Kitchen Science - The Science of Scotch
Interview with
Ben - Hello and welcome to this week's beer related kitchen science, this is one that you can't do at home not only because heating alcohol could be dangerous, but also because this isn't actually legal to do at home, so you could get in a lot of trouble. I've come today to the University of Cambridge's chemistry department, and I'm here with biophysicist Chris Forman.
Chris - Hello!
 Ben - Today's show is all about beer, so what is it that you're going to show us?
Ben - Today's show is all about beer, so what is it that you're going to show us?
Chris - Well actually today we're going to show you how to make whisky from a substance that's actually very similar to beer. As a model substance we're actually going to use some real beer, and we're actually going to distil it in the apparatus that we've set up here.
Ben - So you make whisky by distilling something a bit like beer?
Chris - Yeah, the whisky starts off as barley, water sugar and yeast and then you let it brew. It's the yeast in that mixture that makes the alcohol, but unfortunately the yeast can't get it beyond 15%. So if we want something with a bit more kick, we're going to have to do a bit of separation to concentrate the alcohol in the beverage.
Ben - So how do you go about concentrating the alcohol?
Chris - Well, the process is known as distillation, and it's not concentration so much as separating it out from the mixture. The way that it works is fairly straightforward: If you have a liquid, ther will always be atoms and molecules evaporating, and there will always be atoms and molecules in the gas condensing back into the liquid. But when you have a mixture of liquids, for example alcohol and water, the gas that's produced will have a mixture of alcohol and water but in different proportions - that's very important, in the gas phase there's more alcohol than there is in the water. So consequently, we can capture that gas, re-condense it and then you have a liquid which has a higher concentration of alcohol and that is the process of distillation.
Ben - So it relies on the fact that different substances in your original liquid have different boiling points, effectively, and so will be present in different proportions in the vapour that comes off the liquid?
Chris - That's pretty much on the nose, although it's important to realise that the mixture itself has a boiling point and that all components will evaporate into the gas, so the gas is never a pure form, it's always a mixture. For example, in beer, you've got water, alcohol and all the tasty stuff, but in the gas phase, what you'll have is water, alcohol and all the tasty stuff just in different proportions.
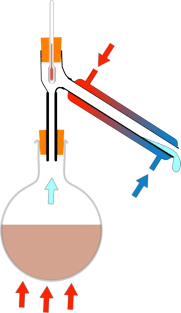 Ben - Well I can see you've got a round bottomed glass flask, and that's in a heater, and coming straight up from that is a tube with an opening in the top, into which you've put a thermometer, and an opening in the side attached to another tube that's angled down to a pear shaped flask. The tube coming out of the side has an extra glass tube around the outside of it, which has got water coming through it to make things cold. But once we've heated the beer up what actually happens to the gas?
Ben - Well I can see you've got a round bottomed glass flask, and that's in a heater, and coming straight up from that is a tube with an opening in the top, into which you've put a thermometer, and an opening in the side attached to another tube that's angled down to a pear shaped flask. The tube coming out of the side has an extra glass tube around the outside of it, which has got water coming through it to make things cold. But once we've heated the beer up what actually happens to the gas?
Chris - As the gas goes up the column above the flask, it will reach the top of the condenser and begin to condense. And it drips out of the bottom into our pear shaped flask here. Hopefully, it wont all go that way, that is to say, pear shaped, it will all work quite well.
Ben - Okay, excellent. Well I have brought along a bottle of beer that I have smuggled into the chemistry department, so shall we crack that open and get on with it?
Chris - Sure, why not? [opens bottle] Okay, so I'm pouring the beer in, and it's draining down through the column and into the flask.
Ben - So how much beer are you putting in there?
Chris - It's about 70ml.
Ben - So we're not putting too much beer in there. Is that because we don't need much? Will this get much alcohol?
Chris - It won't get that much, this is 5.6% so we should get about 3 or 4ml of alcohol out. So we've just got to let the bubbles die down, pop a thermometer in
Ben - So we've put a thermometer in above the beer, why do we need this?
Chris - Right, okay, this is very important. If you put the thermometer in the liquid, you will measure the temperature of the liquid. What matters is the temperature at which that vapour is. Measuring the temperature in the vapour phase is crucial.
Ben - Okay, so we have a tube ready to cool things down, we've got a thermometer ready to measure the temperature of the vapour, where do we go from here?
Chris - We have to start heating the beer, and that's what we'll do now!
Ben - Excellent, so we'll put the heater on. What sort of temperature do we want the beer to be at?
Chris - Well in a pure ethanol and water mixture, that will start to distill at 78 degrees Centigrade. Because the concentration of ethanol in this is much lower, the point where you get distillation will be a little bit higher.
Ben - So how long do you think it will take for our small amount of beer here to get up to the right temperature?
Chris - It wont take too long, only about 15-20 minutes.
Ben - Excellent, well we're going to leave the beer to distill, and we'll come back to you later in the show to let you know what happened. That gives me chance to go to the pub, have a pint and learn a bit more about the damage that alcohol can do to your body.
You can find the interview with Mike Allison, about the damage alcohol can do to your liver,
here.
The second part of this week's Kitchen Science, where we find the result of our distillation, is
here.










Comments
Add a comment