and less mass (at least you end up with some single molecules, and here you go out of the thermodynamic domain and enter into the more fundamental physic), you end up with local interactions that do not have the behavior of the macroscopical law.
So (at equilibrum) you end up with "fluctuations", no "flows".
The fluctuations also exists within the macroscopical domain, but they are discarded (because of the null result at this scale).
So when you talk about heat flow, like when you talk of every flow (flow of liquid in the river per example), you have local molecules doing random transitions ("at equilibrum) having nothing to do with the macroscopical law.
Some water molecules go temporaly up into the river !
To summarize :
At macroscopical level, you have 2 phenomenon occuring : The flow and the fluctuation.
Post by: Bored chemist on 26/08/2022 16:22:11
I didn't specify all the experimental details of how you make sure the temperature is maintained rather better than 0.1C.f I can isolate the system completely by any way, I'd like to test it.It cannot be done, which is why heat experiments are very difficult.
BC's slurry tank will get you some of the way, maybe with in 0.1°C of constant temperature, but you need to keep stirring the slurry to stop the bottom of the tank getting warmer. If you add more ice, you will cool the top below 0°C, so you have to choose the right moment to do your measurements. And you need to seal the top of your experimental rig to stop ambient air getting in. Problem is that with ±0.1°C uncertainly, you won't be working anywhere near close to the textbook statement of "equilibrium". And you still have the problem of not knowing the temperature in the middle of your experimental ice chunks, even if the exterior slurry is well mixed.
So you would probably do better to use a thermostat tank of glycol antifreeze and a chiller coil to bring it down to zero. Keep stirring but at least you know it won't contain any chunks of sub-zero ice or anomalous convection. Then prepare your experimental ice by some magical method that doesn't involve a sub-zero chiller.
The most notable is that you circulate the water with an external pump and ensure that , just before it reaches the "experimental" tank, it flows through a chamber containing lots of ice. If some of that ice is a little below 0C it doesn't matter because it won't supercool the water. The advantage to using water and ice, rather than a cooling coil and glycol is that it's "self calibrating"
You are, of course, perfectly correct in saying it's very difficult.
I have to admit the technique isn't really "mine". It was developed by some "famous names".
https://www.nature.com/scitable/content/ice-calorimeter-developed-by-lavoisier-and-laplace-14898943/
I think you and I could happily while away an hour or two discussing tweaks to improve thermal isolation over a glass of something, that's not really the point.
My point was that, while the answer given to
Have you tried?was
Yes.it seems pretty clear that the correct answer is "no".
Post by: alancalverd on 26/08/2022 18:46:14
Using a well-stirred circulating glycol tank with an external chiller (thanks, BC) you can gradually reduce the temperature of a gently-stirred sample of pure water. The volume of the sample will increase as ice forms. Now hold the temperature at 0°C. If there is a heat flow between the water and the ice, the sample volume will change as the water melts the ice or vice versa.
Post by: hamdani yusuf on 27/08/2022 04:35:43
My experiment shows the limitation.But they might by useful for anyone else trying to stabilize temperature of a system in a real world environment.If that person hadn't previously been aware of an ice bath as a way to get some degree of temperature regulation.
Post by: hamdani yusuf on 27/08/2022 04:38:19
I did not notice the post in which you adopted my suggestion and enclosed the whole apparatus in a tank containing a slurry of ice and water.
Please point it out.
Here's my idea to minimize noise over signal:OK. I've finished my first round of experiment as described above. But I can't get the metal cups, so I just used ordinary drinking glasses. I guess the heat conductance is enough for this experiment since they are quite thin.
- Prepare 50/50 ice-water mixture at around 0°C in a large plastic bowl. Let it in refrigerator for an hour to reach equilibrium.
- Fill a metal cup with 90% water and 10% ice from the mixture.
- Fill another metal cup with 10% water and 90% ice from the mixture.
- Put both metal cups into the bowl containing the remaining of the mixture.
- Let them in refrigerator for an hour to reach equilibrium.
- See the result, if the ratio of ice-water in the cups change.
The temperature of the refrigerator is 4C, as shown by a thermometer left there for an hour. The result is the ice in both glasses decreased from initial ratio.
So I moved the system to the freezer, which is kept at -4C, as measured by a thermometer left there for an hour. The result is the ice in both glasses increased from initial ratio.
These results show that energy transfer between the system and the environment overwhelmed the energy transfered through the glasses. It means that the noise over signal ratio is too high to get reliable conclusion. Hence the experimental setups need to be improved.
I also learned that gravity and buoyancy play significant role in determining the results, which need to be addressed in upcoming experiments.
Post by: hamdani yusuf on 27/08/2022 04:48:12
I have to admit the technique isn't really "mine". It was developed by some "famous names".(https://www.nature.com/scitable/content/ne0000/ne0000/ne0000/ne0000/14898943/f1_dapoian_new1.jpg)
https://www.nature.com/scitable/content/ice-calorimeter-developed-by-lavoisier-and-laplace-14898943/
The top side looks uninsulated, which can cause heat leakage.
Post by: Bored chemist on 27/08/2022 12:16:57
The result is the ice in both glasses decreased from initial ratio.That's the bit which proves that you hadn't thermally isolated them.
And that shows that they were no surrounded by material at 0C.
What sort of lids did you use on the cups?
Post by: hamdani yusuf on 28/08/2022 10:00:55
What sort of lids did you use on the cups?Just a simple plastic plate. I thought the air below it would provide adequate heat insulation.
Post by: Bored chemist on 28/08/2022 10:05:12
So, the test samples weren't actually completely surrounded by ice/water?What sort of lids did you use on the cups?Just a simple plastic plate. I thought the air below it would provide adequate heat insulation.
Post by: JosephCook on 28/08/2022 12:22:46
I'm now considering the matter for amusement only, but it does while away the time on the M25 when all else is silent and unmoving. Here's a suggestion:Uhm, your theory sounds interesting, have you tried it?
Using a well-stirred circulating glycol tank with an external chiller (thanks, BC) you can gradually reduce the temperature of a gently-stirred sample of pure water. The volume of the sample will increase as ice forms. Now hold the temperature at 0°C. If there is a heat flow between the water and the ice, the sample volume will change as the water melts the ice or vice versa.
Post by: hamdani yusuf on 28/08/2022 12:56:51
So, the test samples weren't actually completely surrounded by ice/water?No.
I'll submerge them in next experiment.
Post by: Bored chemist on 28/08/2022 13:31:40
No.So, just to clarify things, when you said
Yes.You actually meant
no
Post by: alancalverd on 28/08/2022 13:33:37
Uhm, your theory sounds interesting, have you tried it?I have used ice/water calorimetry in the past, and as I keep repeating, it is extremely difficult, even with the resources of a national standards laboratory, to do well. To attempt to use it to disprove the most fundamental statement of thermodynamics using domestic kitchen equipment, is beyond foolish.
Post by: Bored chemist on 28/08/2022 14:02:11
To attempt to use it to disprove the most fundamental statement of thermodynamics using domestic kitchen equipment, is beyond foolish.And thus Alan wins today's award for "most understated assertion".
Post by: hamdani yusuf on 29/08/2022 12:49:43
I have tried several tweaks to the experiment. That's what yes means. You seem to have short memory problem, which makes you often make straw man argument.No.So, just to clarify things, when you saidYes.You actually meantno
I replaced the glasses with small plastic bags, which were almost completely submerged. But the results were the same. That's why I tried to find another method.
Quote
I also learned that gravity and buoyancy play significant role in determining the results, which need to be addressed in upcoming experiments.
Post by: hamdani yusuf on 29/08/2022 12:59:02
Do you cover the top of the system with water and ice mixture?Uhm, your theory sounds interesting, have you tried it?I have used ice/water calorimetry in the past, and as I keep repeating, it is extremely difficult, even with the resources of a national standards laboratory, to do well. To attempt to use it to disprove the most fundamental statement of thermodynamics using domestic kitchen equipment, is beyond foolish.
I have to admit the technique isn't really "mine". It was developed by some "famous names".(https://www.nature.com/scitable/content/ne0000/ne0000/ne0000/ne0000/14898943/f1_dapoian_new1.jpg)
https://www.nature.com/scitable/content/ice-calorimeter-developed-by-lavoisier-and-laplace-14898943/
The top side looks uninsulated, which can cause heat leakage.
Post by: JosephCook on 29/08/2022 13:15:27
Got it. Thanks for your answer. I am impressed even by your trial, good one.Uhm, your theory sounds interesting, have you tried it?I have used ice/water calorimetry in the past, and as I keep repeating, it is extremely difficult, even with the resources of a national standards laboratory, to do well. To attempt to use it to disprove the most fundamental statement of thermodynamics using domestic kitchen equipment, is beyond foolish.
Post by: Bored chemist on 29/08/2022 13:35:35
It's irrelevant.
Can you quote the post where you said that you did this experiment properly- well lagged and submerged in well stirred ice and water and told us how you did it and what the results were?
Post by: hamdani yusuf on 30/08/2022 17:40:14
I know what a straw man argument is.I thought you already knew what the results would be before doing the experiment.
It's irrelevant.
Can you quote the post where you said that you did this experiment properly- well lagged and submerged in well stirred ice and water and told us how you did it and what the results were?
I'm keeping the raw footage of the "failed" experiments. I should have publised all of them, but editing the video and adding narration are time consuming. I choose to spend that time to do something more productive. Although it's possible that I 'll just compile and upload them as behind the scenes for the conclusive result.
Post by: Bored chemist on 30/08/2022 18:29:44
I choose to spend that time to do something more productive.LOL
Post by: hamdani yusuf on 30/08/2022 22:36:04
It won't.It seems that the energy flow can be driven by gravity through buoyancy and temperature difference. So, if a liter of ice-water mixture is in an insulated container, and initially well mixed, the top side will gradually become cooler than the bottom side. The ice will be naturally separated from the water through buoyancy.
Because, that would require the transfer of heat to or from teh ice and, because everything is at the same temperature, there is no impetus to drive the energy from one place to another.
Why do you not understand and accept this?
Which bit does not make sense to you?
Heat conductance of water will reduce the difference. But given adequately tall container, its effect can be minimized.
Post by: Bored chemist on 30/08/2022 22:46:10
It seems that the energy flow can be driven by gravity through buoyancy and temperature difference.You can only use temperature difference to move energy if you actually have a temperature difference.
Do you not realise that you ruled that out when you asked "Is there a net heat exchange between water and ice at 0 degree C?"?
If it's all at 0C there is no temperature difference.
The ice will be naturally separated from the water through buoyancy.Yes, ice floats
the top side will gradually become cooler than the bottom side.How?
Where is the energy coming from or going to?
What is providing the impetus to drive that energy transfer?
Post by: hamdani yusuf on 01/09/2022 13:05:43
The energy is provided by the environment, because practically, insulations can't be perfect. Even if the insulation is also made of a bath of ice-water mixture, to several layer like Russian dolls. The outer most layer will still interact with environment, which is then propagated to the inner layers.It seems that the energy flow can be driven by gravity through buoyancy and temperature difference.You can only use temperature difference to move energy if you actually have a temperature difference.
Do you not realise that you ruled that out when you asked "Is there a net heat exchange between water and ice at 0 degree C?"?
If it's all at 0C there is no temperature difference.The ice will be naturally separated from the water through buoyancy.Yes, ice floatsthe top side will gradually become cooler than the bottom side.How?
Where is the energy coming from or going to?
What is providing the impetus to drive that energy transfer?
The other factor is temperature variation inside the system itself. Slight temperature difference due to quantum fluctuation might make slight difference of density, which makes hotter molecules tend to move downward. This effect can be nullified by heat conductance of water. Which effect is stronger can be shown by an experiment.
Post by: alancalverd on 01/09/2022 13:30:58
The energy is provided by the environment, because practically, insulations can't be perfect.And that's most of the problem with heat experiments. Electrical insulators are zillions of times less conductive than heat insulators even if they are made of the same stuff.
Quote
So, if a liter of ice-water mixture is in an insulated container, and initially well mixed, the top side will gradually become cooler than the bottom side. The ice will be naturally separated from the water through buoyancy.Told you so, many times. Which is why you need a continuously-stirred slush. Melting snow is a good start.
Post by: Bored chemist on 01/09/2022 13:52:09
The energy is provided by the environment, because practically, insulations can't be perfect.In which case, you do not meet the requirement stated in the title
"Is there a net heat exchange between water and ice at 0 degree C?"
Why do you keep going round in circles about this?
which is then propagated to the inner layers.Are you deliberately missing the point?
Heat leaks in through the outermost layer.
And it melts ice.
So the temperature there stays at 0C.
So the inner layers are totally isolated.
Now, I realise that, in practice, this is difficult.
But, in principle, the process works.
Which effect is stronger can be shown by an experiment.Or by calculation.
Quantum effects are small.
Post by: hamdani yusuf on 01/09/2022 16:02:52
Told you so, many times. Which is why you need a continuously-stirred slush. Melting snow is a good start.I'm thinking about circulating the cool water using a small pump, like the one used in aquariums.
Post by: Bored chemist on 01/09/2022 17:58:18
Don't forget to allow for the heat it adds.Told you so, many times. Which is why you need a continuously-stirred slush. Melting snow is a good start.I'm thinking about circulating the cool water using a small pump, like the one used in aquariums.
Post by: alancalverd on 01/09/2022 23:51:33
I'm thinking about circulating the cool water using a small pump, like the one used in aquariums.Better to use BC's suggestion of an"extracorporeal" pump and chiller. You can get a good one from a cardiac surgical unit, though we generally run them at something closer to body temperature.
Post by: hamdani yusuf on 02/09/2022 12:21:35
Alternatively, I can use ice blocks held to the bottom of the container using a cage. Slightly warmer water coming down to the bottom will be cooled down by the ice, and flow back up again, producing a natural circulation that stabilizes temperature and makes it more uniform.Don't forget to allow for the heat it adds.Told you so, many times. Which is why you need a continuously-stirred slush. Melting snow is a good start.I'm thinking about circulating the cool water using a small pump, like the one used in aquariums.
Post by: Bored chemist on 02/09/2022 13:21:23
What do you think dives the currents?
Did you think it was magic?
Post by: alancalverd on 02/09/2022 13:23:53
Post by: hamdani yusuf on 02/09/2022 15:25:52
Do you really not understand that, if you have convection currents your system is not at 0oC?It looks like you have difficulties to understand more than one layer of conversation. Why do you think I need to use a pump in the first place?
What do you think dives the currents?
Did you think it was magic?
Post by: hamdani yusuf on 02/09/2022 15:28:59
To repeat the obvious answer to the original question: No, by definition.How does it help to predict experimental results?
Post by: Bored chemist on 02/09/2022 16:03:26
Why do you think I need to use a pump in the first place?Because you can't rely on convection currents, and the ice/ water system needs to be well stirred.
Now, try actually answering this.
Do you really not understand that, if you have convection currents your system is not at 0oC?Especially in the context of
What do you think dives the currents?
Did you think it was magic?
I can use ice blocks held to the bottom of the container using a cage. Slightly warmer water coming down to the bottom will be cooled down by the ice, and flow back up again, producing a natural circulation
Once you have "slightly warmer water" you are no longer at 0oC
Post by: alancalverd on 02/09/2022 18:14:28
It predicts them absolutely, fully, and completely.To repeat the obvious answer to the original question: No, by definition.How does it help to predict experimental results?
But only if you do a very good experiment, and that's very difficult with water.
Post by: hamdani yusuf on 04/09/2022 04:00:07
Geocentric model predicts planet positions accurately, as long as you add enough appropriate epicycles to it. Someone might insist that planets move around the earth by definition.It predicts them absolutely, fully, and completely.To repeat the obvious answer to the original question: No, by definition.How does it help to predict experimental results?
But only if you do a very good experiment, and that's very difficult with water.
Post by: hamdani yusuf on 04/09/2022 04:04:10
Once you have "slightly warmer water" you are no longer at 0oCThe system's average temperature can be still at 0C if another part has slightly lower temperature.
Post by: alancalverd on 04/09/2022 09:26:31
I have $1,000,000, you have $0.
You say I have more money than you, so I owe you $500,000
I say our average balance if $500,000 so I don't have to give you anything.
Post by: Bored chemist on 04/09/2022 09:42:56
Geocentric model predicts planet positions accurately, as long as you add enough appropriate epicycles to it. Someone might insist that planets move around the earth by definition.If they are doing that since Foucault did a better experiment then they are not scientists.
More importantly, they would need to explain why there are epicycles.
Post by: alancalverd on 04/09/2022 11:07:39
Post by: hamdani yusuf on 06/09/2022 23:49:48
What is polycrystalline water?
Quote
Episode 2 of my series on water ice! Admittedly this is probably the least interesting because it's some negative results, but it's not very scientific to only discuss successes right? In this video, I use a lens to melt ice from the inside out, exposing it's polycrystalline structure (and explain how it's different from a single crystal). With this partially-melted ice, I can do a bit of materials forensics, and actually figure out how it formed, hopefully so I can improve the process in the future!
Next video will be on vapor growth, and I'll say right now that it DOES work to grow centimeters-big faceted single crystals! Subscribe to make sure you see it!
Post by: Bored chemist on 07/09/2022 08:41:42
What is polycrystalline water?What do you think?
Post by: alancalverd on 07/09/2022 09:29:32
Here's a video from Alpha Phoenix, which may provide useful information to consider in designing experiment with water and ice.Well I'll be blowed! You add energy to ice, and it melts! Who would have thought it?
Post by: hamdani yusuf on 21/09/2022 13:48:03
The heat generated in the center of the ice block is higher than the outer sides, even when it's farther away from the heat source.Here's a video from Alpha Phoenix, which may provide useful information to consider in designing experiment with water and ice.Well I'll be blowed! You add energy to ice, and it melts! Who would have thought it?
Post by: alancalverd on 21/09/2022 13:55:09
Post by: hamdani yusuf on 21/09/2022 16:41:33
It can be seen in microwave ovens.
Post by: Bored chemist on 21/09/2022 19:44:29
If you think there is, which way does it go?
When does it stop?
Post by: hamdani yusuf on 21/09/2022 23:54:24
"Is there a net heat exchange between water and ice at 0 degree C?"Here are my considerations :
If you think there is, which way does it go?
When does it stop?
Temperature is an average statistical value.
Some points in an object has more kinetic energy than another point, which vary in time.
Heat flows from points with locally-temporally higher kinetic energy to another point in thermal contact with them.
Water has higher heat content than ice, when they are at the same temperature and pressure.
Post by: Bored chemist on 22/09/2022 09:02:12
Post by: alancalverd on 22/09/2022 10:17:23
Post by: Origin on 22/09/2022 13:30:26
Let's try a different analogy.I'm afraid Hamdani Yusuf is never going to get it, he still doesn't know what temperature is...
Post by: hamdani yusuf on 22/09/2022 13:38:57
You seem to have forgotten to answer the questions.I thought you are smart enough to conclude based on those considerations.
Post by: hamdani yusuf on 22/09/2022 13:40:19
You don't seem to know either. At least I don't pretend to know something that I don't know.Let's try a different analogy.I'm afraid Hamdani Yusuf is never going to get it, he still doesn't know what temperature is...
Post by: Bored chemist on 22/09/2022 14:06:34
Did you consider the idea that, no matter how smart I may or may not be, you didn't provide enough information.You seem to have forgotten to answer the questions.I thought you are smart enough to conclude based on those considerations.
So, for the third time
"Is there a net heat exchange between water and ice at 0 degree C?"
If you think there is, which way does it go?
When does it stop?
Post by: hamdani yusuf on 22/09/2022 14:30:02
Let's try a different analogy. Consider two batteries, each with a potential of 1.5 volts. Connect them in parallel. Does any current flow from one to the other? Temperature is the thermal analog of electrical potential.All analogies break down at some points, where they don't resemble the things that they are supposed to represent anymore. Your batteries don't experience phase changes. Hence they are more like two ice blocks with the same temperature.
Post by: hamdani yusuf on 22/09/2022 15:15:19
I don't have enough convincing information to confidently predict the results. That's why I started the thread and did the experiments.Did you consider the idea that, no matter how smart I may or may not be, you didn't provide enough information.You seem to have forgotten to answer the questions.I thought you are smart enough to conclude based on those considerations.
So, for the third time"Is there a net heat exchange between water and ice at 0 degree C?"
If you think there is, which way does it go?
When does it stop?
If it turns out that there's a net heat transfer, it would be from higher heat content to the lower one, i. e. from water to ice.
It would stop when both containers have the same ratio of water and ice. Hence energy transfer rate would be balanced in both directions.
Do you have any objections to the consideration points I mentioned earlier?
Post by: hamdani yusuf on 22/09/2022 15:19:59
https://en.m.wikipedia.org/wiki/Freezing-point_depression
Here I used tap water to avoid supercooling which might occur if pure water is used instead. But it might generate another problem which is not negligible.
Post by: alancalverd on 22/09/2022 17:57:33
All analogies break down at some points, where they don't resemble the things that they are supposed to represent anymore. Your batteries don't experience phase changes. Hence they are more like two ice blocks with the same temperature.I haven't specified the nature of either battery, only its potential. One could be a capacitor with a solid dielectric, the other a wet chemical cell with the same terminal voltage. That's all we know or need to know about the temperature of a body: its ability to transfer energy to or from another body. Equilibrium is equilibrium!
Post by: Bored chemist on 22/09/2022 19:11:27
It would stop when both containers have the same ratio of water and ice.How would they "know" when the ratio was 50%50?
The simple answer is that they can't.
And that's why we know that there's no heat transfer.
But we have been telling you that for 9 pages now, so I doubt you will accept it.
Post by: Bored chemist on 22/09/2022 19:15:21
Does anyone considered this to make their predictions?No.
Because it's irrelevant to the question which you actually asked which was about water and ice not "some random solution of stuff".
Post by: alancalverd on 22/09/2022 20:58:13
If it turns out that there's a net heat transfer, it would be from higher heat content to the lower one, i. e. from water to ice.No!
Heat transfers from a hotter body to a colder one, nothing to do with the heat content of either.
If it were otherwise, you would boil every time you (70 kg of water at 37°C) swam in the sea (bazillions of tonnes at 17°C)!
Post by: hamdani yusuf on 22/09/2022 22:51:13
One factor might be in equilibrium, while other factors may shift it. Are these columns in equilibrium?All analogies break down at some points, where they don't resemble the things that they are supposed to represent anymore. Your batteries don't experience phase changes. Hence they are more like two ice blocks with the same temperature.I haven't specified the nature of either battery, only its potential. One could be a capacitor with a solid dielectric, the other a wet chemical cell with the same terminal voltage. That's all we know or need to know about the temperature of a body: its ability to transfer energy to or from another body. Equilibrium is equilibrium!
(https://www.designingbuildings.co.uk/w/images/6/69/Capillary_action.jpg)
Post by: Bored chemist on 22/09/2022 22:54:33
Water has higher heat content than ice, when they are at the same temperature and pressure.Not really.
The heat- in particular, the free energy- of the two can be calculated at any temperature you like.
And the important bit is that the freezing/ melting point is the temperature at which the two phases have the same free energy.
They have the same "heat content". That's exactly why heat isn't transferred from one to the other (And, that also is the definition of being at thermal equilibrium).
(The problem is your idea of a "heat content" isn't well defined: but the Gibbs free energies is well defined)
Post by: hamdani yusuf on 22/09/2022 23:11:46
How would a light beam know where to bend when going through an interface between two media? How would it know that it will get the fastest route? There's a problem with anthropomorphising physical phenomena.It would stop when both containers have the same ratio of water and ice.How would they "know" when the ratio was 50%50?
The simple answer is that they can't.
And that's why we know that there's no heat transfer.
But we have been telling you that for 9 pages now, so I doubt you will accept it.
We are comparing two ratios, and they don't have to be 50/50.
Post by: alancalverd on 23/09/2022 08:30:40
Are these columns in equilibrium?Obviously not. But if they were all the same height (and the bath at the bottom was enclosed, of course), they would be in equilibrium but with different total potential energies.
Post by: Bored chemist on 23/09/2022 08:35:51
How would a light beam know where to bend when going through an interface between two media?Because the light beam is in the right place to be affected by the change in medium.
they don't have to be 50/50.Did you deliberately miss the point?
How does the system know what the ratios are?
There's a problem with anthropomorphising physical phenomena.And the problem is that you don't understand it.
But, if it makes you heel happier.
What is the mechanism by which the system is driven towards the same ratio?
What parameter does the ratio affect?
Post by: Bored chemist on 23/09/2022 08:37:23
Are these columns in equilibrium?Possibly, but if they are meant to demonstrate capillary action, they aren't well drawn. The meniscuses are missing.
Post by: hamdani yusuf on 24/09/2022 13:18:28
Did you deliberately miss the point?Some of us prefer to be pedantic.
How does the system know what the ratios are?
Just like diffusion.
Post by: Bored chemist on 24/09/2022 13:21:33
Some of us prefer to be pedantic.Fine,
Answer the pedantic version of the same question.
What is the mechanism by which the system is driven towards the same ratio?
What parameter does the ratio affect?
Post by: hamdani yusuf on 24/09/2022 13:28:57
And the problem is that you don't understand it.I was pointing out your problem, not mine.
But, if it makes you heel happier.
Post by: Bored chemist on 24/09/2022 13:30:19
Some of us prefer to be pedantic.Fine,
Answer the pedantic version of the same question.What is the mechanism by which the system is driven towards the same ratio?
What parameter does the ratio affect?
Post by: hamdani yusuf on 24/09/2022 13:35:00
What is the mechanism by which the system is driven towards the same ratio?With different ratio between two sides of the system, heat transfer rate in one direction will be higher than its opposite direction. When the ratio is equal, the transfer rate would be the same.
What parameter does the ratio affect?
Post by: Bored chemist on 24/09/2022 13:49:26
heat transfer rate in one direction will be higher than its opposite direction.Why?
There's no driving force for the transfer.
Post by: Bored chemist on 24/09/2022 14:01:20
If I fill a U shaped tube with water, and I pour more water into one side than the other, the water will flow to equalise the heights.
You can call the parameter "height" or "potential energy" or "pressure", but there is something which starts off unequal, and ends up equal.
Similarly, if I put a hot stone into cold water, heat is transferred from the stone to the water.
The parameter that starts off different but ends up equal is temperature.
If I put iron into copper sulphate solution the iron dissolves and copper is precipitated.
In this case you can consider the parameter to be "electrode potential" or , more formally a parameter called "chemical potential".
In almost every case where something changes, there's a reason for that change, and it stops when some parameter that was previously unequal becomes equal.
So, for about the 4th time, what do you think it is in the case of the ice and water?
Post by: hamdani yusuf on 24/09/2022 14:43:13
Heat transfers from a hotter body to a colder one, nothing to do with the heat content of either.If you want to be pedantic, it's heat content per unit mass. Perhaps it can be called specific heat content.
If it were otherwise, you would boil every time you (70 kg of water at 37°C) swam in the sea (bazillions of tonnes at 17°C)!
Post by: alancalverd on 24/09/2022 17:20:27
Post by: hamdani yusuf on 28/09/2022 13:54:01
Equally irrelevant. Heat flows from a higher temperature body to a lower temperature body, regardless of their specific heat capacity or heat content per unit mass.It's relevant to answer your previous comment.
Heat transfers from a hotter body to a colder one, nothing to do with the heat content of either.
If it were otherwise, you would boil every time you (70 kg of water at 37°C) swam in the sea (bazillions of tonnes at 17°C)!
Post by: hamdani yusuf on 28/09/2022 13:58:04
So, for about the 4th time, what do you think it is in the case of the ice and water?Entropy.
Post by: Origin on 28/09/2022 14:15:30
Post by: Bored chemist on 28/09/2022 21:35:23
That can't be right; it's an intensive property.So, for about the 4th time, what do you think it is in the case of the ice and water?Entropy.
https://en.wikipedia.org/wiki/Intensive_and_extensive_properties
(This is, of course, another example of why I think it would be better if you learned physics rather than posting 23 pages of misunderstandings.)
But, I'm curious.
How did you imagine it would work?
How would "entropy" be the property that went up + down in the right way?
Post by: hamdani yusuf on 29/09/2022 11:52:15
That can't be right; it's an intensive property.Entropy is an extensive property.
But we can get a closely related intrinsic property by simply divide it by the object's mass. Let's just call it specific entropy, analogous to specific weight, specific heat capacity, etc.
Post by: hamdani yusuf on 29/09/2022 12:00:32
But, I'm curious.A system of a container with ice on one side and water on the other side has low entropy.
How did you imagine it would work?
How would "entropy" be the property that went up + down in the right way?
A system of a container with equal mixture of ice and water on both sides has higher entropy.
Here's an analogy. Take a container with two compartments. Left compartment is filled with sugar solution. Right compartment is filled with salt solution. Take out the separator. Diffusion will make both compartments eventually reach equilibrium with equal ratio of salt and sugar. Each sugar molecule doesn't have to know where the other sugar and salt molecules are at any time. The equilibrium will be reached naturally by statistical mean.
Post by: hamdani yusuf on 29/09/2022 12:14:32
23 pages and you still can't understand that heat flow is from hot to cold. Keep trying and maybe you can get it!Take 1 kg of Plutonium. Take 1 kg of iron with the same initial temperature. Put them in an insulated compartment. Will there be a heat flow?
How do you think Parker space probe survive in sun's corona?
You think that you already understand something by simply ignoring cases where you are not familiar with.
Post by: Origin on 29/09/2022 13:52:28
Take 1 kg of Plutonium. Take 1 kg of iron with the same initial temperature. Put them in an insulated compartment. Will there be a heat flow?The Plutonium will increase in temperature due to spontaneous decay and the heat will flow from the higher temperature Plutonium to the lower temperature iron, just like you have been told innumerable times.
How do you think Parker space probe survive in sun's corona?Insulation.
You think that you already understand something by simply ignoring cases where you are not familiar with.I'm not ignoring anything, the physics is actually quite easy on this subject and the answer to the OP remains the same as was stated on the first page, "there is no net heat exchange".
Post by: hamdani yusuf on 29/09/2022 15:12:43
Insulation.Has it reached thermal equilibrium with its surrounding?
Post by: hamdani yusuf on 29/09/2022 15:23:38
The Plutonium will increase in temperature due to spontaneous decay and the heat will flow from the higher temperature Plutonium to the lower temperature iron, just like you have been told innumerable times.You are ignoring heat generated in iron due to impact by emission from radioactive decay.
Post by: Bored chemist on 29/09/2022 19:47:15
Not yet, which makes it irrelevant.Insulation.Has it reached thermal equilibrium with its surrounding?
Why did you mention it?
Post by: Origin on 30/09/2022 00:10:12
You are ignoring heat generated in iron due to impact by emission from radioactive decay.Yes I am, it's irrelevant.
Post by: hamdani yusuf on 30/09/2022 06:00:52
If it's not in equilibrium yet, then its temperature will increase. Will it have the same temperature as its environment?Not yet, which makes it irrelevant.Insulation.Has it reached thermal equilibrium with its surrounding?
Why did you mention it?
Post by: hamdani yusuf on 01/10/2022 03:47:15
My point is, that temperature difference is not the only parameter that can drive material/energy transfer. Some other parameters are known to cause material/energy transfer, such as differences in electromagnetic potential, gravitational potential, and entropy.You are ignoring heat generated in iron due to impact by emission from radioactive decay.Yes I am, it's irrelevant.
But, I'm curious.A system of a container with ice on one side and water on the other side has low entropy.
How did you imagine it would work?
How would "entropy" be the property that went up + down in the right way?
A system of a container with equal mixture of ice and water on both sides has higher entropy.
Here's an analogy. Take a container with two compartments. Left compartment is filled with sugar solution. Right compartment is filled with salt solution. Take out the separator. Diffusion will make both compartments eventually reach equilibrium with equal ratio of salt and sugar. Each sugar molecule doesn't have to know where the other sugar and salt molecules are at any time. The equilibrium will be reached naturally by statistical mean.
Post by: Origin on 01/10/2022 05:39:39
My point is, that temperature difference is not the only parameter that can drive material/energy transfer.Why are you trying to change the subject, you know we are talking about heat not "material/energy transfer". The one constant in your posts is that when you ask a question you will never accept the answer. I really don't 'get you'.
Post by: hamdani yusuf on 01/10/2022 09:01:45
I'm not trying to change the subject. I'm trying to remind you that there are other factors besides temperature that can affect experimental results which haven't been ruled out properly/convincingly.My point is, that temperature difference is not the only parameter that can drive material/energy transfer.Why are you trying to change the subject, you know we are talking about heat not "material/energy transfer". The one constant in your posts is that when you ask a question you will never accept the answer. I really don't 'get you'.
It is a common practice in science where one answer generates more questions. Get use to it. Even more questions will come up when the offered answer is not convincing.
Heat is one form of energy. In another thread I discussed what distinguish heat from other forms of energy.
It was also mentioned previously that heat is not identical with temperature. Heat can be added to an object without changing its temperature. Temperature can be seen as a manifestation of heat.
Post by: alancalverd on 01/10/2022 09:23:26
I'm trying to remind you that there are other factors besides temperature that can affect experimental results which haven't been ruled out properly/convincingly.They generally have been by those people who do proper, convincing experiments.
Once more: heat experiments are very difficult.
Post by: Origin on 01/10/2022 23:56:41
I'm not trying to change the subject. I'm trying to remind you that there are other factors besides temperature that can affect experimental results which haven't been ruled out properly/convincingly.Yes, I have run many experiments, hopefully we all know that other factors must be taken into account.
It is a common practice in science where one answer generates more questions. Get use to it. Even more questions will come up when the offered answer is not convincing.Unfortunately your questions are largely irrelevant and just confuse you. Your questions and doubts seem to paralyze you and you can't seem to move forward.
Heat is one form of energy. In another thread I discussed what distinguish heat from other forms of energy.That's great but in this thread the question is, "Is there a net heat exchange between ice and water at 0C?" The answer is no, but for some reason you can't see that so it seems kind of important to stay on track.
It was also mentioned previously that heat is not identical with temperature.I would certainly hope that someone with a basic knowledge of physics would know that heat and temperature are two completely different things.
Heat can be added to an object without changing its temperature.True.
Temperature can be seen as a manifestation of heat.Heat can change the temperature of matter if that's what you mean.
Post by: hamdani yusuf on 02/10/2022 12:05:54
Heat can change the temperature of matter if that's what you mean.Heat can also change the phase of matter without changing its temperature.