Ingredients
In theory this is a relatively simple experiment to do, but in that form it is really quite dangerous, so DO NOT try it at home! I spent a long time building a box with lots of interlocks and thinking about where electrified gherkin juice was likely to flow...
Anyway, a pickled gherkin (or pickle to some) is just a small type of cucumber which has been boiled and then pickled in a mixture that is mostly made up of salt and vinegar. The boiling breaks open all the cells so you effectively have a tube of salty water in a convenient package. Salty water conducts electricity quite well, so a large current flows and the gherkin starts to heat up, then something rather wonderful happens.
The electric currents are concentrated near the fork prongs and heat up the gherkin so much that water starts to boil and escape as steam.
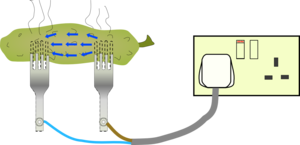 This will dry out the area around the prongs until it ceases to be conducting any more.
This will dry out the area around the prongs until it ceases to be conducting any more.
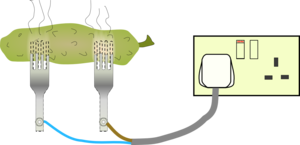
This means that there is a large voltage across a very small insulating gap and this is large enough to rip the electrons off the air causing it to become conducting and electricity flows through the air in the form of a spark.
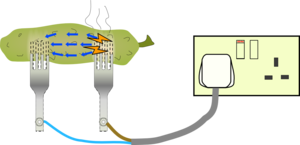
Sparks are extremely energetic and they will transfer energy to all the atoms around them. One common atom in a pickled gherkin is Sodium from the salt (sodium chloride). Sodium is a very efficient emitter of orange light so the sparks look orange.
Sodium is such an efficient emitter of light that it is used in streetlights, which is why they are often the same bright orange colour. Find out more about the light they give off by
clicking here










Comments
Add a comment