Scientists have found a way to selectively shut down the components of the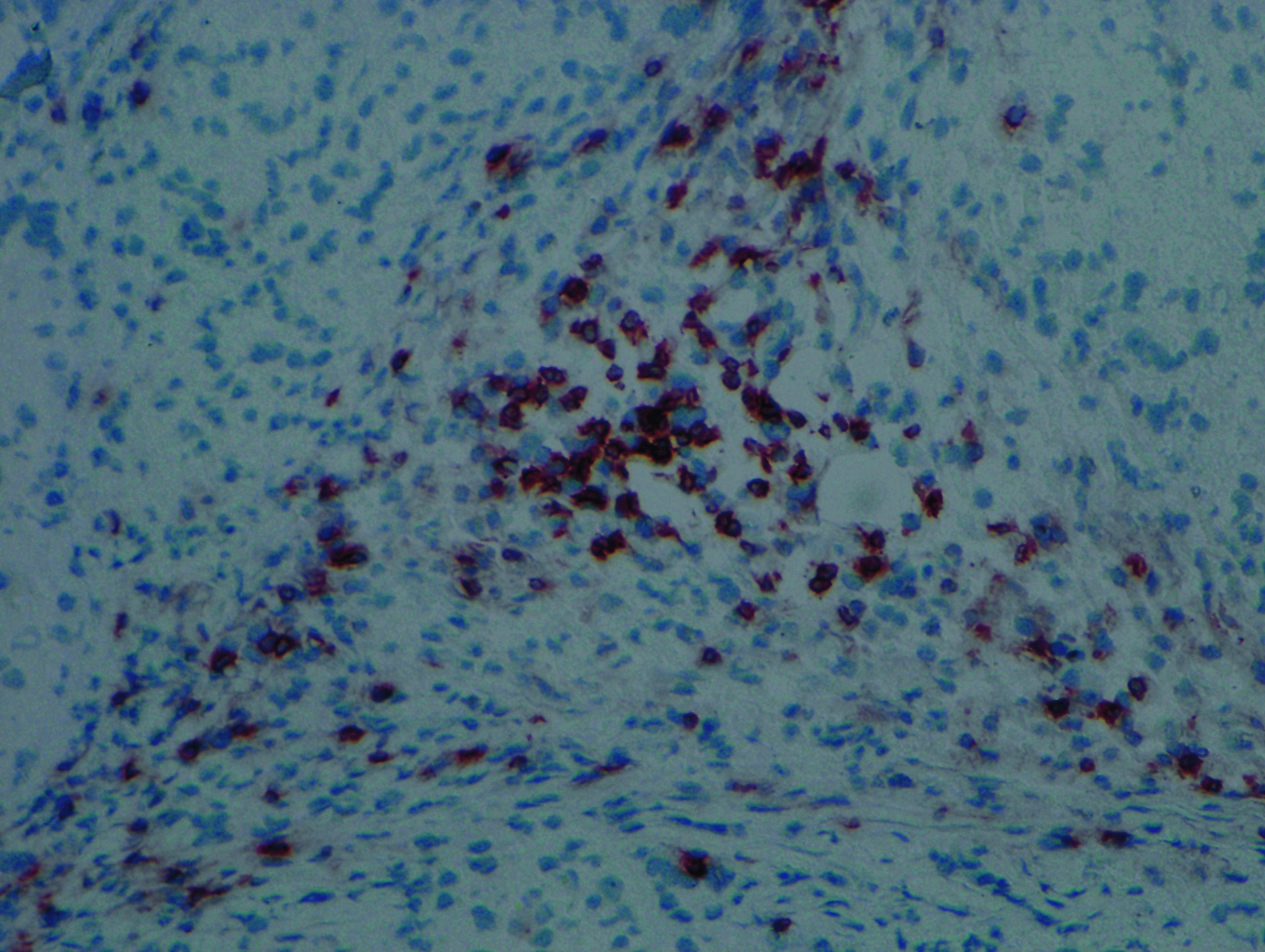 immune system responsible for triggering autoimmune diseases and rejecting transplanted organs.
immune system responsible for triggering autoimmune diseases and rejecting transplanted organs.
The discovery hinges on a population of rare immune cells called regulatory T lymphocytes, or Tregs. These cells, which make up about 1-5% of all the white blood cells in the body and were first discovered in the 1990s, work to stop other immune cells from reacting to certain antigens. Consequently, doctors have for a number of years sought to harness them as a natural form of immunosuppression to prevent patients from rejecting donor organs.
But the relative rarity of the cells, coupled with the fact that they can switch off immune surveillance against potential pathogens and even cancer, has frustrated attempts to exploit them.
Now, writing in Science Translation Medicine, a team based at King's College London and led by Giovanna Lombardi have developed a technique to sift out only those Tregs capable of recognising donor tissue. These handpicked cells can then be grown in a dish before being reinfused into the body to fend off immune attack against a donor organ.
Using experimental mice as a model for the technique, the Kings team first grafted a piece of human skin tissue onto the animals, which have been modified to accept human tissues.
After the grafts had "taken" the team then infused white blood cells mis-matched with the skin to provoke rejection. In one group of the animals, they also infused a million T reg cells selected previously to recognise the skin cells, while control animals received just a salt solution lacking any cells.
Skin biopsies subsequently showed that the control animals had significant signs of immune attack within the grafted skin, consistent with immune rejection of the foreign tissue. But the animals that had received the skin-specific T regs was significantly protected against the immune-onslaught.
This shows that it should be feasible to tailor-make donor-specific cell therapies, using a recipient patient's own cells, to combat immune rejection of donor organs or possibly also prevent autoimmune disease, but without the attendant risks of causing cancer or rendering a patient highly susceptible to infectious diseases.
- Previous Amazing Amazonian geometry
- Next Gossip makes faces more memorable










Comments
Add a comment