Microwaving Ice - why defrosting is so slow.
Ingredients
 | A microwave |  | Some ice |
 | A couple of microwave proof glasses or cups |  | Scales |
Instructions
Take 2-3 ice cubes out of the freezer, weigh them and put them in a microwaveable cup.
Weigh the same amount of water into the another cup.
Put both cups in the microwave in symmetrical positions - equidistant from the centre of the turntable.
Turn the microwave on and watch the cups to find out whether the ice will melt or the water will boil first.
Result
Although it only takes an increase of a few °C to melt the ice, and the water has to heat up by about 80°C to boil, the water will normally boil before the ice melts!
Explanation
There are two effects happening here, one of them is that to melt ice takes a huge amount of energy, the equivalent of heating water by about 80°C. This is because in ice water molecules are locked together by quite strong hydrogen bonds, and to melt it has to break a lot of these bonds, which takes a lot of energy.
The second effect is that ice just doesn't absorb microwaves nearly as well as water - this means it actually heats up less.
Why does water absorb microwaves better than ice?
A microwave oven cooks by creating microwaves, a form of light or electromagnetic radiation. These are produced on the right hand side of the oven and are sent into the main oven. They reflect back and forth creating a standing wave.
 | 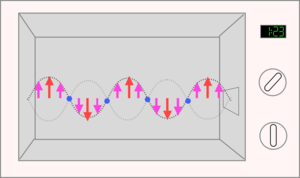 |
| The microwave oven gives out microwaves (a form of electromagnetic radiation) which then reflect back and forth across the metal box. | As they do this they create an electric field which will point up and then down about 2.5 billion times a second. |
In the microwave the electric field will point upwards and then downwards about 2.5 billion times every second. This means that anything charged will be pulled upwards and downwards by the field produced by the microwaves.
Although water molecules don't have an overall charge, their oxygen atom is slightly negative and their hydrogen atoms are slightly positive (they have a positive and negative pole so are called polar). This means that in an electric field they will rotate to align with the field.
 | 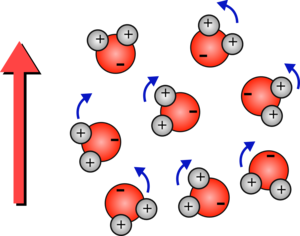 |
| Each water molecule is made up of one oxygen atom which is slightly negative and two hydrogen atoms which are slightly positive. | If the electric field points upwards then the positive hydrogen atoms are pulled upwards and the negative oxygen atoms are pulled downwards, so the molecules rotate. |
If the field keeps changing at 2.5 billion times a second they will keep rotating which will give them energy so the water heats up.
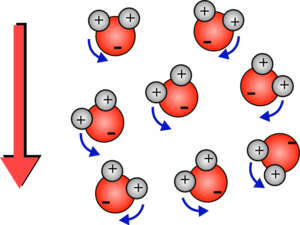 | 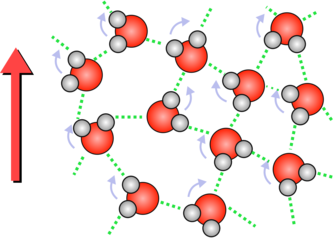 |
| If the field now points downwards then the oxygen atoms will try an reverse their direction. All of this rotation transfers energy to the water and heats it up. | Although the ice molecules also feel a rotational force they are locked into a crystal and rotating would involve breaking bonds between molecules. So they hardly move and they absorb very little energy. |
In ice the water molecules are all locked together in a crystal structure by hydrogen bonds. These bonds will stop the water molecules rotating, which means they can't absorb much energy from the microwaves. This, in turn, means that the ice doesn't heat up.
This is why you shouldn't defrost a chicken on full power. As soon as some of the frozen chicken melts, the water it contains will be in the form of liquid water - which will absorb microwaves far better than the surrounding frozen water. This means you will end up with part of your chicken fully cooked while the rest is frozen, and your meal will be horrid, if not dangerous!
The defrost setting on microwaves gets around this by by only heating for a few seconds every minute and essentially waiting for the hot bits of your chicken to melt their neighbouring areas.
What is a hydrogen bond?
The slightly negative oxygen of one water molecule will attract the slightly positive hydrogen from another. This attraction will pull the molecules together and forms what is known as a hydrogen bond.










Comments
Wow! Science is magic! Thank
Wow! Science is magic! Thank you for letting us know the reason behind it
Awesome, was just wondering
Awesome, was just wondering this!
Add a comment