901
Physics, Astronomy & Cosmology / Re: Is there a net heat exchange between water and ice at 0 degree C?
« on: 24/03/2022 15:02:06 »
Hi.
Where the container expands, work is done on the container by the system (the water region with net freezing). The container walls acquire the energy, the system loses that energy. If it's a container of the type you described (something elastic that tried to maintain a a constant pressure) most of the energy in the walls would be described as elastic potential. In the more general case, we don't care what form the energy takes in the walls of the container, it's usually heat which ultimately escapes to the outer environment. All that's important is that the system has done work on something external to the system.
Opposite idea where the container walls contract. Here the external environment has done work on the system.
This is some work done on the environment by the system (or vice versa if its negative). There can also be other ways of doing work on the system but that doesn't matter. Changing the volume of the container while the system was exerting pressure on the container walls is certainly one way. It doesn't matter if the system is a mixture of solid and liquid, an ideal gas, or something else.
Best Wishes.
ES Said: Sadly, that's still a minor issue. Work, pΔV, is done on a system if there's a volume change ΔV while maintaining constant pressure p.
Hamdani replied: Where does the energy come from, and where does it go to?
Where the container expands, work is done on the container by the system (the water region with net freezing). The container walls acquire the energy, the system loses that energy. If it's a container of the type you described (something elastic that tried to maintain a a constant pressure) most of the energy in the walls would be described as elastic potential. In the more general case, we don't care what form the energy takes in the walls of the container, it's usually heat which ultimately escapes to the outer environment. All that's important is that the system has done work on something external to the system.
Opposite idea where the container walls contract. Here the external environment has done work on the system.
Here is what I found in Wikipedia. Does it also apply to liquid and solid?Yes, for a container that maintains constant pressure, which is what you suggested, that integral reduces to p.ΔV.
Quote
(Integral formula appeared here)
This is some work done on the environment by the system (or vice versa if its negative). There can also be other ways of doing work on the system but that doesn't matter. Changing the volume of the container while the system was exerting pressure on the container walls is certainly one way. It doesn't matter if the system is a mixture of solid and liquid, an ideal gas, or something else.
Best Wishes.
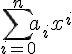 because you don't have to keep writing the constant term a0 separately from the sigma notation when x could be 0. (But that's just a convention for ease of notation).
because you don't have to keep writing the constant term a0 separately from the sigma notation when x could be 0. (But that's just a convention for ease of notation).