Scientists have uncovered the genetic switch system that activates and deactivates the herpes viruses that cause cold sores.
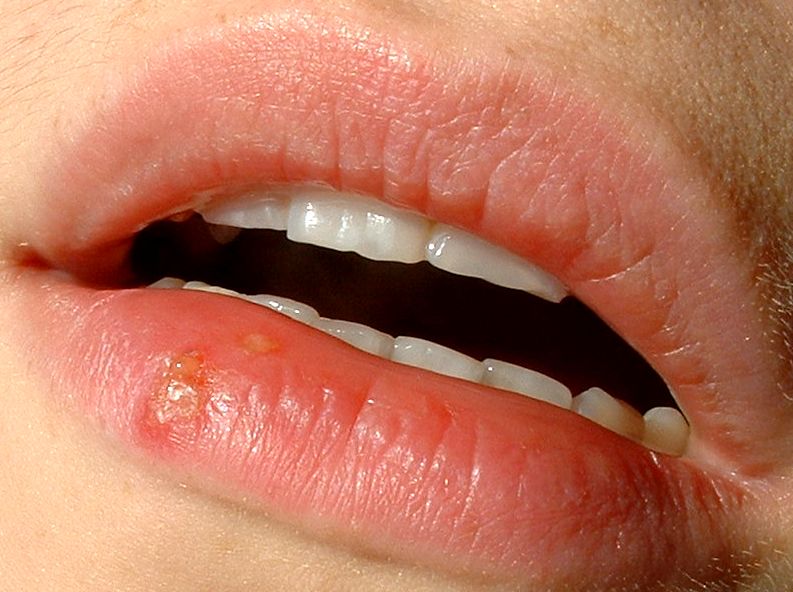 80% of the population carry the virus, which is normally picked up in early childhood, in their nervous systems. The virus loiters in the form of a small piece of DNA, which sits inside the nucleus of sensory nerve cells supplying the skin. This state is called latency, but periodically the viral DNA is re-awakened in some nerve cells and orchestrates the production of new virus particles which are shipped back along the nerve to the skin surface. There they enter skin cells to produce infectious cold sores, allowing the virus to be passed on to other susceptible individuals.
80% of the population carry the virus, which is normally picked up in early childhood, in their nervous systems. The virus loiters in the form of a small piece of DNA, which sits inside the nucleus of sensory nerve cells supplying the skin. This state is called latency, but periodically the viral DNA is re-awakened in some nerve cells and orchestrates the production of new virus particles which are shipped back along the nerve to the skin surface. There they enter skin cells to produce infectious cold sores, allowing the virus to be passed on to other susceptible individuals.
Now Duke University researcher Jennifer Umbach and her colleagues, writing in this week's Nature, have discovered that the virus exploits a Nobel prizewinning technique to enable it to lurk, undetected like this, inside nerve cells for the majority of the time. The team focused their attention on a region of the viral DNA code called the LAT - latency associated transcript, which is the only part of the viral genome that remains active while the virus is dormant. They found that this region of DNA produces a family of small molecules called micro RNAs. These turn out to be the genetic mirror images of parts of some of the key genes that drive the wake-up programme that normally reactivates the virus and culminates in a cold sore.
The team think that these micro RNAs damp down this process by blocking the expression of these key genes using a process called RNA interference, which won the Nobel prize for the two American scientists, Mello and Fire, who discovered it. This keeps the virus genetically "on ice" until some other stimulus, such as damage to the cell or chemical stress, triggers it to reactivate.
The researchers think that this discovery might be the key to ridding sufferers of the scourge of recurrent herpes. They are now testing a drug that can precisely and selectively block these micro RNAs, which should provoke the virus to reactivate at which point it can be killed with the already well-established anti-herpes drug aciclovir.
"In principle you could activate and then kill all of the virus in a patient," says principal investigator Bryan Cullen.










Comments
Add a comment