Coke Can Eruption
24 June 2007
Ingredients
At least 2 cans of fizzy drink
A hand
Instructions
Shake up both cans
Leave them on their side for 10-15sec
Turn them upright.
Tap one sharply on the top 4-5 times
Wait 10sec
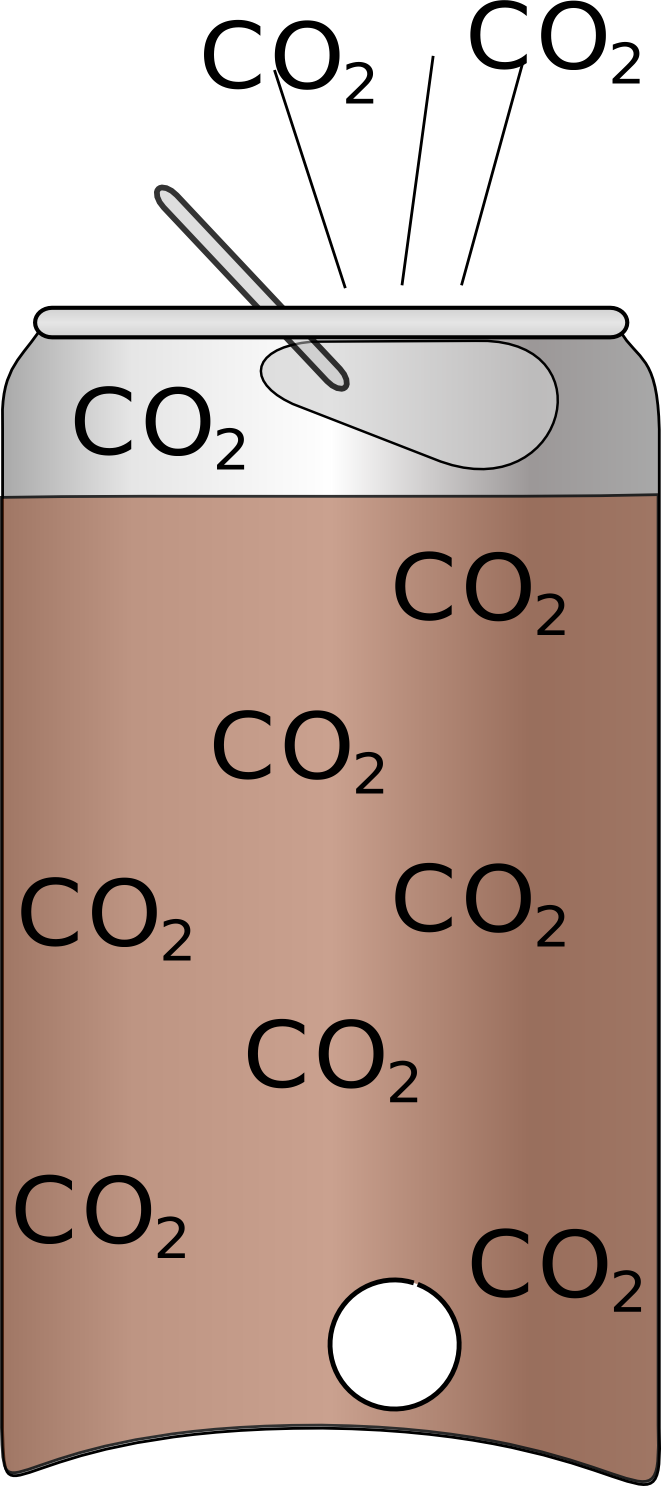
Open both cans.
Is there any difference.
Result
In theory, the can that you tapped should froth out less than the one that you didn't!
Explanation
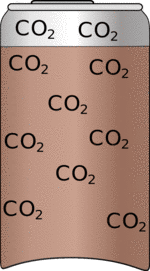 |  |
| Fizzy drinks are fizzy because they have loads of carbon-dioxide dissolved in them under pressure. | So when you release the pressure by opening the can the carbon-dioxide will come out of the liquid. Bubbles are difficult to start so if the can is clean not many bubbles will form. |
 | 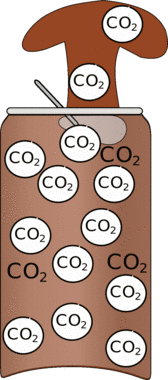 |
| If you have shaken the drink you will have mixed a whole lot of bubbles into the liquid. | These bubbles have already started so if you open the drink immediately the bubbles can expand rapidly creating foam. This foam is much larger than the can so the drink will splurt out. |
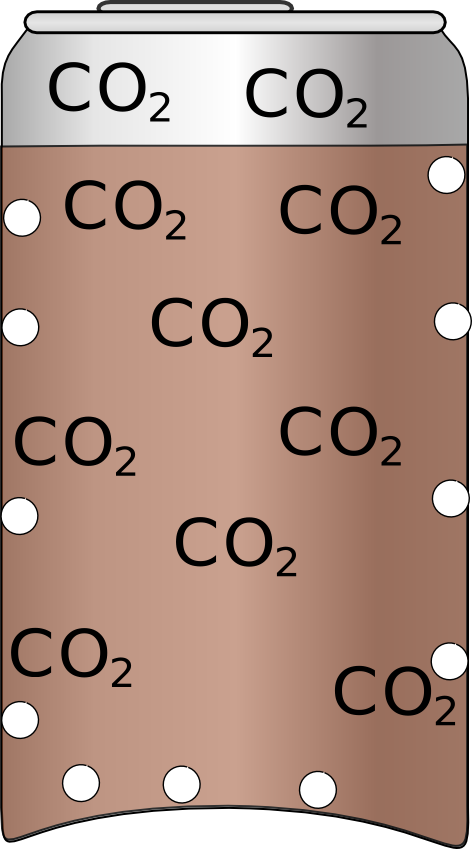 | 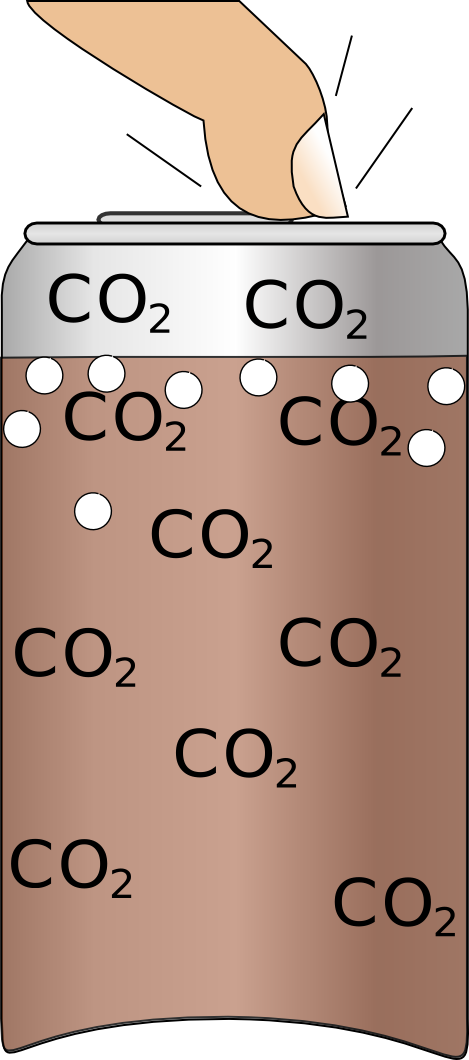 |
| Often when you shake a can bubbles get stuck to the sides which will stay there until they dissolve which will take 10s of minutes. | If you tap the can it will dislodge some of the bubbles and if you give them long enough to reach the surface it should reduce the amount of foam produced. |
- Previous Cup Fountain
- Next How fat would you have to be to stop a bullet?










Comments
Add a comment