Eroding Coastlines and Holy Grails - A look back at 2010
This month we look back at Diamond's scientific highlights of 2010 to reveal how microbes are eroding away our coastline and how metal organic frameworks could help find the holy grail of chemistry! We also hear how the synchrotron was improved to provide more beamlines as well as bring you the latest research from these beamlines including stresses on jet engines and the never-ending fight against antibiotic resistance.
In this episode

01:41 - Look back at 2010
Look back at 2010
with Trevor Rayment, Diamond Light Source
Meera - 2010 saw many changes and improvements to the Diamond Synchrotron as well as good news as to how it will continue to improve in the future. Diamond's Physical Sciences Director, Trevor Rayment, explains more about the synchrotrons journey to completion as Diamond gears up for Phase III;
Trevor - This is important for Diamond because it's always been seen as a 3 phase development project. In Phases I & II we built the machine and are close to completing 22 Beamlines and in Phase III we'll add a further 10 of these Beamlines. So, Phase III is going to improve our capacity for doing science and engineering technology by about 50%.
Meera - So how are the Phases broken up? What make Phase I & II & III separate?
Trevor - Well, the way a synchrotron operates is that it provides a number of quite independent scientific facilities that we call Beamlines, which just simply describes the fact that the light is a beam and it comes down the line of equipment. But perhaps a better way of thinking about these is as independent scientific laboratories. Every laboratory is designed for a specific task and if you want to have a broad scope then you need to have a large number of beamlines because each one is very specialised and designed for a specific scientific purpose. By adding a further 10 of these Beamlines, we're improving the scope of what we can do enormously.
I'd like to give you a couple of examples; There are a series of methods that have been developed outside synchrotrons, in other areas of science, called High Throughputs where you deliberately employ automation, robotics, artificial intelligence, to squeeze the very most out of your instruments. And this does two things; first of all it enables more people to access the facility, but not only that, it enables you to do things that you couldn't dream of doing before, simply because it would take too long. You could ask questions that are more difficult simply because you could do more experiments, more quickly. Phase III gives us increased volume and it gives us hugely increased scope of what we can do.
Meera - You mention the light generated by the core synchrotron itself is what changes and what is altered for each beamline, but in 2010 a change was actually made to the core synchrotron and the way it produces its light.
Trevor - Yes it was. If you imagine a synchrotron, looking down from above, the way we normally explain it is as a many-sided coin, a 24 sided polygon. That gives you the impression of the electrons travelling in a straight line between magnets that steer the beams, the electrons will go in a straight line and then be steered round a corner, they'll do that 24 times all together to go round. Then on the long sided sections of this polygon, we put what we call Insertion Devices - purpose built magnetic structures that produce the light that we want. But electrons are not really like that. All electrons are negatively charged and if you just put them together and leave them alone, they'll repel each other. And so what that means is that as the electrons are travelling along in a straight line, they're also moving apart from each other which mean the beam gets bigger. So part of the structure of the machine are components to refocus the beam back down to a very small point. So what you have to imagine is not only do you have the electrons moving in a straight line but they're diverging and you have to refocus them. Now in the original structure of Diamond they were arrange so the electron beam was focused at the centre of each of these straight sections because that is where we would put the insertion devices.
Some of these straight sections are actually quite long, they're 5 metres long and there is in fact space to put 2 of these Insertion Devices. So in principle you could have 2 sources of light rather than 1, and that would mean you could build 2 beamlines rather than 1 beamline. However, the fact that the electrons are focused at the centre means that if you have 2 Insertion Devices, they've both got to be off centre and neither is in the optimum position. So what they've done this year is they've changed the structure of the machine such that they can insert additional focus lenses at the end of the straight sections and also in the middle of the straight section so that now the electrons, as they enter this straight section, are focused very early on by diverge and then they're focused back in again on the straight section and then out. So it becomes, instead of having one dip in the middle, it's got a double dip.
Meera - So essentially this is leading to more regular focusing of electrons along their route around the synchrotron?
Trevor - More specialised, I wouldn't say it's more regular, but it's more specialised. The take home message is that the light delivered out to the Users is going to be brighter and more flexible.
Meera - You mention that Diamond's priority is always the Users that come in and use the Facility for their Research and you actually reached a milestone in 2010 in terms of the research and publications resulting from this research.
Trevor - Yes we did indeed. We reached the milestone of 1000 publications produced from Users and from people who work at Diamond. And that's 1000 publications since the start of Diamond, we started operating in 2007.
Meera - Are there any papers that are particularly noteworthy or stand out in your memory?
Trevor - There have been some very interesting studies carried out at the University of Leicester where they have been looking at the fate of depleted Uranium - an Environmental study. There has been work on Lead-free ceramics to make Piezo electrics, that's an important Material Science application and there had been work carried out at Imperial College where they've been able to solve a 20-year old mystery about the way that HIV attacks the body.
Meera - Everything that you've mentioned just reiterates the fact that such a wide variety of scientific disciplines are researched at Diamond, but another important aspect of the facility is that you develop workshops and you have a great deal of Outreach.
Trevor - Yes, that's true. Diamond exists for people who don't use it, as much as it does for those who do. We are now a mature facility and we are keen to attract a very large number of new users and experienced users and one way we do that is we host meetings. These meetings range from Training, Meetings that simply liaise with Users - we simply bring them in, listen to them and listen to their concerns - and we host external scientific societies for example. And of course we welcome lots of visitors from the local area on our Inside Diamond Days.

08:52 - Coastal Erosion at the microbial level
Coastal Erosion at the microbial level
with Charles Cockell, Open University
Charles Cockell - Erosion is a big problem and a large percentage, well over 15% of the UK's coast is subject to coastal erosion. Now this is a natural problem of course and wave action and tidal action causes coastlines to erode but of course it's become more of a problem recently  given our concerns about rising sea levels, also human manipulation of coastal regions that can cause some areas of coast to erode much more quickly than others. Tidal breaks and these sorts of things can cause artificially fast rates of erosion.
given our concerns about rising sea levels, also human manipulation of coastal regions that can cause some areas of coast to erode much more quickly than others. Tidal breaks and these sorts of things can cause artificially fast rates of erosion.
Meera - But you've been particularly looking at an area of the coast along the North Yorkshire coastline.
Charles - That's right, this is an area where there are shales that are weathering quite quickly.
Meera - So what have been thought, up until now, are the causes of this erosion on that coastline?
Charles - So up until now, people have been focusing on the large scale, so they've mapped the faces of these cliffs and seen large scale rock falls, and really begun to understand the physics of how erosion occurs down to the cm scale. But it's still not really understood what's happening at the very small scale, literally the micron scale, the scale of micro-organisms. How do those chemical processes cause erosion and what sort of processes might be involved in causing our coastal erosion. And that's where we come in, because of course microbes are very, very small, a micron in size, and they might be relevant to erosion at these very small scales.
Meera - So the coastline you've been looking at is made up of shale as you've said - which microbes are thought to be having an effect here and what are they actually doing in this area?
Charles - Well we know that microbes can oxidise iron in shale, so they oxidise it and turn it into iron oxide, rust basically, and those new minerals  that are formed can form in the cracks of rocks and contribute to their degradation. But in our rocks we found that very few of the iron oxidises that we found, or if in fact very few of the microbes in that environment were related to things that had been studied before. So we think that there's a novel microbial community in there that is responsible for weathering the rocks. But what's basically happening is that the microbes are basically eating the various minerals in the rocks as a source of energy and in the process they are breaking down the rocks but they're also producing new minerals that are formed on the rock surfaces. And they form in tiny fractures inside the rock and they contribute to the weakening of the rock and eventually to its large scale deterioration and collapse. And that's how a little creature, a micron in size, can actually contribute to erosion on a kilometre scale.
that are formed can form in the cracks of rocks and contribute to their degradation. But in our rocks we found that very few of the iron oxidises that we found, or if in fact very few of the microbes in that environment were related to things that had been studied before. So we think that there's a novel microbial community in there that is responsible for weathering the rocks. But what's basically happening is that the microbes are basically eating the various minerals in the rocks as a source of energy and in the process they are breaking down the rocks but they're also producing new minerals that are formed on the rock surfaces. And they form in tiny fractures inside the rock and they contribute to the weakening of the rock and eventually to its large scale deterioration and collapse. And that's how a little creature, a micron in size, can actually contribute to erosion on a kilometre scale.
Meera - So what microbes are these and how much are they thought to be contributing to erosion on this particular coastline?
Charles - The varieties of microbes are very large. We've extracted the DNA, we've looked at the community and there are all sorts of microbes in there that we don't really understand and we don't know what they're doing. What their contribution is a big question. We know they're there, but we don't know which ones are active in breaking down the rocks and how much they contribute towards forming the minerals. Because what we do know is that weathering can also occur chemically without the microbes, so the next challenge is really to understand the contribution of the microbial community.
Meera - Let's go a bit into how you've been looking into the effects of these microbes, or perhaps how they're even working. This is where you've been using the Diamond synchrotron, but an interesting aspect is that you've been using quite a few different techniques at Diamond to look at very many aspects of the microbes.
Charles - That's right, we use a number of methods. We've been using biological methods where we extract the DNA and we try to understand what the microbial community is doing inside the fractures and how they are distributed and what types of microbes are there. Essentially what we're asking is the question 'Who's in there?'. But we are also interested in understanding the mineral that surround them. What are those minerals, what structure do they have, could they have been produced by microbes? And that's where Diamond comes in, because we can use Diamond to map the different types of iron minerals on the surface of the rock. Try and understand their relationship to the rock surface and what characteristics they have and so we've been using Diamond to map the surface of the weathered fractures in the coastally eroded cliffs. We will then go on to see whether microbes can produce these types of minerals.
Meera - How useful then would you say these techniques are?
Charles - They are extremely important because they allow us to really probe the structure of the minerals. It's one thing just to know what their composition is, but we wanted to know their structure. We also wanted to know how they are distributed on the surface of the rock; are they localised in one place, are they evenly distributed? That tells us a bit about the way in which the rocks are breaking down.
Meera - How would you summarise your findings so far and I guess at the end of it all, what's the aim? I guess we can't obviously prevent it, what could be done using this information?
Charles - Well in terms of stopping these things, it's interesting you say that, the assumption is that we can't do anything about it, the microbes just eat the rocks, but in fact there are fascinating questions about if you know what microbes are weathering these rocks, could you slow them down? Is it possible that as the sea level rises, salt water will actually kill off some of these weathering microbes and slow the process down? Or will new types of microbes take over?
Meera - Another important thing to consider is that the impact of these microbes isn't just limited to the erosion of the actual coast, there's greater effects spanning from this.
Charles - Yes, absolutely. I mean the work that we do gives us a much broader insight into the role of microbes breaking down rocks generally. What role do these microbes play in other shale environments? Shale has a very important role in engineering structures. Many Motorway bridges, for example, in the UK are built on shale, or near shale, and the microbes that break down shale might have serious implications for civil engineering and civil infrastructure as well. So these studies of how microbes break down rocks, has quality of life implications as well.

15:36 - Diamond News Update - Fighting antibiotic resistance
Diamond News Update - Fighting antibiotic resistance
with Sarah Boundy, Diamond Light Source
Meera - This is the Diamond Light Source Podcast and this month we're looking back at some of the key developments and achievements at Diamond in 2010. So now let's join Sarah Boundy from Diamond's Communications Team to find out what happened at the facility towards the end of the year.
Sarah Boundy - It recently had a peper published in the journal of microbiology
which is looking at antibiotics. It reports on the molecular interaction between a protein from a common bacteria called Pseudomona Originosa, which can cause disease in animals and humans, and they're looking at that and the 2 main antibiotics to combat it.
Meera - What aspect of these antibiotics were they actually looking into?
Sarah - Well they used Diamond to solve the structure of the protein bound to the antibiotic to get a picture of how the drugs work against the bacteria. The results allow them to look at the structure and see ways that the drugs may be modified and one of the problems with Pseudomona Originosa is that it can be particularly dangerous to those who are already ill, it can cause secondary infections, and so they are looking at how they can overcome the problem of drug resistance, when the bacteria becomes resistant to the antibiotic.
Meera - and I guess knowing this, they can develop new antibiotics or  drugs that can be more effective?
drugs that can be more effective?
Sarah - Yes, from looking at the structure, they are able to look at ways they can aid the existing inhibitors and come up with new drugs that could be more effective.
Meera - The workings of antibiotics are always going to be crucial but now moving away from Life Sciences, the main thing about Diamond is that it looks into a whole range of scientific disciplines, including Engineering.
Sarah - That's right. This one is most interesting because most of the Beamlines at Diamond are looking at samples of things that are absolutely tiny. They could literally fit on a pin head, but we've had a new development on our Jeep Beamline which is a Joint Engineering and Environmental Processes beamline.
It's the only place in the UK where the internal stresses and strains of components up to 2 tonnes in weight and beyond a metre in length can be studied at the atomic scale.
Meera - So these are much bigger objects than have ever been analysed in the past at Diamond. So what kinds of objects are we talking about here - aircraft design and aircraft components?
Sarah - That's right, it's actually the external hutch of the JEEP Beamline, it can create molecular scale 3D images such as Aerospace and engineering components and the first users were actually from Rolls Royce. They were looking at a fan blade from one of their Trent 1000 engines. They were able to use JEEP to look inside the fan blade and measure all residual stresses without having to remove any of the metal and Rolls Royce routinely measure residual stress in their components and the JEEP Beamline is an alternative way for them to do this.
Meera - Now moving back down to small scale, the nano-scale in fact, you've had users on another Beamline as well?
Sarah - Yes, so we've now got 19 Beamlines in operation. Towards the end of last year we had first users on the Beamline for Advanced Dichroism Experiments, we call it BLADE for short. They were investigation multi-ferroics which are the class of materials which simultaneously exhibit magnetism and electricity.
Meera - So this obviously has to do with computing and computer chips as this is how data storage essentially works which is a crucial part of science and technology to be looking into at the moment.
Sarah - Yes, they were trying to understand the principles involved, with a view of being able to develop multi-ferroic materials which could work at room temperature. If this could be cracked, they could hold huge potential for new solutions for data storage.
Meera - Ok, so they were the first users on Beamlines that were developed last year, but you've also recently had 'First Light'.
Sarah - So this is 'First Light' on I13, our X-Ray Imaging and Coherance Beamline, and it simply means that the Beamline Team were successful in getting synchrotron light from the main machine into the Optics Hutch of their Beamline. So now they're busy finishing the beamline off and they hope to welcome first users later this year.
Meera - And what, essentially, is this Beamline? What makes it stand out and what kind of work will be done on it?
Sarah - So this one will allow researchers in a wide range of fields to create high quality 3D images of samples. So things such as engineering components, biomaterials, fossils, organic materials, and energy devices such as fuel cells, and it's a bit different for Diamond because its experimental Hutch sits 250 metres away from the energy source, whereas most of the other ones are within the main building. We're also developing it a little differently because we've entered into a collaboration with the University of Manchester.
Meera - So plenty of development and plenty of new research taking place but as well as research, another important thing that happens at Diamond is Outreach.
Sarah - That's right, throughout the year we try to run lots of events for A-Level students and people interested in a career in Science and Engineering and in particular, towards the end of last year, we had a day called 'Engineering your Future'. It was a careers day for A-level students who might like to take up a career in Engineering and they did lots of fun things like wiring up a mini RAF Pad, Programming a Space Robot to carry out manoeuvres, they had to build a prototype sturdy base for a wind turbine out at sea, so they did all kinds of things. We run that event on a yearly basis and we've got more of that kind of stuff coming up with our Particle Physics Master Class in March, as well as an Inside Diamond event especially for A-level students.
Meera - How can people keep up to date or aware of when these events are happening?
Sarah - The best thing to do is to look at our website. There's plenty of information there about public events. We've actually got the next public Inside Diamond event which is coming up on Saturday 26th March, and registration can be made on our website which is www.diamond.ac.uk.
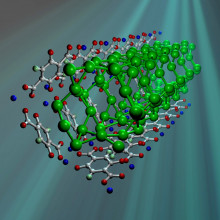
21:29 - Metal Organic Frameworks
Metal Organic Frameworks
with Neil Champness, University of Nottingham
Meera - One recent, and fast developing, area of Chemistry is the design and use of Metal-Organic Frameworks. They have potential uses in Gas Storage and Catalysis, but can also provide greater insight into the fundamental workings of chemical reactions as Nottingham University's Neil Champness explains.
Neil Champness - So metal-organic framework is a material, always a solid, which is made by reacting metals with organic molecules which will 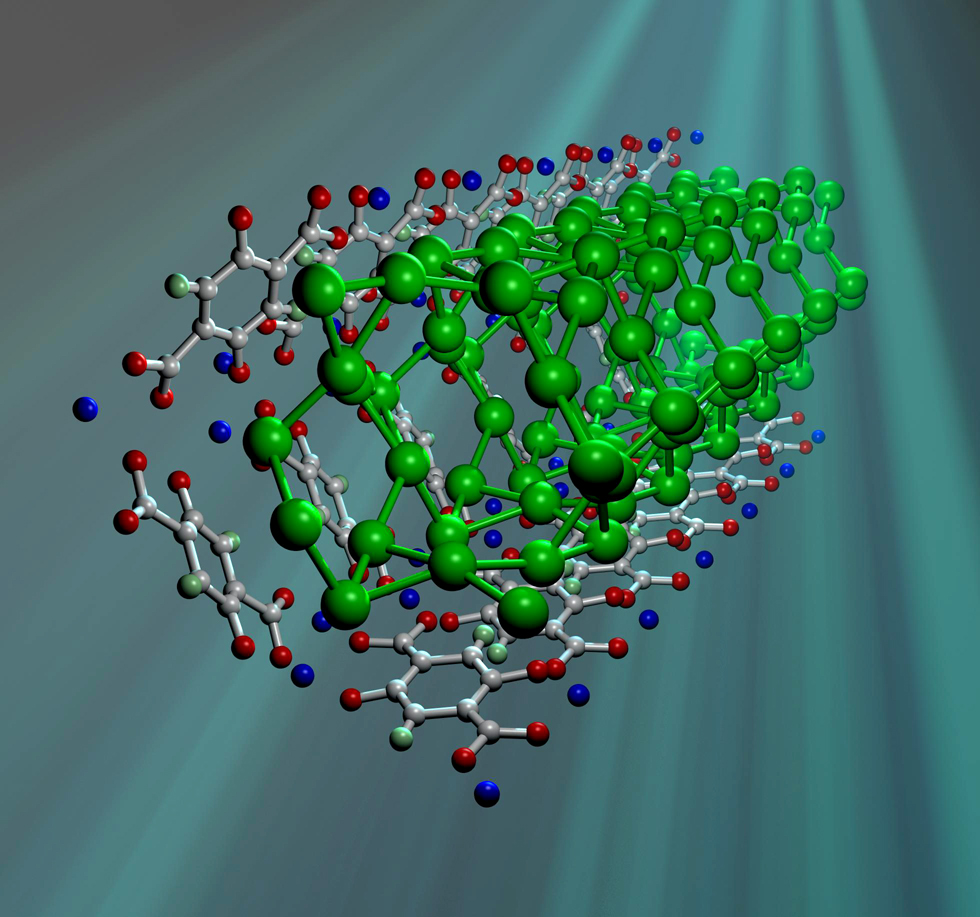 act as a bridge between different metal centres. So, you react the 2 together and you make a 3 Dimensional structure, so it will look at bit like a climbing frame. And typically they will have holes within that structure. But more importantly, beyond that, metal-organic frameworks will have this subset of materials where you can, well react metals and ligands.
act as a bridge between different metal centres. So, you react the 2 together and you make a 3 Dimensional structure, so it will look at bit like a climbing frame. And typically they will have holes within that structure. But more importantly, beyond that, metal-organic frameworks will have this subset of materials where you can, well react metals and ligands.
Meera - So, essentially it's a 3 dimensional, porous structure that you can encapsulate things into, but on a very, very small scale?
Neil - Yes, absolutely. The types of dimensions that we are talking about will be 2 nanometres and below. But that is the molecular scale.
Meera - What would you say is the general aims of these structures are and what is the purpose of them?
Neil - The beauty of metal-organic frameworks is that you can vary the metal that you use, so typically we use transition metals, things like copper and zinc, or you can use lanthanides. So there's a lot of variation there and you can have different properties that they bring with them, but also you can vary the organic molecule a lot as well. And therefore, you can have almost a limitless library of possible framework structures. A lot of the work is directed towards Gas Storage, this is a big theme, and our own research group work with Diamond in fact on developing Hydrogen Storage materials. You can use it for trapping Carbon Dioxide, is another possible direction, there is a lot of interest in developing them for pollutant separation, those types of things.
Meera - So essentially you can really design and tailor these to be specific to the kind of use or the things that you want to look into?
Neil - Absolutely, that's the great desire and in many ways that is more true for metal-organic frameworks than it is for anything else.
Meera - Tell me more about your specific use for these structures. You look into Photochemical and Photophysical reaction; reactions that really need the presence of light?
Neil - Yes. One of the interesting things in Photo-Chemistry is that you can shine light on a molecule and it becomes excited. It absorbs the light and becomes excited. And those excited states then undergo different reactions or use that energy, the energy of the excited state, in different ways. The actual properties of those excited states depends very much upon the environment in which they are sitting and also, how easily they can meet other excited molecules and react with them. What we have been doing is trying to incorporate those molecules which can be photo-excited within the metal-organic framework. You are controlling the environment of the excited state, of that excited molecule, and also you are stopping it meeting other excitable molecules, so therefore you are changing the properties of the molecule by putting it within the framework. Metal-organic frameworks are ideal for this because you can design the framework structure, as we've just been discussing.
Meera - What is the benefit then of keeping them in this rigid state and analysing what does happen to them?
Neil - So it gives you real insight into what is the nature of this photo-excited state. Probing those excited states is difficult because many of them are short lived, they can be less than a nano-second. It can react, it can undergo chemical transformations into all kinds of things, you need to understand that process, but also by putting it within the framework, we can prolong the lifetime of those excited states, we can change the actual properties in fact and actually we can probe them differently. Often, the way that you look at these excited states is by using spectroscopy. They give you lots of information, but it's not quite the same as the details that you can get from other techniques such as crystallography. This is where our work with Diamond comes in. At Diamond, we can get what are called crystal structures of single crystals of our frameworks, using the high energy x-rays that can be generated at the Diamond synchrotron, which we can't do in our own labs here. What this allows you to do is to actually get detailed information about the precise positions of the atoms within our molecules. This allows us to see what happens to the photo-excited state after it reacts.
Meera - Could you give an example of a particular reaction that you have been looking into at Diamond?
Neil - A particular case we can incorporate into our framework structure, a particular rhenium complex. Now, rhenium is a pretty unusual metal and this particular type of rhenium complex has been widely studied and is actually used, it's kind of a classic of photo-chemistry, and it has been used in all kinds of applications, studied in solutions, looked at how it interacts with DNA, used in photovoltaic applications. What we did was we incorporated this basic group within a framework, modified its properties and were then able to look at how it behaves after photo-excitation.
Meera - What would the next step be? With a really thorough understanding of the process, say for the rhenium complex and other examples and other reactions as well, what would it be hoped would be the next using this information?
Neil - In this process there are carbon monoxide molecules attached to the rhenium centre and actually on photo-excitation there is a short period of time where this carbon monoxide molecule comes away from the metal centre and there is what is called a 'Vacant Site'. Normally, and indeed in the studies we've done so far, what happens is that the carbon monoxide goes back on. What we really want to do is to prolong the lifetime of that situation where there are vacant sites on the metal and try to put different things onto it. There are 2 directions here, one is to try to put alkanes on to there - this is things like Methane, methane gas -and try to incorporate it and get a rhenium interaction and that leads to what is called 'Activation' of the alkane and make it react differently in chemical terms. But the one we'd love to do, is to try to put Xenon onto it, try to make a metal to xenon bond and actually characterise it so that we exactly where this xenon is sitting. The xenon is incredibly un-reactive. It's actually being able to identify and characterise a bond to xenon. It's almost one of the Holy Grails to chemistry.
Meera - So it would be quite an advancement in fundamental chemistry to try and get this bond.
Neil - Absolutely, it's fundamental understanding here. I mean, it gives us a far greater understanding of how these things can behave, it advances science all together. There is still a beauty in understanding how different atoms interact with each other, 'cause that's the basis of all chemistry.










Comments
Add a comment