"The wheels on the bus go round and round, round and round, round and round. The wheels on the bus go round and round, all day long..."
Do you remember this childhood song? It will stick in your head now -  circulating there for at least a few days. I think of it often - not because I have particularly fond memories of riding the bus to school, although they are not bad memories either - but because this song is one of the best ways to think about the nitrogen cycle. Yes, nitrogen.
circulating there for at least a few days. I think of it often - not because I have particularly fond memories of riding the bus to school, although they are not bad memories either - but because this song is one of the best ways to think about the nitrogen cycle. Yes, nitrogen.
While life as we know it cannot survive without nitrogen, too much nitrogen can lead to deadly consequences in the marine environment. In the next several essays we are going to explore how nitrogen has transformed our coastal oceans. But first we must learn about nitrogen and how it cycles on Earth.
Simultaneously, in 1772, the Scottish physician Daniel Rutherford and a Swedish chemist Carl Wilhelm Scheele, noted that air contained two primary but very different "fluids". The first was oxygen and the second was di-nitrogen, or N2 gas. The scientists learned that organisms (in this case a mouse) as well as fire were extinguished in the presence of N2 and thus, in time, it earned the name "azote", from the Greek for "without life".
Of course, this is a bit ironic as in truth  nitrogen is a fundamental element necessary for all life. It is a critical component of proteins and of DNA and RNA - the blueprints that help define the shapes of our bodies, the colours of our eyes and whether or not our ears attach to our heads. In fact, your body is approximately 3% nitrogen by weight (the rest is predominantly composed of carbon, oxygen, and hydrogen).
nitrogen is a fundamental element necessary for all life. It is a critical component of proteins and of DNA and RNA - the blueprints that help define the shapes of our bodies, the colours of our eyes and whether or not our ears attach to our heads. In fact, your body is approximately 3% nitrogen by weight (the rest is predominantly composed of carbon, oxygen, and hydrogen).
Nitrogen can be found in a variety of forms including the lifeless gas as well as in dissolved and particulate phases. Scientists separate nitrogen into two categories: 1. un-reactive nitrogen or N2 gas; and 2. reactive nitrogen (sometimes called Nr), which includes ammonia (NH3), ammonium (NH4+), nitrate (NO3-), urea and proteins. All of these forms allow nitrogen to cycle continuously through every part of the biosphere, just like the wheels on the bus. And once nitrogen becomes reactive it passes ceaselessly from one form to another, over and over again, round and round.
The largest pool of nitrogen on Earth, and the one that Rutherford and Scheele first discovered, is found in the atmosphere.  In fact, N2 gas accounts for approximately 78% of the air we breathe. But this vast pool of N2 swirling and whirling around us is unusable to most organisms on Earth, apart from nitrogen fixers. Nitrogen fixers are bacteria with the unique ability to take inert N2 gas out of the atmosphere, break apart the two triple bonded nitrogen atoms, and turn them into a new form of nitrogen - ammonia (NH3). You are already familiar with these bacteria if you have munched on a peanut or sneaked a mouthful of peas off the summer-ripened vine. All of these plants are also known as legumes and they have nitrogen-fixing bacteria living on their roots in bumps or nodules. These bacteria help to naturally replenish soil nitrogen taken up by plants when they grow. In fact, since ancient times farmers have planted legumes as a way of "reinvigorating" the soil after growing a crop of plants without this nitrogen-fixing ability - say wheat or maize (corn). Legumes are also protein rich and thus they are important components of our diet.
In fact, N2 gas accounts for approximately 78% of the air we breathe. But this vast pool of N2 swirling and whirling around us is unusable to most organisms on Earth, apart from nitrogen fixers. Nitrogen fixers are bacteria with the unique ability to take inert N2 gas out of the atmosphere, break apart the two triple bonded nitrogen atoms, and turn them into a new form of nitrogen - ammonia (NH3). You are already familiar with these bacteria if you have munched on a peanut or sneaked a mouthful of peas off the summer-ripened vine. All of these plants are also known as legumes and they have nitrogen-fixing bacteria living on their roots in bumps or nodules. These bacteria help to naturally replenish soil nitrogen taken up by plants when they grow. In fact, since ancient times farmers have planted legumes as a way of "reinvigorating" the soil after growing a crop of plants without this nitrogen-fixing ability - say wheat or maize (corn). Legumes are also protein rich and thus they are important components of our diet.
So why does it matter that most nitrogen on Earth is an inert gas? It matters because nitrogen is a key ingredient in building and maintaining all forms of life. This is particularly important when it comes to growing plants - both on land and in the sea. Nitrogen is the "limiting" nutrient in these ecosystems. That is, it is often found in least supply compared to the amount required to form life, so whether we are talking about the grass in your backyard or phytoplankton in the ocean (the microscopic grass of the sea), plant  growth is ultimately restricted by the supply of nitrogen. Until just over a hundred years ago nitrogen-fixing bacteria were the only organisms that could tap into the vast, un-reactive pool of N2 gas in the atmosphere. Thus plants and ultimately human population were capped by the amount of reactive nitrogen naturally available on Earth. In the past if we wanted to grow more crops to feed more people we had to harvest fertilizer from other locations. For example, we have applied cow and pig manure to our farm fields, we have harvested seaweed for our vegetable gardens, and we have traveled across the globe to mine guano (or bird waste) deposits. We've even used our own sewage.
growth is ultimately restricted by the supply of nitrogen. Until just over a hundred years ago nitrogen-fixing bacteria were the only organisms that could tap into the vast, un-reactive pool of N2 gas in the atmosphere. Thus plants and ultimately human population were capped by the amount of reactive nitrogen naturally available on Earth. In the past if we wanted to grow more crops to feed more people we had to harvest fertilizer from other locations. For example, we have applied cow and pig manure to our farm fields, we have harvested seaweed for our vegetable gardens, and we have traveled across the globe to mine guano (or bird waste) deposits. We've even used our own sewage.
But none of these activities were actually adding reactive nitrogen to the earth. Instead, we were simply, and perhaps wisely, recycling already available nitrogen. For many years scientists tried to mimic the capabilities of nitrogen-fixing bacteria so we would be able to add nitrogen to the soil and increase our ability to grow food. While many attempts were made and various pieces of the puzzle discovered, it wasn't until the early 1900s that we learned to fix nitrogen in what we now call the Haber-Bosch process. The Haber-Bosch process uses high temperature and pressure to make ammonia and is considered to be the most "important technical invention of the twentieth century" (Smil 2001). In fact, over 48% of the 7 billion people alive today are living because of a chemical engineering feat of the Haber-Bosch process (Erisman et al. 2008).
Because nitrogen can be transformed through various chemical and microbial processes from one form to another it constantly flows through the environment. You can think of nitrogen as a shape-shifter as it can be taken up by biology, secreted as a waste, and taken up again. It can be transformed from a gas to a particulate form bound up in cell and then it can be dissolved in water and make its way to the sea. Between cultivating nitrogen-fixing crops, burning fossil fuels, and fixing nitrogen in the Haber-Bosch process humans have doubled the amount of nitrogen cycling through the biosphere! While this additional nitrogen has been beneficial to many it has also caused unanticipated and negative consequences to terrestrial and aquatic ecosystems and even human health.
In marine systems nitrogen stimulates plant growth - both microscopic phytoplankton as well as larger macro algae. At first, increased growth of 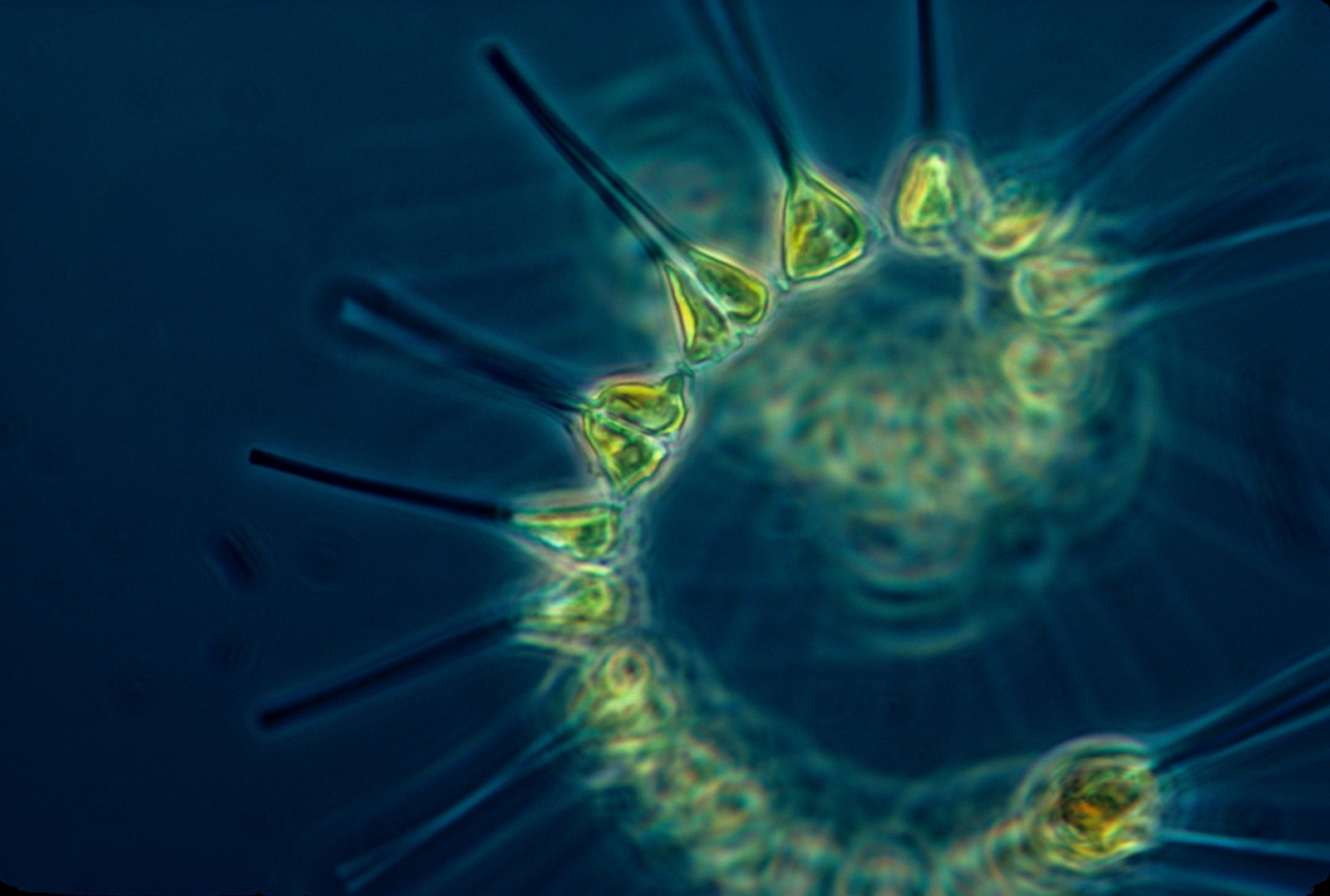 phytoplankton can be beneficial as they are the base of food chains and ultimately support the growth of fish. But as nitrogen additions increase too many phytoplankton and macro algae grow. First, as they grow in the surface waters, this increased phytoplankton or macro algae growth may block light from reaching the bottom thus killing submerged aquatic vegetation (or SAVs). SAVs are critical nursery habitats for important fin and shellfish. In addition, increased nitrogen loading can change the species composition of phytoplankton and harmful algal blooms, like red tide, which are associated with excess nitrogen loading. When the phytoplankton die they sink to the bottom and the natural decomposition by bacteria will use up the oxygen in the water column thus creating hypoxic (little oxygen) and anoxic (no oxygen) conditions. For organisms that cannot move away - like shellfish - these low oxygen conditions can kill them. Thus too much nitrogen leads to excess phytoplankton growth, low oxygen conditions, habitat destruction, and a decrease in biodiversity.
phytoplankton can be beneficial as they are the base of food chains and ultimately support the growth of fish. But as nitrogen additions increase too many phytoplankton and macro algae grow. First, as they grow in the surface waters, this increased phytoplankton or macro algae growth may block light from reaching the bottom thus killing submerged aquatic vegetation (or SAVs). SAVs are critical nursery habitats for important fin and shellfish. In addition, increased nitrogen loading can change the species composition of phytoplankton and harmful algal blooms, like red tide, which are associated with excess nitrogen loading. When the phytoplankton die they sink to the bottom and the natural decomposition by bacteria will use up the oxygen in the water column thus creating hypoxic (little oxygen) and anoxic (no oxygen) conditions. For organisms that cannot move away - like shellfish - these low oxygen conditions can kill them. Thus too much nitrogen leads to excess phytoplankton growth, low oxygen conditions, habitat destruction, and a decrease in biodiversity.
In Part II of this series we'll focus on low oxygen conditions in marine environments - also known as Dead Zones.
References
Erisman, J. W., M. A. Sutton, J. Galloway, Z. Klimont, and W. Winiwarter. 2008. How a century of ammonia synthesis changed the world. Nature Geoscience 1: 636-639.
Smil, V. 2001. Enriching the Earth- Fritz Haber, Carol Bosch, and the Transformation of World Food Production. MIT Press.
- Previous Barnacles "mussel" in
- Next The Doctors who Poisoned Children










Comments
The increase of avaiable
The increase of avaiable Nitrogen in the ecosystem has lead to an inccrease in the Earths population, is this a good thing already there are more of us than the Earth can comfatablye support.
Add a comment