The flu is an infectious disease of which we are all likely to contract at some point in our lives. While some disregard it as just a bad cold, this infection can actually result in 500, 000 deaths every year. Many of the deaths are from high-risk groups: people with a weaker immune system like the elderly, chronically ill or very young [1].
 |
|---|
Influenza viruses that cause flu in humans mostly show up in seasonal epidemics and are more easily spread at lower temperatures, targeting our respiratory tract [2]. There are three types of influenza viruses: A, B and C. While type C viruses are responsible for mild forms of the disease, and are not considered to cause epidemics, types A and B are responsible for the seasonal manifestations almost every year, with type A being the most spread amongst humans [3]. Although these types are all influenza viruses, they are distinguished on the basis of relatively small structural variations that can still account for the difference in their impact. Given their wider distribution and historical impact on the public health, this article will be focused mainly on Influenza A viruses.
As well as seasonal epidemics, influenza can also show up in a pandemic, which is when a new kind of virus becomes able to transmit very efficiently amongst people and eventually spreads across the world. These novel viruses generally emerge from animals, e.g. the influenza A H5N1, which derived from birds and spread worldwide in the early 2000s [4, 5]. When animal-derived viruses emerge in humans, the media talk about this topic very frequently (the reader might have hear of "swine" or "avian" flu), sometimes reporting the potential risks associated with these global epidemics and mentioning the race that the scientific community undertakes in the search of an effective, preventive vaccine.
Later, I'll look at why these animal-derived viruses drive the media into such a storm, and ask if the race to create vaccines is justified, or simply the fruit of pure scaremongering, but first let us start with some information on the biology of influenza viruses.
Influenza Viruses: Structure and Diversity
Viruses need to hijack cells to replicate
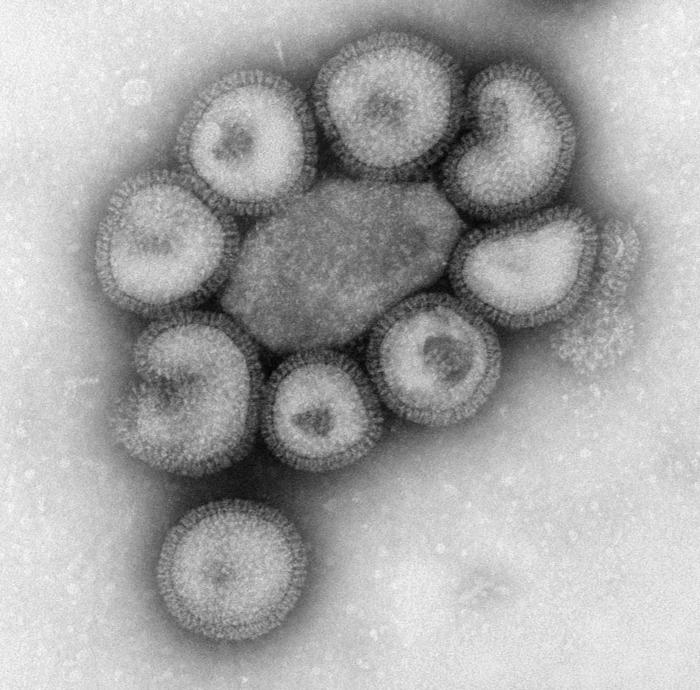
 While the cells that make up you and I are microscopic, autonomous forms of life, generally in the order of micrometers, encapsulated by a membrane and able to create copies of themselves and grow independently, viruses are very different. In general, viruses are hundreds to thousands times smaller than cells, with their dimensions ranging in the order of nanometers. Although these microorganisms come in many shapes and structures, a good share of them are made up by a round protein capsule containing one or more strands of genetic material, either DNA or RNA.
While the cells that make up you and I are microscopic, autonomous forms of life, generally in the order of micrometers, encapsulated by a membrane and able to create copies of themselves and grow independently, viruses are very different. In general, viruses are hundreds to thousands times smaller than cells, with their dimensions ranging in the order of nanometers. Although these microorganisms come in many shapes and structures, a good share of them are made up by a round protein capsule containing one or more strands of genetic material, either DNA or RNA.
Due to their small dimensions viruses have given up some characteristics that could allow them to be fully functional like cells: they cannot create copies of themselves independently, hence their need to look for a host cell to invade. They take advantage of its mechanisms to replicate, and hijack it to create more and more viruses. These then spread, find new cells and restart the cycle.
But how does a virus give indications to a cell on how to assemble new copies of itself? That is through the genetic material and, in the case of influenza virus, RNA.
The RNA that the virus carries is like a book with the instructions on how to build new viral particles (virions). Released from the infected cell, the virions include their own copies of RNA, together with all they need to recognise new target cells to be infected, and restart the cycle.
However, like a book is useless if nobody ever reads it, a virus left alone without a cell to infect will just stay there and do nothing. Inside a cell there are some "carpenters" (enzymes) that can read the instructions and build new viruses. Sometimes the enzymes are so committed to creating new copies of these microorganisms that they forget to look after whatever else is going on in the cell, which will eventually die.
 |
|---|
Influenza virus is one of those pathogens that hijack the cells to the point in which they wear out and die. In contrast some other viruses are not as greedy. For instance, HIV can allow a cell to keep working on its own tasks for a long time (sometimes years) before bursting into replication.
The immune system and influenza viruses
A healthy, young person who gets the flu can generally recover from the infection pretty smoothly in just one or two weeks by staying home and drinking hot tea. That is because there are defined populations of viruses that keep circulating in human communities: once we acquire immunity against a virus (to see how this happens, please refer to my previous article
"how vaccines work"), we will generally be protected against that one. Later, once the virus comes back the following winter with little, natural differences accumulated over time and cycles of transmission (with the same mechanisms that set the basis for evolution of every living being), we will get sick again, and generally -if not belonging to the high-risk categories- just as mildly. However, if for some reasons the virus is too unrecognisable; our immune system will need some time to build an effective plan of defence. This unpreparedness can create a time window in which the virus can potentially lead to a more severe progression of the disease, even in individuals who are not considered at high risk of complications.
In the past, the accumulation of mutations lead to the diversification of the influenza viruses, thanks to that very famous process named evolution. That is very similar to what happened to a common ancestor that differentiated over time in the modern monkeys and us. However, instead of differentiating between monkeys, humans, primates etc., for influenza viruses the distinction is based on the types and subtypes according to the structural difference of some of their components (see below).
Although these differences (mutations) appear randomly, not all of them persist in a population of viruses: that is because of the selective pressure applied by our immune system. Briefly, if some modifications make the virus more likely to remain unnoticed to our defence mechanisms, then its characteristics will be passed along and possibly stabilise in the viral population. In contrast, if during these random modifications the virus accidentally changes a characteristic that is necessary for its survival, thus reducing or eliminating its ability to replicate, then it will be more likely to be selected against and killed, and the mutation will not persist.
The trump card of influenza viruses
In addition to the slow, random mutations accumulated in the race to escape the host's defences, a mechanism shared in some extent by all viruses, some characteristics can suddenly make an influenza virus very diverse from the one that usually circulates in a population: the naturally fragmented genome and the ability of more than a viral particle to infect the same cell.
 |
|---|
Influenza viruses are not like other viruses, many of which have a linear, single genome containing all the assembling instructions one after the other on the same book. Influenza viruses have their genome naturally divided in 8 parts, all included inside the same viral particle. Just imagine that the 8-page instructions for a shelving unit that you need to build were delivered unstapled and without page number. With some dedication, you can still manage to find the correct orders of the paper sheets and build the furniture piece. Similarly, the cell can still build a viral progeny of functional viruses.
However, let us consider the event of superinfection, i.e. a phenomenon that takes place when two parasites get into the same host at the same time.
When infected by two influenza viruses, the cell has to manage as many as 16 pages of instructions, i.e. 16 genomic fragments, to build a progeny of viruses. Because the cell has no capability of defining what fragments came from which of the two viruses, the result will be the production of mixed, "chimeric" viral particles containing some components coming from the one virus, and some from the other, which might be unknown, and therefore catastrophic, to our unprepared immune system.
Classification of influenza viruses
In the modern virology, influenza viruses are classified according to the sequence of two molecules that they expose on the surface: Hemagglutinin (H) and Neuraminidase (N). For Influenza A viruses, 18 types of H and 11 types of N have been described. For instance, the widely spread H3N2 Influenza A virus exposes Hemaglutinin of the 3rd type on its surface, and Neuraminidase of the 2nd. Another virus widely spread among humans is H1N1.
While the function of Hemagglutinin is to recognise and start the phases of entrance into the target cell, Nerauminidase has the opposite role, that is to release the newly formed viral particles from the cells, in the context of a mechanism called "viral budding".
The fact that so many different types of these two molecules exist is a consequence of their position on the viral particle: exposed on the surface, H and N are more easily identified by our immune system. The virus, driven by the mechanism of evolution, will need to change some parts of these molecules so that they can pass unobserved to the host's defences [6].
However, considering that our immune system can get trained in identifying the Hemagglutinin and Neuraminidase circulating in humans and generally keep up with the changes that are accumulated, where do viruses pick up new forms of these molecules during superinfection?
Transmission from Animals [7]
While influenza B viruses almost exclusively infect humans, Influenza A viruses can also be transmitted within and between animal populations. This sometimes results in the transmission from animal to human of viruses with characteristics that were "borrowed" from the animal population and previously unknown to the immune system of the patient. As a result, the immune system of the infected person will need a while to build an effective response against this new virus, creating a window of time that can potentially be harmful for the host. This has some serious implications for the public health, and explains why influenza B viruses, generally unable to take advantage of this animal-to-human transmission, are not prone to cause pandemics [17].
The most diverse pool of influenza A viruses is not found in humans, but in avian species. While mostly H1N1 and H3N2 are spread among people, which means that the population of viruses infecting humans usually displays only two main kinds of H and N, as many as 16 H and 9 N subtypes circulate among wild waterfowl and shorebirds. This makes it possible to hypothesise the existence of up to 144 combinations of H and N, of which 112 have already been identified! This enormous diversity is also encouraged by the migratory patterns of the birds, which meet in common breeding grounds or stopover points thus "sharing" with other birds the subtypes they are infected with.
The avian virus can then be transmitted to domestic birds (e.g. chickens, turkeys, ducks...), with the subtypes H5 and H7 being the most spread in poultry. Eventually, humans can contract the virus either from wild, or domestic birds.
However, birds are not the only animal reservoir of influenza viruses: many of them are in fact spread among swine populations too. Because of the presence of some target molecules in their respiratory tract, swines are very susceptible to infection with viruses that are adapted to humans or birds.
Simply put, both you and a chicken can pass flu to a pig!
If it actually happens that a bird and human influenza virus simultaneously infect the same animal, then some combinations of the two can emerge. This is why pigs are sometimes labelled as the "mixing vessel" of novel influenza viruses.
An exemplar case of animal-to-human transmission is that of the year 1997, when the H5N1 "jumped" from poultry populations to humans in China, in a particular combination of genomic segments that allowed it to gain the capability to infect people. In the early 2000s the virus had spread from Asia to Africa and Europe, resulting in millions of infections in poultry, as well as hundreds of reported human cases, with many casualties [5].
Similarly, in the spring of 2009, a swine-origin virus spread to humans causing the first influenza pandemic recorded since 1968. In this virus, some segments were derived from an Eurasian avian-like swine lineage (among them the one that dictates what N to expose on the surface) and some other segments from a purely swine H1 virus subtype. This pandemic resulted, according to the U.S. Center for Disease Control and Prevention, in a range of deaths spanning from 151,700 to 575,400 [8].
However, if the number of casualties was this close the range of fatalities of the seasonal flu, why was the virus so dreaded, and the scientific community on the lookout?
Learning from the Past: the Race to Vaccines
Although the 2009 pandemic had a tragic death toll of about half a million victims, there is a particular time in history when things got particularly serious [9]. The influenza pandemic of 1918 and 1919, "Spanish Influenza" killed 40 million people worldwide, and claimed more victims than World War I itself. Outnumbering in one year the casualties that the Black Death Bubonic Plague provoked from 1347 to 1351, the Spanish Flu is now one of the deadliest epidemics ever recorded [10; 11]. It is now accepted that the 1918 disease was caused by a virus source with avian features, which had not been circulating in humans in the decades before the epidemic [12].
Many cases reported people dying within hours of first feeling ill, with the mortality rate highest among adults between 20 and 40 years of age: in contrast to what generally observed with seasonal influenza, the young adults were particularly vulnerable to the disease. This was due to their immune system, stronger than that of the common categories at risk, which provoked reactions too violent for the young adults to bear [13]. The name "Spanish Influenza" is due to the particularly deadly reports that we have from this country as a testimony. However, considering the neutrality of Spain during World War I, it is likely that it was rather the military censorship of other countries involved in the conflict to limit the transmission of accurate information on the counts of their casualties.
The fist cases appeared in Kansas by the early spring of 1918. Weeks later, many events started being reported in Europe as well, although -please note- there was absolutely no signal that this influenza epidemic could be more dangerous than those of the usual, circulating varieties.
In the following months, the virus accumulated some mutations and returned from September to November 1918 with unpredictably deadly features. From that moment, influenza spread extremely rapidly in waves of infection that left many fatalities behind.
Early symptoms of the disease included some of the normal signs of influenza infection like fever, sore throat, exhaustion, headache, aching limbs and cough. In addition, some patients could experience vomiting, diarrhoea and violent nosebleed. Most of the patients with these symptoms could make it to a full recovery, while others suffered a relapse. Their temperatures would increase again and they would experience serious respiratory problems, some of which with massive pulmonary haemorrhages that would, eventually, cause the death the patient [14].
In 1918, there was no particular treatment for Influenza, nor a preventive vaccine. In truth, at that time there were neither the knowledge nor the technology to prove the existence of viruses themselves beyond pure conjecture, and it was believed that influenza was caused by a bacterial infection [15]. Still, as vaccines can also be produced against bacterial pathogens, experts started trying to develop preparations against what they thought to be the cause of the pandemic. This inaccuracy did not bode well for their purpose, and all of the vaccine candidates proved ineffective [14].
With the fading hope for a vaccine to successfully prevent infection, practitioners started committing to other ways to treat influenza. Unfortunately, the medicines of that time (e.g. Vicks Vapo-Rub and atropine capsules) did very little in terms of ameliorating the conditions of the infected [14].
Unexpectedly, after the second wave struck in late 1918, the number of casualties plunged abruptly, and the pandemic was considered ended by summer 1919.
The pandemic influenza viruses that followed in 1957, 1968 and 2009 descended, via different pathways of transmission, from that influenza virus that caused the pandemic of 1918. The fact that there were many similarities between the Hemagglutinin of the 1918 virus and that of the 2009 pandemic had some practical implications even after that much time: although more than 90 years had gone by since the deadly world epidemic of 1918, the elderly survivors that were exposed to the 1918 virus during their youth were still protected by the infection, thanks to the memory of their immune defenses. It is somewhat fascinating that after all that time, the cells of their immune system were still able to recognise the structural characteristics of the old influenza virus (for more details on the mechanisms of this memory, see "how vaccines work") [16]. That deadly virus, while killing lots of people, provided a tool for protection from future, similar infections in those individuals that were infected, but managed to survive. Taking advantage of the memory of our immune system is at the basis of the vaccination, but thanks to vaccines we now don't need to run the risk of being infected by a circulating, potentially harmful virus to gain immunity. Nowadays, by exposing our organism to a dead, modified or shattered microorganism contained in a vaccine, we can protect ourselves and our children from dangerous, fully infective viruses or bacteria.
Besides these structural characteristics shared between the virus that arose in April 2009 and that of the 1918 pandemic, some other traits were communal between the two: the early behaviour that H1N1 displayed in 2009 (low mortality rates and appearance during springtime) highly resembled the first, more moderate wave of the Spanish flu. A few years ago, when facing the emergence of a new virus, the virologists of 2009 had no way to exclude that the virus would come back mutated in a second, much deadlier wave like in 1918.
At that point, with the possibility to develop a protective vaccine and no way to predict what would be the actual outcome of the epidemic (like Niels Bohr allegedly said, "It is hard to make predictions, especially about the future"), one may understand that the experts preferred to take a lesson from the past and be cautious, encouraging the population to get vaccinated against a virus they could not rule out to return in a pandemic of catastrophic consequences.
Better safe, than sorry!










Comments
Add a comment