For over 70 years scientists have been trying unsuccessfully to produce sperm in a petri dish. Now they've finally succeeded.
Writing in Nature, Takehiko Ogawa and his team at Yokohama City University reasoned that sperm probably depend for their development upon the complex three-dimensional environment provided by multiple nurturing cell types found within the testis. To recapitulate this sperm cell nursery, the team developed a system to culture small pieces of newborn mouse testicular tissue which they suspended in an agarose supporting gel and perfused with culture medium and a limited cocktail of growth factors.
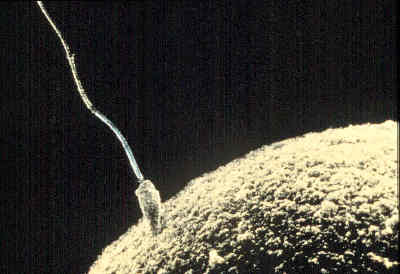 To make the job easier they also used tissue that had been genetically programmed to glow green when mature sperm cells were being produced. Using these tactics, and by carefully manipulating the chemical environment they eventually hit upon just the right conditions to persuade spermatogonial stem cells to turn first into spermatocytes, the first step in sperm maturation, and then to take the crucial step of halving their complement of genetic information and growing tails.
To make the job easier they also used tissue that had been genetically programmed to glow green when mature sperm cells were being produced. Using these tactics, and by carefully manipulating the chemical environment they eventually hit upon just the right conditions to persuade spermatogonial stem cells to turn first into spermatocytes, the first step in sperm maturation, and then to take the crucial step of halving their complement of genetic information and growing tails.
The ultimate proof, though, was when they were able to use these sperm to fertilise mouse eggs and produce viable - and fertile - offspring. This, they say, shows that testicular tissue can be triggered to produce mature, functional and fertile sperm under artificial conditions, which could have huge clinical implications for the treatment of infertility.
In particular, individuals facing chemotherapy for cancer could "bank" a sample of healthy testicular tissue prior to undergoing treatment. Kept in liquid nitrogen in the interim, this material could then be used to rear sperm for the patient at a later date, should the treatment render him infertile.










Comments
Add a comment