Blood Under a Microscope
This week, we’re taking a look at blood, from regenerating blood vessels, to one of our team going to their first blood donation! Plus in the news, concerns that Ebola might be back with a vengeance in future: 60% more often and with 4 times the impact; plus NASA’s first all-female spacewalk: we hear from one of the astronauts involved...
In this episode
00:55 - Climate change may worsen Ebola
Climate change may worsen Ebola
David Redding, University College London
Ebola outbreaks could become 60% more frequent and affect 4 times as many people as they have in recent years, owing to the effects of climate change and over-population. That’s the sombre message in Nature Communications, emerging from a new disease outbreak simulation, that Chris Smith heard about from UCL’s David Redding…
David - Ebola is a very interesting disease. It's what we call a zoonotic or animal-borne disease, so we catch this disease from contacting animals and then it spreads through human populations. What we were really interested in: can we think about the animals that have this disease where they live, and also where the people live and what conditions they live in? And if we can bring those two things together we can understand how Ebola outbreaks are likely to happen in the immediate future and in the more distant future. So what we did is that we constructed what we call a model; that is, do a very simple version of reality that hopefully still contains the important transmission processes for this disease. And then we can run that model lots and lots and lots of times to see where might be at risk, and what might happen if outbreaks happen in certain areas.
Chris - Essentially a computer simulation, then, where you can tweak numerous variables to ask what factors might be big drivers in the future?
David - Yes. So we try and build a simulation that, when we run it for present day, it matches what we do know about where outbreaks have occurred. And when we're happy this simulation is simulating reasonable scenarios, we can then think about what happens if we move this model to different environments. So for instance, where we've changed the landscape to more human-dominated landscapes. And as the climate continues to increase in warmth, how is this then going to play out in those conditions?
Chris - And when you run these scenarios, what do they predict?
David - One of the things that we predict is fewer smaller outbreaks, and they're shifting towards larger outbreaks. And that's likely to be to do with how the disease is then caught by people; there's more people, high connectivity because more people are moving, and therefore we get these bigger outbreaks.
Chris - How big is the difference that you're seeing between what we see today and the predictions made by your study, in terms of the upscale of the size of the caseload and the frequency of these outbreaks?
David - We looked at several different scenarios. In the worst-case scenarios we see a 50 to 60 percent increase in epidemics, which is what we define as outbreaks with more than 1,500 human cases. And we see about four times increase in the total number of cases per year on average.
Chris - And what about the geography over which they are manifest? Do you see an expansion of that? And if so by how much?
David - Under the best-case scenarios we see an increase of just 1 percent, so effectively the same area. Under the worst-case scenarios we're looking at a 15 or so percent increase in the area that's likely to be infected.
Chris - And what factors in the model wield the most influence? So in other words, if you're a policymaker and you're saying, “I'm going to go to Africa and I'm going to have a strategy to try to minimise the Ebola threat,” what's the top three things to target which would have the most dramatic effect turning your worst-case scenario into your best-case?
David - In our model we see the biggest effect in terms of socioeconomic scenario. We have three different scenarios: we have one where we're kind of carrying on as usual; we have one where we work cooperatively to reduce poverty, and increase education, and increase healthcare resources; and one where we start being more isolationist and trying to deal with problems on our own. We definitely see, in that more isolationist scenario, we see a large jump in the number of epidemics and the size of epidemics. That has the biggest influence in terms of our results. And so if we work together when we say, “okay let's improve the healthcare resources in these areas where we have these dangerous diseases that have the potential to spread,” not only are we going to protect from Ebola if we have better containment facilities, we're less likely to see any kind of global importations of the disease into other countries, and we're also going to protect in terms of future diseases that we don't currently know about. It's a win-win situation really if we work to improve healthcare facilities in these high-risk hotspots.

The tiniest guitar ever
Edward Laird, Lancaster University
As we’re all aware, science rocks! But sometimes, science rocks a little smaller than normal, Adam Murphy found out about a rocking study in Nature Physics, from the Lancaster University's Edward Laird…
Adam - We all love a good guitar riff. Slash, Keith Richards, Rory Gallagher, Pete Townsend; everyone's got a favourite. I'm partial to a bit of Brian May myself. But it seems even quantum physics is getting in on the guitar. I spoke to Edward Laird from Lancaster University about the tiniest guitar that's a little bit different.
Edward - Yes. It is a single-stringed instrument and the string is a carbon nanotube, which is the smallest wire that we can make. It's about 100,000 times thinner than an actual guitar string and it's also much shorter, it's less than one millionth of a metre long. It is like a guitar string because it's suspended between two clamps, one at each end, and we pass an electrical current through it and then we measure the electrical resistance. When it moves that resistance changes. Like a guitar string it vibrates at one particular frequency; because it's so small and so light that frequency is correspondingly much higher than an actual guitar, so it's 231 million hertz, which is a high A. What is new about this particular device is that it oscillates by itself.
Adam - A guitar that plays itself, playing the highest of As. Rock stars around the world would be envious. But why do you want to do that?
Edward - Well there’s two interesting things. The first one is, I would say, the pure scientific motivation of studying the effects of quantum mechanics and of quantum measurements on small moving objects. The theory of quantum mechanics was developed 100 years ago to describe the behaviour of individual particles. In everyday life we don't see any of those things. So what happens in between? And we're now in a position that we can explore this experimentally by taking medium-sized objects - so medium-sized in this case means a million atoms or so, which is about the size of one nanotube - and trying to find out how quantum mechanics makes them behave differently. There is also an application as well which is that you can use small resonators for detecting small forces. Just like in a real guitar string, if you put your finger on it, even if you touch it very lightly, it's very easy to hear the change in frequency or the change in damping that changes the note. If you have our nano guitar it can measure a small force applied to that, and we can detect that by a change in the motion.
Adam - Which means by dragging it across the surface you could get a super powered microscope, or a weighing scale that can weigh atoms. But how on earth can it play itself?
Edward - Ah, okay. So what do you need to set up an oscillation is some kind of feedback effect between the motion and the electrical current.
Adam - Feedback, at least in sound, is where noise from the speaker goes through the mic, which goes through the speaker, which goes through the mic...
Adam - Real guitar players, though - some of them like it. So Jimi Hendrix uses feedback in some of his recordings: he points his guitar at a microphone, and if he sets the parameters properly then the feedback stimulates the guitar string, which stimulates the microphone, which then stimulates the guitar string again and so you amplify the oscillation. What we did was to have a feedback… we set up the feedback loop entirely within the carbon nanotube itself. There is then feedback between the motion and the electrical current that’s flowing through it. And the reason for that feedback is in the details of the way the electrons quantum tunnel down the length of the nanotube. But the effect is exactly the same. The feedback makes the guitar string hum to itself and that's why we call it the self-playing guitar.

First all-female spacewalk
Jessica Meir, NASA
Last week NASA made history with its first all-female space walk; two astronauts dropped feet-first out of the airlock to spend the day mending a failed power unit on the outside of the International Space Station. One of those astronauts was physiologist Jessica Meir. Chris Smith chatted with her last month when she was still at Star City, the cosmonaut training facility outside Moscow, just before she blasted off, destined for six months in space aboard the ISS…
Jessica - On the space station we are participating in a wide variety of experiments. So everything from how microgravity and the spaceflight environment affect the human body. There are some specific hot issues that we're looking at right now in terms of the health of astronauts’ eyes. We're seeing some vision changes in astronauts that are coming back that we need to make sure we have a good understanding for, when we start thinking about the future of space exploration. We have some evidence now that the arteries of astronauts are actually thickening in spaceflight. Even in a six month mission, we have about the equivalent of 20 to 30 years of aging here on the ground. So pretty significant increase and we need to understand more about that mechanism as well.
Chris - So what you're saying is you're going to come back looking 30 years older with the arterial tree of someone in their 70s, possibly in need of new eyes. But other than that, it’s going to be fine.
Jessica - Hopefully it won't be quite that extreme, but of course the benefit of researching these things is to make things better for the future, and those are just a few examples of those physiology studies, of course my interest really lies there, but we are doing things like combustion experiments, even flames spread differently in space. As you can imagine, if we can eliminate any of these gravity driven effects that we would have here on Earth, we might expose a whole new world of other factors that might otherwise be masked.
We can grow more pure and bigger protein crystals on the space station. So that has actually led to the development of drugs for things like Duchenne muscular dystrophy, and more recently we are looking at Alzheimer's and Parkinson's. The Japanese space agency even has a drug and development in clinical trials for Duchenne muscular dystrophy based on that space station protein crystal growth research. Some of the other work in the combustion facility will help us hopefully improve fuel economy processes here on Earth and also we'll look toward engines and fuel systems for future spacecraft.
And when we're on the space station we're also doing a lot of routine maintenance and repair. The space station's actually getting a little bit old now, it's about 20 years old. So as you can imagine, if a light bulb needs to be changed or we have to fix the toilet, we can't just call a plumber or an electrician. We have to do all that ourselves. So it's part of the routine operations that we're doing up there. We go for spacewalks as well. Anytime we need to upgrade a system, or if we need to conduct some kind of unanticipated repair that has to be done on the outside of the space station, we put the spacesuit on and have to go work out there for the day. So one of the things that I really like about the job as an astronaut is that it is very active and very diverse, doing something different every day.
Chris - Was this always an aspiration of yours? I'm very good at doing this on Earth but actually there are some questions I can answer even better in space.
Jessica - So I just applied to become an astronaut since it was a dream of mine, since I was five years old. And luckily I think some of my experiences working with the physiology of organisms and extreme environments, helped them see that perhaps my backgrounds would be pretty useful in these space environments as well.
Chris - And how’s your Russian? Because that was the thing that many people are quite surprised to learn that you have to become extremely good at Russian because, isn't a lot of the control materials all written in Russian.
Jessica - Yes. The International Space Station is not just American NASA astronauts. We are up there with the Russian cosmonauts, and then also the European, Japanese, and Canadian astronauts. Those are all the international partners for the space station program. There are two official languages on the space station and those are English and Russian. So everybody up there has to be competent in both languages. We like to describe it as a little bit of Ruglish that really gets used most of the time. For over the past year and a half I've been over here, off and on, Star City, the cosmonaut training center outside of Moscow, and I've been learning to be the co-pilot of the Soyuz spacecraft. That's what I'll be launching in, and all of that training is actually in Russian. Pretty interesting to learn how to not only be a co-pilot given my background, certainly not in that field, but to learn how to be a co-pilot in Russian. It has been an absolutely incredible experience.
Chris - Would you like to sign off for us now with a little bit of Russian, just to prove that you've really mastered it?
Jessica - Sure. I might need a second though, of thinking about what would be a good way.
Chris - Can you say live long and prosper?
Jessica - Not without looking that one up. I can say “Schastliv i do Stantsii”. So I basically said be happy and until the space station

16:15 - Menu labels impact calorie content
Menu labels impact calorie content
Dolly Theis, University of Cambridge
Obesity is becoming more and more of an issue, and with that, there’s been a greater push towards healthy eating. More and more restaurants these days are putting nutritional information in plain sight for us all to see. But does that make any difference? A new study in PLOS One suggests that it does. Chris Smith and Adam Murphy were joined by Cambridge University Researcher Dolly Theis, who’s been looking into this...
Dolly - We have found something different to what the evidence base has looked at so far. So a lot of the evidence is focused on whether it impacts individual choice, whether having calories on the labels changes what people choose when they eat out or order in. And what we did instead was compare the restaurants in the top 100 by sales UK chain restaurants. We compared the ones that had nutritional information on their websites versus those that had it in store and found that those that had in store had 45 percent less fat and 60 percent less salt in the food and drink they serve. So this is new evidence for the U.K. they've conducted it in the States and it indicates that menu labelling potentially incentivises restaurants to improve the healthfullness of their food. So we don't even have to make the choice.
Adam - Now that's a massive difference especially in the salt content. Why do you think it's such a stark change from one to the other?
Dolly - We aren’t able to infer, because it's a cross sectional study, which way the association goes so it could be that the restaurants that already serve healthier foods were more inclined to voluntarily introduce menu labelling in store, but the indication shows that if we are able to see what impact it might have over time it might be that other restaurants that aren't even aware of the nutritional information of their own food and drink, that given that information they can know whether where the improvements are required.
So that is the big message from our paper that it's quite bonkers to think how many restaurants don't even know what's in their own food and drink, let alone the consumers.
Chris - How were they making the patrons aware of those food constituents Dolly? Was this a traffic light system like you see on sandwiches these days or was it just the raw numbers?
Dolly - Absolutely depended restaurant to restaurant. So it was everything from having it right by the pictures, they've got the calorie information alone, and then some only had it on the menus that they give out and others have it separately.
So it's a complete mixture which also again points to the fact that the inconsistency isn't always easy for consumers to know where to even look for it in the first place. So we don't know whether you were introduced, you would have something of a more consistent process and then you can start talking about whether you introduce traffic light labelling on top of it.
Adam - Now I don't know if anyone else has the same kind of morbid curiosity as I do but we were talking about how good restaurants can be, how bad can it get?
Dolly - Well we found one item that was almost six thousand calories! It’s a burger called the Apocalypse Cow. Fantastic name but not great for health! That had almost 6000 calories.
Chris - Who was selling that?
Dolly - Do I mention names?
Chris - It's just a restaurant chain?
Dolly - Yeah. Just a restaurant chain.
Chris - And what’s in it?
Dolly - I wasn't quite in the kitchen when it was being made. That was actually a restaurant that didn't have it in store, menu labeling in store. So a consumer would technically turn up and I'm sure that they would know it probably doesn't look like the healthiest thing…
Chris - I know that around the time that he died, Elvis is alleged to have been eating roughly the number of calories that would have.
Dolly - On the loo.
Chris - Well admittedly he did die on the loo. But when you realize he was eating roughly the number of calories that would have sustained an African elephant each day I think the intake was in the order of 21,000 calories a day. It's probably not far off a couple of those burgers!
Dolly - Exactly and that's the other thing is that we found so many items! That was an extreme example but there are so many items that are just over an entire adult's recommended daily amount. You know one menu item is just bonkers to think that that is the norm now. So it's not only about individuals being aware of it but it's the restaurants themselves, if they don't even have that information how you're going to target interventions?
Adam - So speaking of interventions, what recommendations do you have now going forward?
Dolly - So the government conducted a consultation at the end of last year on menu labeling and they had actually already committed to introducing it. So the consultation was much more about the detail of how they would introduce it.
And we are still, coming up to a year, waiting for a decision to be made. So I think first thing would be great to hear a decision and to understand what the government's thoughts are on this issue and perhaps a good place to start is exactly where we looked with the top 100 chain companies. Because there have been some issues around the capacity smaller companies have to introduce menu labeling and the cost on that. So yes we would like a response from the government.
Adam - Will making restaurants do this actually do anything to change behaviour of people going into those restaurants?
Dolly - We didn't look at this in our own study, so the evidence is pretty weak on the impact on individual choice, but potentially that might change if it's mandated so we have to wait and see.
A new way of sticking
Orlin Velev, North Carolina State University
A team from North Carolina State University have created a new type of super-sticky particle called a soft dendritic microparticle. They look like hairy microscopic meatballs, and they’re made by whipping a polymer solution at very high speeds; this creates chaotic turbulence that pulls out the fine filaments that form the hairs. And those hairs give the particles their stickiness because they generate attractive forces - called van der Waals attractions - with the surfaces they sit on. Phil Sansom got stuck into the paper, published in Nature Materials, with its author, Orlin Velev...
Orlin - This is a new type of particle, big hairy ball of fibres that will stick very strongly to any surface.
Phil - How strongly?
Orlin - Comparable to the form of double sided Scotch tape.
Phil - That sounds so bizarre! How do you make them.
They're made in the highly turbulent flow. Highly turbulent flow is considered to be very chaotic. We inject into this highly chaotic flow polymer solution so we'll use a very chaotic medium in order to make something which is organized in a way that will be called dendritic or hierarchical, because it has different size scales.
Phil - Okay what is this stuff actually made of?
Orlin - It is made of polymer such as polystyrene or cellulose acetate. You have a polymer which is dissolved in a solvent, so the moment you mix the solution of the polymer with the medium, you have the polymer begin precipitating because the polymer does not like the medium into which its solution is being mixed.
Phil - How do you actually make that turbulent flow? Do you stir it really hard?
Orlin - Stirring it really hard is the way to describe it in simple words! The device that they use is called a colloidal mill. You have a static encasing around a rota that rotates as very high speed. Think of them as having highly serrated surfaces.
So when this rotating cylinder in this case moves very fast is that the liquid is going to be sheered very strongly.
Phil - These particles that you get out. What's so strange about them?
Orlin - So there will be a material which is commonly called nanofibers, similar to the nanofibers that you would find on the legs of gecko lizards. Nanofibers have very interesting properties because they're very fine. They have very strong adhesion to any material because of the so-called Van der Waals forces. You can think of this as natural stickiness of any material on the nanoscale.
The reason gecko legs are sticky is because they have this very fine mat of nanofibres and those nanofibres have the ability to stick to any surface because of the Van der Waals forces.
Phil - So you're saying these particles are sticky just like gecko feet are.
Orlin - Exactly so when you have the particles approach a surface the nanofibers would like to stick to that surface and because you have so many nanofibers sticking together you are going to have a high adhesion force, that is you are going to have high stickiness.
Phil - What kind of things can you use this for?
Orlin - We believe that there will be many potential applications in different technologies. One of the properties that we describe is how easy it is to make gels. So in order to make a gel you put particles in liquid until the particles are sticky. The liquid is going to stop flowing and it’s going to become a gel.
We only need less than 1 percent of our particles in any liquid in order to turn the liquid into a gel, so you can use them in products where do you need the small volume of particles in order to make a large volume of gel. For food, cosmetics or similar type of applications.
If you take those particles and dry them on a surface. If you take a hydrophobic polymer, the surface that you dry them on top is going to become super hydrophobic, water repellent. If you take hydrophilic particles it is going to become superhydrophilic.
You can make a sheet which is going to be super hydrophobic on one side, super hydrophilic on the other side, which can be used in different applications such as biomedical applications for example.
Phil - And presumably also we can make gloves that help us climate walls like Spider-Man!
Orlin - I would not say this is a very feasible application! In our case our particles stick strongly once we dry them on the surface. But then prying them off and then attaching them to the surface again is typically not possible.
Phil - So you'd have to chisel Spider-Man off the wall?
Orlin - If you tried to do that with this type of particles probably.

26:25 - Mailbox: Big fans and big burgers
Mailbox: Big fans and big burgers
We're taking a look at the messages you've been sending in...
Adam - And now it's time for the mailbox which is the part of the show where we look at what you've been sending us in, and we just wanted to say a big thanks to Schuyler Moore who's part of the most impressive of clubs which is people who have listened to all our episodes which must be a thousand plus going back nearly 20 years at this stage!
So thank you very much Schuyler. And just a reminder that if you're interested in any of the stories we've covered all the transcripts and papers can be found on our website.
Chris - Thanks very much to Ed Wilson who's tweeted @nakedscientists and sent us a wonderful picture of an Apocalypse Cow burger. That does look like actually it would sustain me for about a week. Probably about 10 inches tall that burger and that looks like it's one two three four five six seven seven or eight burgers? And that's not including the chips which fill the entire tray that it's on!
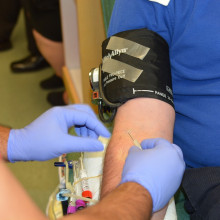
27:59 - The science of giving blood
The science of giving blood
Steven Thomas, NHS Blood and Transplant
Blood is vital: it has a variety of functions, from carrying oxygen around the body in red blood cells, to transporting our immune system’s soldiers, white blood cells and antibodies. It also contains small cell fragments, called platelets, and coagulation factors to help wounds to close. The average person has about 5 litres of blood. But you don’t need all of it. And sometimes other people do, as Adam Murphy found out at his first blood donation, before chatting about his donation with Steven Thomas, from NHS Blood and Transplant...
Adam - I've always wanted to be a blood donor, but until recently I haven't been able to. Now I live in Cambridge, I can. So I took a trip to the donor center there to give my first pint. Once I had been through the consultation process and had my iron levels tested, I sat in the chair with John - who was amazing throughout the whole thing - and started my first donation.
Adam [mid-donation] - And how am I going so far?
John - You’re romping along at 67 mils per minute, and you're just coming up to 300 mil.
Adam - When I had finished my donation - which was pretty painless - I sat down with Steve Thomas, Associate Director for Manufacturing Development at NHS Blood and Transplant, to find out what would happen to my blood next.
Steve - We'll have taken some sample tubes as well as the bag of blood. The tubes will go off to one of our main testing centers where they'll be screened for infectious diseases. The blood itself will go to a manufacturing centre where it'll be separated out into the different components of blood. So most people think of blood as being red cells; so those are the cells that carry oxygen, and what most people think of as a blood transfusion - if someone's losing a lot of blood they'll be given red cells quickly. A fraction of the blood will be separated to be pooled together with three other donations; a platelet fraction to make a platelet concentrate; and possibly the plasma will be separated - so that's the liquid that all the cells are suspended in - that then gets separated and frozen down as a fresh frozen plasma component, and again that's given to people who have deficiencies in their clotting, for instance.
Adam - Right. So that's red blood cells, which carry oxygen around the body; platelets, which help with clotting and forming scabs; and plasma, which is the liquid that the rest of the stuff travels in. Once out of the body and packaged up, the blood goes through a series of tests to check if you're infected with any viruses that might rule you out from donating the blood; how long you might have been infected for; and how your body was coping with the infection. I actually couldn't donate blood until moving to Cambridge from Ireland because I lived in the UK when I was a child. So what's that about?
Steve - So between 1980 and 1996 the food chain in the UK was proven to be contaminated with BSE. And so whilst we had to maintain a blood supply in the UK, and we were collecting blood from donors who lived here in that time as well, other countries excluded people who had lived in the UK in that time just to minimise the risk to their countries’ recipients.
Adam - BSE - or bovine spongiform encephalopathy - might be more known as mad cow disease, and is a disease of the brain that cows get when they eat tainted products. That can be spread to humans where it's known as CJD or Creutzfeldt-Jakob disease, and it is a debilitating and terminal neurological condition.
Steve - It's been very difficult to develop a reliable test for variant CJD. And that's never been successfully brought to market. So there was a lot of work done on that over many years, but nobody ever got to the stage of having a reliable test.
Adam - So overall how is the UK doing when it comes to donation?
Steve - It's quite healthy. We are constantly looking for new donors because there is a turnover of donors. Hospitals are using less blood than before due to many initiatives to reduce unnecessary use of blood. But we still need 5,000 donations every day. We're also very keen to recruit donors from black and minority ethnic backgrounds because there is a high need for donors with those particular blood types, specific blood types, and so that's something that we're very keen to promote.
And we're also very keen to recruit more male donors, so many... there is a skew towards female donors rather than male donors joining the database, and so we're keen to encourage male donors. Primarily because they're more likely to be able to donate due to high haemoglobin levels, for their own safety, but also males are much less likely to have antibodies in their plasma that can cause adverse reactions in recipients. And that's due to females potentially making antibodies to paternal antigens on babies when they've been pregnant.
33:06 - The history of blood
The history of blood
Elise Burton, University of Cambridge
After you go for a blood donation, you get a little letter through the post, telling you, among other things, your blood groups. But what are those, and how did we find out about them? Chris Smith was joined in studio by Elise Burton, an historian with an interest in genetics, at Cambridge University...
Elise - Well, the first blood groups to be defined was the ABO blood group system, which is the most well-known. And it was discovered right around the turn of the century by a scientist working in Vienna named Karl Landsteiner. And what he found at that time was only three blood types, because he was working with only 22 people and his sample didn't include the AB blood type.
Chris - Oh. So how did he actually fathom that there might be differences in blood groups in the first place? Because it's not intuitive to think that, when you see this red stuff coming out of people, that there may be differences.
Elise - That's right. The science he was working with, the context he was working in, was when blood transfusion was not at all a common thing to do and it was not a very successful medical procedure. What he did was try to understand why sometimes, when blood transfusions were attempted, the blood would coagulate or clump together and cause really severe reactions that would make the transfusion fail. So what he did in his first experience was really just take samples from two different people at a time, and mix them together to see if he could find a pattern; you know, some sort of logic for when the blood would coagulate or not.
Chris Obviously this is a bit of a volte-face from earlier medical practices where people were really keen on getting blood out of the body. So when did that switch-around happen, and people realised it's pretty important to keep hold of your blood, and rather than blood-let and take it away, actually we wanna put some back? When did that happen?
Elise - That was really in the 19th century, especially later in the 19th century, when people started having a better idea of how blood pressure worked, and the importance actually of maintaining constant blood pressure; in the event that too much blood was let out, that was recognised as a serious problem. But at the time, in the 19th century, people didn't actually agree necessarily that you should be putting blood back into people. They would actually inject people with things like milk with saline solution, that was a popular one, and worked well for some reasons; and sometimes they would try to inject non-human blood as a replacement.
Chris - I was going to say, did they go down the animal route? Because they'd say, “well this looks the same,” and they had microscopes, they would have been able to see there are cells in there, and they would have looked grossly the same. So it was presumably something that people logically tried, “well let's put some pig blood or sheep blood into a person and see what happens”?
Elise - They did indeed try that out. And it really wasn't until World War One when blood transfusions using human blood became an increasingly routine procedure, and everyone came to an agreement that in fact human blood is the best thing to use in a transfusion.
Chris - And were they adopting those techniques of knowing which bloods were compatible or not at that time, or was it still very much a potluck thing? Where people would say, “this guy's bleeding to death, he needs blood, we’ll just give him some,” and take the first person they come to and just say, “hold your arm out”.
Elise - Well this is an interesting story. So in the lead up to World War One, between 1900 when they were first discovered and 1914, Landsteiner himself recognised that it was probably very important for a transfusion that you should test the donor and the patient and try to come up with compatible matches. But a lot of physicians didn't necessarily agree that that was important, because usually the rare conditions under which they would even consider a transfusion were so dire and so time sensitive that they didn't have time to check for compatible blood type. The test that Landsteiner developed would take an entire day to get results.
Chris - And so what would they do? Just literally couple one person up to another?
Elise - Yes. In fact some of the early techniques for direct blood transfusion, they would literally sew together the artery and the vein of a donor and a patient.
Chris - Goodness. How long did... how did that work out? I would have thought that the donor wouldn't last terribly long either, because you're going to hose out most of your circulating volume!
Elise - Well it was an extremely dangerous procedure, and delicate.
Chris - I bet it was!
Elise - And it required a very well-trained surgeon to even link up the blood vessels in the first place. And yes, it was found that this was not at all sufficient for an emergency medical situation.
Chris - So when did things become more standardised and organised, so that actually we had an idea as to what these blood groups were and what you could and couldn't do in terms of donors and recipients; and also having blood banks, so we store blood, along the lines of common practice today?
Elise - World War One was an incredibly important time for the development of all of these things. So it was in the context of war where you had a lot of guinea pigs, in the form of unfortunately maimed soldiers, where these physicians could do a lot of experimental work. For example, how to keep blood from coagulating through the addition of chemicals like sodium citrate, for example; much, much faster ways to test for blood types so that you could test it within a minute, for example. Those were all things that were developed during the war. And also indirect procedures, where you could withdraw blood using syringes and later have it available for other people to accept as a transfusion. That all happened in the later war period, 1916 to 1918.
Chris - And then what happened since then to bring us to the present era? Because obviously this field has exploded since then and we know the value of blood, but we've learned a lot more about it: we know a lot more about certain diseases that can be inherited that can affect the way the blood works; we know a lot more about certain things that you can be exposed to when you're pregnant, for example, which will affect your likelihood of having problems later.
Elise - Well it was actually in the 1930s and 1940s, with the addition of certain mundane technologies like refrigeration, that we were able to develop institutions like blood banks that would enable you to study blood for longer periods of time without constantly needing fresh infusions from a ready stream of donors. So it was technologies like that in the 30s that were really important. And also the development of an idea of medical genetics which really boomed in the 1940s and after. The blood groups are important to that story because in 1910, that was when it was first found that the ABO blood types are inherited according to a Mendelian pattern. Until then no-one had proved that Mendelian genetics actually apply to any human characteristic.
39:53 - Making new blood vessels
Making new blood vessels
Jo Mountford, Scottish National Transfusion Service
Your body needs more than just the blood flowing around it. It also needs something to flow through: your blood vessels, or your vascular system. And arterial diseases - where the blood vessels become blocked - including coronary artery disease which causes heart attacks, and carotid artery disease, which causes strokes, are the leading causes of death and disability worldwide. One way to tackle these disorders is to try to unclog blocked vessels, but in some cases the disease is too extensive; in these instances, what’s needed are replacement blood vessels. Adam Murphy spoke to Jo Mountford, at the Scottish National Transfusion Service, who is developing a way to re-grow blood vessels from scratch...
Jo - We’re trying to repair blood vessels that are not working properly, or that have been lost through disease or any other disorders. And in order to do that we're, rather than giving drugs to treat the problem, we're trying to replace the cells that have been lost. So in order to do that we have to grow new cells in the laboratory, or eventually in a clean manufacturing facility, and turn those cells into vascular cells or blood vessel cells. And we're doing that by starting with a population called pluripotent stem cells. And these are a particular type of cell that's similar to those found in the embryo, that are able to make any cell in your body at all. And we have to try and coax them into being just vascular cells and nothing else, so that they can then perform the function that we want them to have.
Adam - How do you make it turn into a vascular cell and not say, a brain cell, or a muscle cell?
Jo - Yeah that's the really tricky bit. So that's why we, to begin with, always struggle, and where we have to devise new protocols and new processes for doing this. We take our cues from developmental biology, so we understand how the vascular system and all of the other organs form in an individual during, you know, embryology and as a person grows, and by understanding how that biology works, we then have to try and make that happen in a dish in the laboratory.
So we deliver what are called growth factors. So these are small proteins that deliver signals to the cells, and they specifically tell the cell to, for example, turn into a vascular cell and not a bone. And also we support them with growth media. So this is the liquid that they grow in. It contains proteins, sugars, salts, all of those things. And it effectively is the nutrients, but also it can be selective. So only certain types of cells will grow in certain types of media. So if we use a vascular media those cells will preferentially survive and other cells like bone or brain cells for example will die off.
Adam - And how do you end up with something that is a blood vessel and not just a clump of cells?
Jo - These cells are programmed by nature of the fact that they're endothelial cells, vascular endothelial cells. They're self programmed to know what to do, if you like. So as long as they're in the right environment they will regulate and organise themselves to form tubules. And they'll attract other cells from the periphery to form muscle layers, to form the bigger vessels for example, so you end up with a multilayered blood vessel.
Adam - And when you've got your cells grown, how do you put them into subjects?
Jo - So again, it's pretty simplistic, in our model systems and eventually hopefully in human trials, we're going to look at areas of the body which ischemic, so they have very poor blood supply or compromised circulation. So what we're going to do is directly inject into those areas of poor circulation, and the intent is there, that the cells we put in will either form new vessels themselves, or that they will encourage the cells that are already there and perhaps dormant, or not working properly, to buck up their ideas, if you like, and to regenerate or to restore function themselves. So direct injection and then either they make the vessels or they incorporate into, or encourage vessels that were already there to regrow.
Adam - Are there any particular conditions you're targeting that this could help with?
Jo - Yeah so our first indication is actually one that's really quite common, so it's peripheral arterial disease or critical limb ischemia is the final stage, and this is where particularly in the leg, you get compromised circulation, and the final stage of the actual treatment for it is amputation. So we'd hope that you could treat the disease earlier and avoid those very late stage complications.
Adam - What stage are you at now?
Jo - So that's a really good question, and we're at the stage which is always the difficult one for these type of projects. We have all of the efficacy data that we need, so from animal models for example, so we know that this therapy works in the systems we've looked at it. We're now at the point of needing to go to clinical trial in humans, in order to do that we need funding. And the first step of that is to manufacture the cells in a manufacturing environment that's qualified and regulated to do so, so to produce them at a sufficient grade and clean enough and safe enough. And then we'll have to do safety testing and then we can go to human trials.

Making blood in the lab
Cedric Ghevaert, University of Cambridge
The volume of blood needed every day by the NHS, and around the world, is staggering. But what about growing blood in a lab, could we do that? Chris Smith was joined by Cedric Ghevaert from the department of haematology at the University of Cambridge, to find out more about lab grown blood...
Cedric - So platelets are one of the three main blood components. They’re actually the smallest cell in the body, if you look at a millimetre on a ruler you can line 250 platelets in that millimetre. The interesting thing is that some people argue they're not even a cell, because they don't contain a nucleus, they don't have DNA.
Chris - When I was learning biology at school they said platelets are bits of cells.
Cedric - That's absolutely true. They are fragments of a parent cell that lives in the bone marrow, and called a megakaryocyte, and one megakaryocyte will release about 1000 to 2000 of these little fragments called the platelets.
Chris - Every day?
Cedric - Every day we produce 10 to the 11 platelets
Chris - 100 billion Every day?
Cedric - That’s right.
Chris - And so these cells are just budding off little bits of themselves, which then go circulating around in the bloodstream.
Cedric - That's absolutely right.
Chris - And what are they doing there? What is their role?
Cedric - The main role of the platelets is to monitor your blood vessels, so they contain two things: Their outer layer, which is called the membrane. And on that membrane they have all sorts of little receptors, and these receptors will tell the platelets that the blood vessel has been damaged. When the platelet detects that it does two things, it will attach to the damaged blood vessel, and it will become activated and by that, it will then tell other platelets around, “you need to come and help.” These platelets will then stick to each other and form a plug to literally block the hole.
Chris - Do they change shape or anything? Do they become, sort of spiky or anything, to become more jagged so they jam in the hole.
Cedric - So they do become spiky, indeed you see that under the microscope when you activate the platelets. They go from a disc to this sort of spider, and that allows them to indeed interact with each other even better. The thing that they also do, is to then pull. They literally pull the wound together to try to stem the blood flow.
Chris - And they're presumably the first responders when you have a wound. They're there first because they're at the scene of the crime already because they're in the blood. And then as more come along, they're recruiting more of their mates from the numbers that come in the blood flow, and what do they provide the initial foundation of a blood thrombus or a clot.
Cedric - That's absolutely right. So once they become activated, inside the platelets there are granules, and those granules contain things that tell the blood, “you need to clot.” These things are released when the platelets become activated. And that leads to an amplification of the blood clotting that proteins are linked together, they form a polymer, and that polymer is a sort of a mesh that will capture more platelets and really plug the hole.
Chris - So the platelets are pulling more raw materials that are dissolved in the blood into that wound site, and then turning it into this dense mesh work that's gonna be a stable repair.
Cedric - That's absolutely right.
Chris - So they're really critical aren't they?
Cedric - Absolutely.
Chris - We can't do without them. And what's the problem with just growing them in a dish, because we can grow loads of things in dishes these days. We can you know, cells grow in dishes easily. So why can't you just churn out platelets in a dish?
Cedric - The main challenge with producing platelets in a dish is to do it so efficiently that actually we have a product that can be used, for example by the NHS and cost efficient. So if you look at a bag of platelets which we give for a transfusion, it contains three times ten to the eleven.
Chris - So 300 billion platelets.
Cedric - So where the platelets score as it were, is that we only need to produce one megakaryocyte, to produce a thousand platelets. And we can grow the megakaryocyte from stem cells. So the idea is that we can take stem cells, grow them into a megakaryocyte, and then right at the last stage of production, suddenly you have this massive amplification, a thousand times more platelets than you had megakaryocytes.
Chris - But if it's that easy to just grow these things in a dish, why are we not doing it? What is the what's the problem at the moment.
Cedric - The main problem is that particular last step. When the megakaryocyte is in the bone marrow it gets its cue from its environment. And it will detect the blood flow. It will be talked to by the cells that are around it, and that, it's very difficult to reproduce in the dish. If we produce megakaryocytes in liquid, in a culture dish they can produce 1 to 10 platelets, so we are at least a hundred times below what a megakaryocyte can do.
Chris - So you've got to have some way of recreating that very specialised three dimensional relationship in the bone marrow, where all these cells are in contact in a particularly special arrangement which seems to be the cue to them, to churn out platelets with the efficiency that they do when they're inside the body.
Cedric - And that's exactly the challenge that my group and several other groups across the world are trying to answer. And there are two ways to do this. First we need to tell the megakaryocyte there’s a flow. They sense the flow, and that makes them release the platelets. So we put them in a bioreactor where they're exposed to shear, which is basically fluid going along them.
Chris - That's kind of mimicking the blood flowing through the bone marrow. So that would normally be bending and distorting the cells a bit, I presume, and that's what makes them churn off or snap off bits.
Cedric - That's right. So they produce these long digits which we call proplatelets, and these long digits elongate in the bloodstream and then snap off these platelets.
Chris - And when you make them, having mimicked this as best you can, do the platelets that you produce in the dish look like, and critically work like, the ones that are made naturally in the bone marrow?
Cedric - That's the critical thing that we are trying to address at the moment. They are bigger when we produce them in the dish, and they don't seem to quite react like normal platelets. However that doesn't mean that it won’t work really well. What we need to do is to test them through a range of assays to really make the statement; these platelets are good, they will monitor your blood vessel, they will last in circulation.
Chris - Is your aim to make platelets bespoke for a patient? Or would you make off the shelf platelets, a bit like we'd currently do with transfusion medicine, where we just make a big bag of platelets collected from a range of donors?
Cedric - So at the moment we can produce platelets from either four donations, from four different donors, or we take them off a special machine where we have one pool of platelets coming from one donor, but we've talked the blood group before, one of the challenges with platelets, is that some people are immunised and they need platelets of a very specific blood type.
Chris - When you say immunisation, you mean that they've made an immune reaction to certain types in the past, so you've got to basically restrict what types you give them?
Cedric - Exactly. The beauty of working with stem cells is that we can edit the DNA somewhat, and because we can edit the DNA we can actually make platelets that don't express blood group, that are universal platelets. The one we produce in the dish can go to anyone. And that's one of the beauties of this technology.
Chris - And are you far away?
Cedric - We are not that far away. We are looking at human clinical trials in the next two to three years.
Were dinosaurs edible?
Mariana Marasiou reached out to Steve Brusatte at the University of Edinburgh, to find out...
Dan - I watched a survival show where the host cooked a skunk, cooked it and ate it but said it was awful. It got me wondering, are most animals edible? And taking that to an extreme, what are the odds that dinosaurs were actually edible?
Mariana - Dan is definitely on to something. Most animals are indeed edible but a edability is not the same as being nutritious tasty or culturally accepted. But how about eating dinosaurs? Now that I was very curious to find out and so I reached out to Steve Brusatte a palaeontologist at the University of Edinburgh.
Steve - Dinosaurs definitely were edible, at least to other dinosaurs. Some of the most iconic dinosaurs of all, things like T-Rex and velociraptor, made their living as predators, eating other dinosaurs like the horned dinosaurs Triceratops for instance or long neck dinosaurs like brontosaurus.
And so those prey dinosaurs must have been pretty tasty to the meat eaters.
Mariana - But would they have been tasty to us?
Steve - I would imagine, yeah! Actually they probably would be! In fact they probably would have tasted like chicken. I guess everybody always says that everything tastes like chicken. And I'm being a little bit funny but I'm not really being that facetious here because birds evolved from dinosaurs, which means they are dinosaurs. So a chicken is as much of a dinosaur as Triceratops or Brontosaurus.
Mariana - Wow. But not all birds taste like chicken though.
Steve - Maybe some Mesozoic age dinosaurs may have tasted like duck or Turkey or game birds. Who knows? And as with birds and mammals today, it was probably the plant eating dinosaurs that would have tasted best, they would have been the tastiest, the meat eaters not so much. This is why we eat cows and sheep but we don't eat wolves and bears so much.
Mariana - That may also have something to do with how difficult they are to catch! On the forum Chiral SPO also pointed out that cooking can turn many inedible things into at least somewhat edible foods. And besides that, let's not forget that cooking gets rid of lots of bacteria that might make food unsafe to eat.
Related Content
- Previous What actually is a shooting star?
- Next Could you eat a dinosaur?









Comments
Add a comment