A 3d-printer capable of building organs and tissues by printing complex arrangements of cells has been unveiled by US scientists.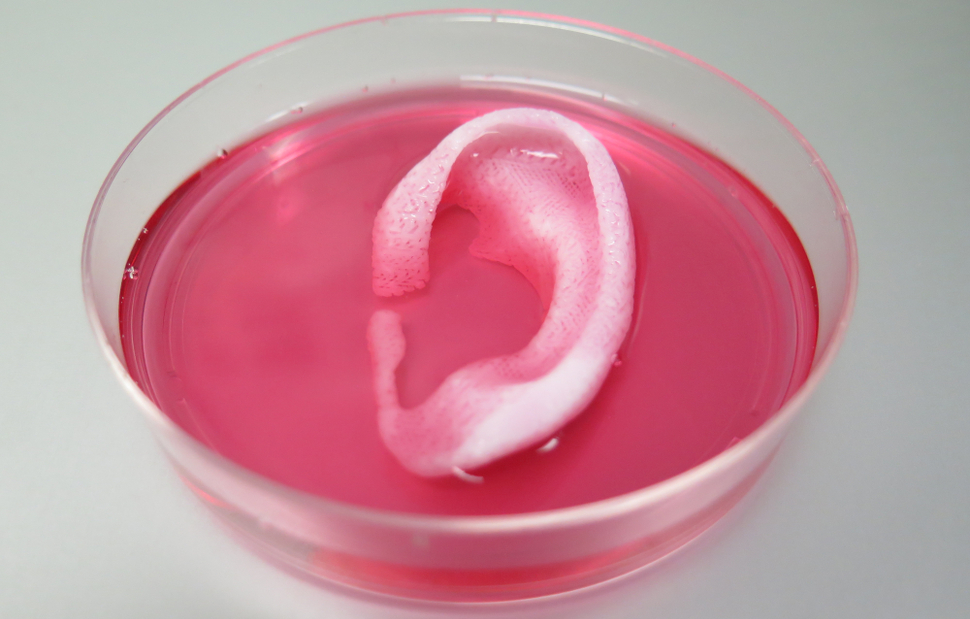
3D-printers are revolutionising science, technology and engineering. Specialist manufacturers are using them to print parts for jet engines; chemists are creating miniature test-tubes guaranteed to produce the right reaction products; archaeologists printed the spine of Richard III to recreate his back deformity and confirm his identity, and doctors have used the technique to create templates for bespoke body implants including previously a replacement piece of windpipe.
But the medical applications of 3D printing remain limited by the fact that the body is composed of many different types of living cells, all of which cooperate in special ways to form tissues. And printing mixtures of cells in a way that keeps them alive and also in the right position to form a functional tissue has been impossible.
Now a team led by Wake Forest School of Medicine researcher Anthony Atala have announced the 3D bioprinter, which can produce human scale complex tissues made up multiple cells types.
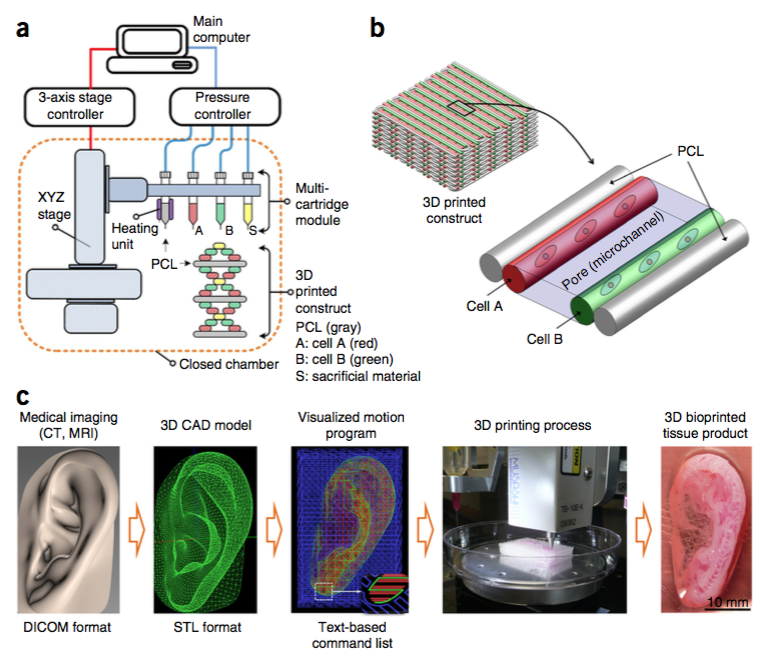
Dubbed ITOP, which stands for integrated tissue-organ printer, their system consists of a series of spray nozzles that release a nutrient-rich hydrogel packed with cells.
Different nozzles are loaded with different cell types. Another nozzle delivers a glue-like matrix that contains, among other things, the same substance that makes blood clot.
Initially, a computer programme takes the data from a CT or MRI scan of the desired structure and converts this into a series of movements required by the print head and stage on which the tissue rests.
The spray heads then deposit cell-rich droplets in precisely the right configurations to grow the desired tissue.
The cells are then cemented in place by rows of the glue material. Critically, this approach allows spaces or pores to be left between the rows of cells. These function like blood vessels, allowing nutrients to reach the cells and waste products to diffuse safely away as the tissue grows.
Once printing is complete, the signal used by the blood to trigger clotting, an enzyme called thrombin, is added. This activates the clotting chemical in the glue, fixing everything in place.
In their paper in Nature Biotechnology, the team test the approach by printing a 3cm piece of human jawbone using human bone-producing stem cells. More than 90% of the printed cells survived the process and remained viable more than a week later
The team also printed replacement pieces of skull bone that were successfully implanted into the heads of rats. Examined five months later, the new bone tissue was healthy, had knitted effectively into place and had plumbed itself into the local blood supply.
Next, the researchers printed a human ear using data from a CT scan. Cartilage-producing cells called chondrocytes were deposited and the ears were implanted onto the backs of mice where they grew and developed over the following two months.
Finally, the researchers also tested the ability of the approach to print new muscles using muscle stem cells called myoblasts. These were connected to a nerve in the back legs of a group of rats and, after two weeks, showed electrical responses to nerve stimulation.
"With further development, this technology may produce clinically useful tissues and organs that incorporate multiple cell types at precise locations to recapitulate native structure and function," the researchers say.










Comments
Add a comment