Making a Mess with Milk
Ingredients
 | A glass of milkIdeally with some fat in it |
 | 2-3 tablespoons of vinegaror lemon juice. |
Instructions
This is incredibly simple, just put some milk in the glass and then add a couple of tablespoons of vinegar to it.
What happens to the milk?
Result
The milk will turn into a mixture of a clear liquid with white lumps mixed in.

If you filter the mixture through a piece of kitchen paper or a coffee filter you can seperate the clear liquid from the solid. The solid can be pressed into a fairly solid lump.

Explanation
Milk is made up of water, globules of fat and little lumps of caesin protein and Calcium phosphate (the mineral that makes our bones hard) called micelles. The caesin is repelled by water which is why it forms the micelles, the outside of the micelles is negatively charged so they will repel other micelles and they don't stick together.
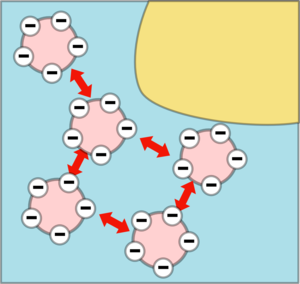
Vinegar is an acid which means it contains lots of positively charged hydrogen (H+) ions which will neutralise the negative charges until the micelles stick together forming a big sticky network. This tends to catch the fat globules and form the the white lump you saw in the experiment which are called curds. The watery stuff left over is called whey. This process is called curdling.
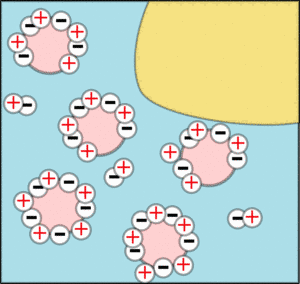 |  |
| The negative charges are cancelled out by the acid. | So there is nothing to stop the micelles coagulating and taking the fat with them. |
What has this got to do with cheese?
 Cheese is made from the coagulated lumps - known as the curds, the liquid is called whey, hence curds and whey. There are various ways of doing this although the most common is to add an enzyme called Rennet, this is used by calves to curdle the milk in their stomachs to make it easier to digest. It attacks the caesin and causes it to precipitate.
Cheese is made from the coagulated lumps - known as the curds, the liquid is called whey, hence curds and whey. There are various ways of doing this although the most common is to add an enzyme called Rennet, this is used by calves to curdle the milk in their stomachs to make it easier to digest. It attacks the caesin and causes it to precipitate.
Cottage cheese is basically just the curds roughly strained out, other cheeses are mostly pressed to remove water, and matured sometimes with bacteria or fungi to add to the taste.
- Previous Cloud in a bottle
- Next Pinhole camera










Comments
Add a comment