Cooling Glow Sticks
Ingredients
At least one Glow Stick
A bowl of ice
Some Salt
Instructions
Go somewhere not very bright
1- Snap the glowstick - Don't shake it yet - look at it closely
2- Now shake it.
3- Add some water to the ice so it comes about half way up the ice
4- Add a few tablespoons of salt to the ice-water mixture
5- Put the glow stick half way into the ice-water
6- Take it out again - What has happened?
7- Try warming the glow stick up
Result
When you add salt to the ice it will get colder and go below zero
When you snap the glowstick you should see little swirls of glowing liquid in the glowstick.
After being shaken the whole glowstick should be glowing
When you cool down half of the glowstick, this piece should become much less bright, but when you warm it up again it will start glowing again.
Explanation
A glowstick is made up of two plastic tubes, the inner one is filled with hydrogen peroxide (this is a bleach that is often used on hair) and in the outer one is a chemical called Cyalume and a dye. If you want a different coloured glowstick you use a different coloured dye.
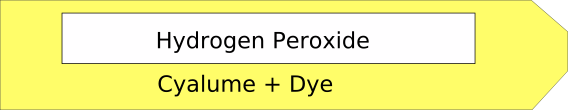
The inner tube is quite brittle and when you bend the outer tube the inner one snaps letting the two liquids mix together

Where they mix the Hydrogen Peroxide reacts with the Cyalume producing an unstable molecule with loads of energy. If this collides with some, dye the molecule breaks down into carbon-dioxide and transfers its energy to the dye, which then releases the energy as light. The more often this happens the brighter the light is.
The beautiful glowing swirls when you cracked the glowstick were where the two liquids were slowly mixing together.
Why does it go dim when it gets cold?
If anything is hot it means that the molecules have lots of energy, which means that they are moving very fast so they hit each other more often and when they do collide there is plenty of energy to allow them to react.
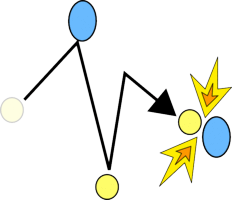
If you cool the molecules down, you are taking their energy away, so they move more slowly, hitting fewer other atoms every second, and when they do collide they often won't have enough energy to react.
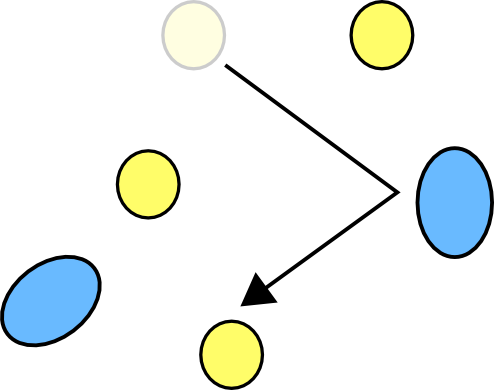
So when the glowstick is cooled down the reaction slows right down making it very dim, but making the reaction last much longer because the chemicals are used up much more slowly. So if you want to make your glowstick last, put it in the freezer.
What has this go to do with snakes?
 Snakes and lizards are cold blooded, which means that they don't control their own body temperature. So when the weather gets cold, they get cold. LIfe is driven by a huge set of complex chemical reactions a bit like in the glowstick, so when a snake gets cold, all of its reactions slow down, which means it can't do anything as fast when it is cold. This is why snakes and lizards can often be found on a sunny rock warming up on a cold morning, speeding up their chemical reactions so they can get on with their day.
Snakes and lizards are cold blooded, which means that they don't control their own body temperature. So when the weather gets cold, they get cold. LIfe is driven by a huge set of complex chemical reactions a bit like in the glowstick, so when a snake gets cold, all of its reactions slow down, which means it can't do anything as fast when it is cold. This is why snakes and lizards can often be found on a sunny rock warming up on a cold morning, speeding up their chemical reactions so they can get on with their day.
- Previous Strange Cornflour Slime
- Next Butter from cream










Comments
Add a comment