How long can you hold your breath - 30 seconds? 2 minutes? Enough to cross from one end of the pool to the other? The world record for breath holding is 19 minutes and 21 seconds... but don't try it at home - this was accomplished after breathing pure oxygen for ten minutes and doctors worry such feats might have serious health complications. For most of us, while we go about our land-based business, we think little about breathing. We notice that when we run too fast we breathe more heavily, that when we get too excited we breathe faster, or - if you are like me - when we get stressed sometimes we hold our breath. We take our breathing for granted and we don't typically worry that we might run out of oxygen. But if you lived in the ocean, this would be a real and, I am sad to say, ever increasing concern. There are two main reasons for this - one due to the physical properties of water, and the other due to human activities on land.
We don't think about running out of oxygen on land for good reasons. First, air holds more oxygen than water. In fact, a given volume of air contains approximately 40 times more oxygen than the same volume of seawater. The actual amount of oxygen (or any gas) dissolved in seawater also varies according to the temperature and salinity of the water. The warmer and saltier the water is, the less oxygen it can hold. In addition, water is harder to mix than air, so if winds or currents do not stir up the water then the supply of oxygen won't be rejuvenated from the overlying air. In turn, as organisms breathe the local oxygen concentration drops. This is exacerbated in summer when the sun warms the water surface, setting up a temperature-induced stratification (layering of the water column). In addition, in estuaries - the coastal zones where freshwater and seawater meet and mix - stratification develops as freshwater enters the sea. The freshwater is lighter and more buoyant, so it floats on top of the seawater like a lid. Together, freshwater and warming temperatures decrease mixing, so the oxygen concentration towards the bottom of the water column is rapidly depleted with no way to replenish it.
 Typically, such low oxygen events caused by freshwater and warmer temperatures are transient and water column oxygen would be rejuvenated with the changing of the tide or the summer-afternoon southerly ocean winds. However, these days low oxygen conditions are found in many coastal waters and, alarmingly, are occurring at both an increased frequency and duration. If you were to look at a map of these low-oxygen hot spots (and there some good ones available, see the links below) you'd notice that most of these locations are adjacent to major population centres. This is because where there are humans there is nitrogen, which is the limiting nutrient in marine ecosystems. This means that the amount of nitrogen available in the environment will determine how much plant growth occurs. In the marine environment, nitrogen can stimulate the growth of various plants including phytoplankton (the microscopic grass of the sea), submerged aquatic vegetation (e.g. eel grass, turtle grass) and macroalgae. Like all things, too much nitrogen leads to excess plant growth most often seen as increased phytoplankton or macroalgae production. If you can remember those long-distant science lessons you might recall learning that plants photosynthesise and in doing so release oxygen, so you're probably wondering why it is that more plant growth could lead to lower oxygen conditions?
Typically, such low oxygen events caused by freshwater and warmer temperatures are transient and water column oxygen would be rejuvenated with the changing of the tide or the summer-afternoon southerly ocean winds. However, these days low oxygen conditions are found in many coastal waters and, alarmingly, are occurring at both an increased frequency and duration. If you were to look at a map of these low-oxygen hot spots (and there some good ones available, see the links below) you'd notice that most of these locations are adjacent to major population centres. This is because where there are humans there is nitrogen, which is the limiting nutrient in marine ecosystems. This means that the amount of nitrogen available in the environment will determine how much plant growth occurs. In the marine environment, nitrogen can stimulate the growth of various plants including phytoplankton (the microscopic grass of the sea), submerged aquatic vegetation (e.g. eel grass, turtle grass) and macroalgae. Like all things, too much nitrogen leads to excess plant growth most often seen as increased phytoplankton or macroalgae production. If you can remember those long-distant science lessons you might recall learning that plants photosynthesise and in doing so release oxygen, so you're probably wondering why it is that more plant growth could lead to lower oxygen conditions?
Well, at first, as the phytoplankton grow in the surface waters they transform sunlight and nutrients into minute green cells. Oxygen is produced and its concentration within the water column does initially increase. But, when they stop photosynthesising - either temporarily at night or permanently when they die - they stop creating oxygen. In time, their small, delicate bodies fall through the water column to the depths of the sea floor or the estuary bottom. Here, these nutrient-laden carbon-rich morsels are decomposed by hungry bacteria. As the bacteria decompose the dead phytoplankton they also consume oxygen. This ultimately leads to hypoxic (low oxygen) or anoxic (no oxygen) conditions. In some cases these hypoxic conditions last just the night; in other cases these hypoxic and even anoxic conditions persist in both space and time earning the name of "dead zones."
Perhaps the most infamous dead zone is the one located in the Gulf of Mexico. 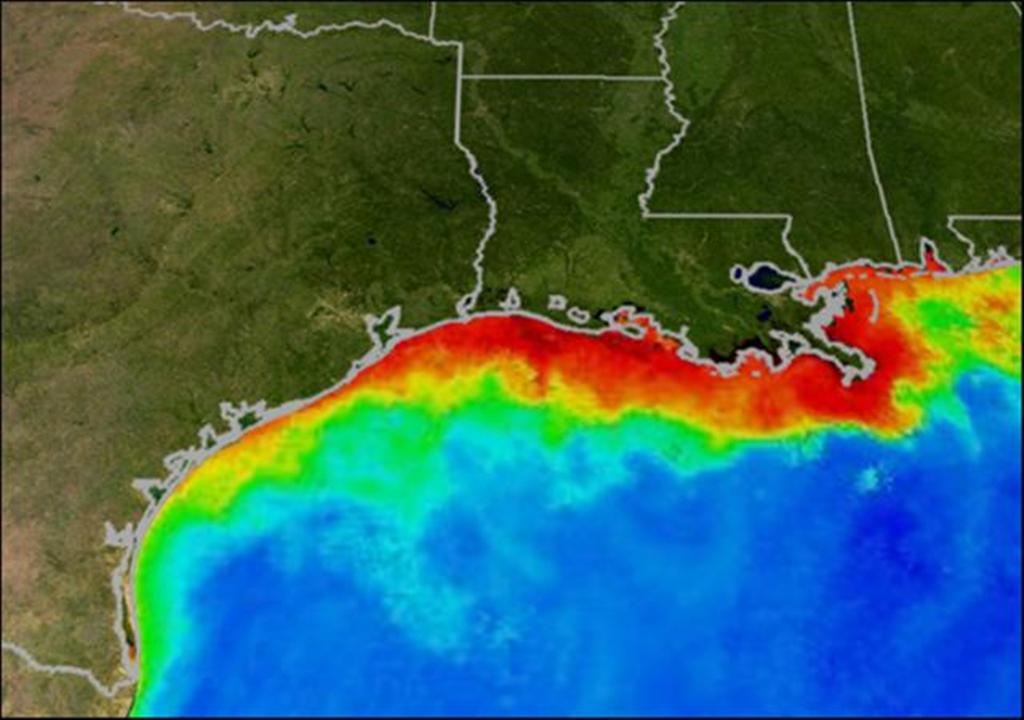 This dead zone has been linked to nitrogen fertiliser runoff from the agricultural basin in the United States. Owing to heavy flooding this year, the Gulf of Mexico dead zone is predicted to be the largest yet recorded - somewhere between 22,000 and 24,000 square kilometers. That's almost the size of the Island of Sicily or the State of New Hampshire. Unfortunately, the prevalence of known dead zones is increasing, and since the 1960s their total number has doubled each decade (Diaz and Rosenberg 2008).
This dead zone has been linked to nitrogen fertiliser runoff from the agricultural basin in the United States. Owing to heavy flooding this year, the Gulf of Mexico dead zone is predicted to be the largest yet recorded - somewhere between 22,000 and 24,000 square kilometers. That's almost the size of the Island of Sicily or the State of New Hampshire. Unfortunately, the prevalence of known dead zones is increasing, and since the 1960s their total number has doubled each decade (Diaz and Rosenberg 2008).
Of course, the ultimate question is does it matter? Why do we care that vast areas of the coastal ocean are experiencing low oxygen? And what does it mean for marine systems or for us? In Part III I will discuss the many negative consequences of dead zones. Meanwhile, please don't hold your breath...
References
Diaz, R. J., and R. Rosenberg. 2008. Spreading dead zones and consequences for marine ecosystems. Science 321: 926-929.
Maps of Dead Zones:
1. http://earthobservatory.nasa.gov/IOTD/view.php?id=44677
2. http://www.cop.noaa.gov/stressors/extremeevents/hab/features/Diaz_08.aspx










Comments
Add a comment