In this week's NewsFlash, a new way to finger criminals using the trail of bacteria they leave behind, combating cancer with synthetic lethality, and how scientists have turned mosquitoes into flying vaccinators. Plus, how to create, and cure Alzheimer's disease in fruit flies.
In this episode

00:10 - Targeting cancer's Achilles' heel
Targeting cancer's Achilles' heel
Cancer is a disease caused by gene faults - both inherited from our parents and picked up during our lifetime as our DNA gets damaged. And increasingly, researchers are working out how to use these faults to our advantage to fight the disease.
This week, research from Professor Alan Ashworth and his team at The Institute of Cancer Research, funded by Cancer Research UK, has revealed another Achilles' heel in cancer cells that we can try and target. They've just published their results in the journal Cancer Cell.
 The new research is based on the concept of synthetic lethality. In our cells, we have multiple ways of doing the same thing, which help to protect us if something goes wrong. It's a bit like a pair of trousers with a belt and braces holding them up - so if the belt breaks, then the braces still work to preserve your dignity.
The new research is based on the concept of synthetic lethality. In our cells, we have multiple ways of doing the same thing, which help to protect us if something goes wrong. It's a bit like a pair of trousers with a belt and braces holding them up - so if the belt breaks, then the braces still work to preserve your dignity.
But cancer cells have a lot of faults, and in many cases they're just relying on one mechanism to do something - to use our analogy, the belt is broken, and they're relying solely on their braces. So if we can figure out what they're dependent on, and target it with drugs - to snip the braces - then that might be a good way to treat cancer.
Professor Ashworth and his team looked at cancer cells that had faults in specific genes involved in repairing damaged DNA - genes called MLH1 and MSH2. Faults in these genes have been found in a number of different cancers, including bowel cancer.
Faults in either of these genes mean that cells tend to accumulate DNA damage, helping to turn them cancerous. But other pathways in the cell - the metaphorical braces - can still compensate for the lack of MLH1 or MSH2, so the cancer grows and spreads.
Here's where it gets clever. The researchers realised that molecules called DNA polymerases - which help to copy DNA when cells divide - were also repairing DNA, and compensating for the lack of MLH1 and MSH2.
The researchers used a technique called RNAi to knock out certain DNA polymerases in cancer cells lacking either MLH1 or MSH2. And they found that knocking out a polymerase called POLB could kill cells lacking MSH2, while targeting another polymerase called POLG could kill cells without MLH1.
We already know that this kind of approach works to treat cancer - we're starting to see very promising results from early clinical trials of new cancer drugs called PARP inhibitors, which work along the same principles, but targeting different pathways in the cell.
At the moment, the technique the researchers used to block POLB or POLG isn't easily transferable to patients (as we'll hear in my second news story). So scientists now need to find small chemicals that could be developed into drugs that would have the same effect. Although that's still some way away, this is a promising approach.
This research also highlights that we need to focus on the specific faulty pathways in cancer cells, rather than just the overall type of cancer. For example, faults in MLH1 and MSH2 are found in a subset of several different types of cancer, so they might all respond to polymerase-blocking drugs. We're starting to move away from thinking about treating "bowel cancer", or "breast cancer" etc., and towards treating "MLH1-deficient cancer cells", targeting the genetic faults that are specific to an individual patient.

03:44 - Bacterial fingerprint: a new way to catch crooks
Bacterial fingerprint: a new way to catch crooks
Scientists have found a new way to track down criminals - using the unique collections of bacteria they leave behind on things they touch.
The human body is plastered in a highly diverse community of bacteria that form a microbial fingerprint unique to each individual. So much so in fact that, even after a scrub with soap, after a few hours the same bugs are back on the average pair of hands.
 Now researchers have shown that this bacterial badge could even be used forensically. Writing in the journal PNAS, University of Boulder Colorado scientist Noah Fierer and his colleagues took swabs from computer mice and keyboards and were able to produce a genetic profile of the bacteria present and then pick out correctly - from more than 250 possible pairs of hands - the computer owner.
Now researchers have shown that this bacterial badge could even be used forensically. Writing in the journal PNAS, University of Boulder Colorado scientist Noah Fierer and his colleagues took swabs from computer mice and keyboards and were able to produce a genetic profile of the bacteria present and then pick out correctly - from more than 250 possible pairs of hands - the computer owner.
Although they caution that further investigation and validation of the technique will be necessary to establish its forensic credentials, the researchers think that it could be a powerful adjunct to existing methods. "This approach might represent a valuable alternative to more standard techniques," they point out.
"Given the abundance of bacterial cells on the skin surface and on shed epidermal cells, it may be easier to recover bacterial DNA than human DNA from touched surfaces."
06:43 - Gene switch technique shows promise in clinical trials
Gene switch technique shows promise in clinical trials
RNAi is a powerful technique used to switch off DNA polymerases in cancer cells grown in the lab. The technique has been shown to switch of genes in cells, and in small organisms like worms and flies. But until now we haven't seen that it can work in humans - it's really been a technique restricted to lab studies. Now research from Mark Davis and his colleagues in the US, published in the journal Nature, shows the first inklings that we might be able to get RNAi to work in patients.
RNAi stands for RNA interference. When a gene is switched on, it produces a little message in the form of RNA, a chemical that's similar to DNA. This RNA message then goes into the cell, where it's 'read' by the cell's protein 'factories', and the appropriate protein is produced. It's a bit like copying a recipe out onto a piece of paper, then taking the paper into the kitchen to bake a cake, rather than the whole recipe book.
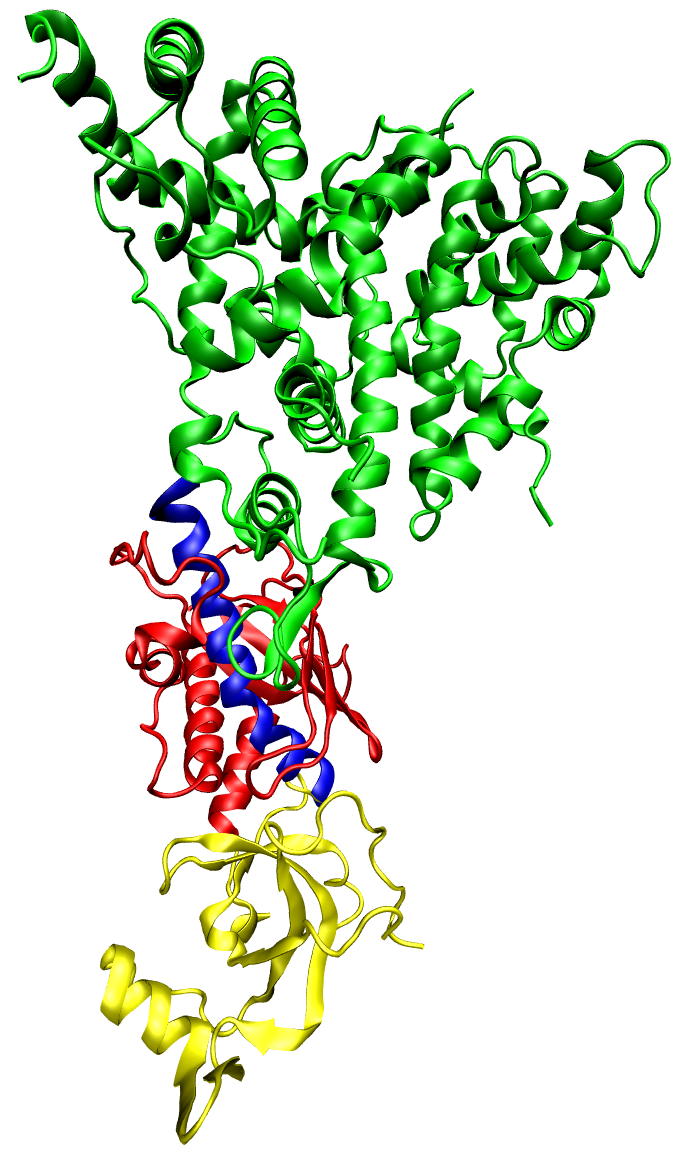 |
| One molecule of the Dicer-homolog protein. Dicer is an RNase that cleaves long double-stranded RNA molecules into short interfering RNAs (siRNAs) as a first step in the RNA interference response, and also initiates the formation of the RNA-induced silencing complex (RISC). © Opabinia regalis |
Researchers discovered that short reverse stretches of RNA could effectively silence RNA messages, and individually switch specific genes off. It's an incredibly powerful technique that helps researchers to switch genes on and off in the lab, as we heard in my earlier story, but it's not been clear whether it actually works in larger animals, or in humans.
In this case, the researchers were running a small-scale clinical trial to test RNAi in patients with cancer. They were using tiny nanoparticles to deliver the RNA to the tumours - for some reason that we don't understand, nanoparticles seem to be attracted to tumours.
The scientists discovered that the nanoparticles had effectively delivered their RNA payload to the cancer cells, and that the therapy was working as expected - the specific gene they were targeting, called RRM2, was getting switched off.
This is a very small-scale, early trial and in this paper, the researchers only present data from three patients, all of whom had melanoma skin cancer. And we don't know whether the RNAi actually helped to treat their cancer. Still, it's an impressive demonstration that the RNAi technique can work in humans, and bodes well for future research in this area.
09:28 - Turning mosquitoes into flying vaccines
Turning mosquitoes into flying vaccines
Scientists have found a way to use mosquitoes as mobile vaccinating machines!
Mosquitoes are ranked as the world's most dangerous animal because the diseases they transmit, including malaria, dengue and yellow fever, kill several million people per year. But now scientists having turned the tables and are working on ways to reprogramme mosquitoes to convert them from agents of illness into low cost flying vaccines!
 The work is based on the premise that when mosquitoes take a blood meal they first inject saliva, containing anticoagulants and immune-evading agents, around the blood vessel puncture site. This is what provokes the itchy inflammatory aftermath but is also responsible for transmitting infectious agents, which the mosquito regurgitates into the wound when it feeds.
The work is based on the premise that when mosquitoes take a blood meal they first inject saliva, containing anticoagulants and immune-evading agents, around the blood vessel puncture site. This is what provokes the itchy inflammatory aftermath but is also responsible for transmitting infectious agents, which the mosquito regurgitates into the wound when it feeds.
But Shigeto Yoshida, from Jichi Medical University in Japan, reasoned that it ought to be possible to exploit this unpleasant aspect of the insect's behaviour in a beneficial way. They set about genetically modifying Anopheles mosquitoes to make them produce in their saliva a protein called SP15, which is critical to the spread of another major disease-causing parasite, called leishmania.
Mice bitten repeatedly by these modified mosquitoes, the team found, developed antibodies to SP15, which other researchers have shown can protect against leishmania transmission.
"Following bites, protective immune responses are induced, just like conventional vaccination but with no pain and no cost," says Yoshida. "What's more, continuous exposure to bites will maintain high levels of protective immunity, through natural boosting, for a lifetime."
Now the team are testing whether mosquito-vaccinated mice really can be protected from leishmania infection; the same SP15 protein has been successfully tested as an experimental vaccine previously so the odds are that it should work.
But whether this flying vaccinator technology will take off in general is another matter. Some may feel slightly stung, however, by the idea of a natural and uncontrolled vaccination system delivering unmetred drug dosages and boosters indiscriminately, a sentiment prempted by the scientists themselves in their paper, published in Insect Molecular Biology.
"The concept of a "flying vaccinator" transgenic mosquito is not likely to be a practicable method of disease control, because "flying vaccinator" is an unacceptable way to deliver vaccine without issues of dosage and informed consent against current vaccine programs. These difficulties are more complicated by the issues of public acceptance to release of transgenic mosquitoes."

11:06 - Preventing Alzheimers Disease in Fruit Flies
Preventing Alzheimers Disease in Fruit Flies
Dr Leila Luheshi, University of Cambridge
Kat - Also in the news this week, scientists have discovered a new molecule that can block the build up in the brain of beta amyloid. This is the toxic protein that causes Alzheimer's disease. But rather than working on human subjects, Dr. Leila Luheshi who's based at the Genetics Department here at the University of Cambridge made a discovery using fruit flies that were genetically engineered to develop the insect equivalent of Alzheimer's disease. Meera Senthilingam went along to meet her to find out more.
Leila - So it turns out that if you would like to study Alzheimers disease really you  need to have some sort of model of the disease in your own organism other than humans which you can try out new treatments on, and you can test new and rather crazy ideas. It turns out Drosophila are very good for this. They have quite complicated brains, a lot of the same genes that there are in humans and also, they have very short lifespans, so we can test lots of different treatments very rapidly in them. It turns out that we can give them something like Alzheimer's disease and then go out, and try and find different ways of curing it.
need to have some sort of model of the disease in your own organism other than humans which you can try out new treatments on, and you can test new and rather crazy ideas. It turns out Drosophila are very good for this. They have quite complicated brains, a lot of the same genes that there are in humans and also, they have very short lifespans, so we can test lots of different treatments very rapidly in them. It turns out that we can give them something like Alzheimer's disease and then go out, and try and find different ways of curing it.
Meera - So how do you set about giving them Alzheimer's disease?
Liela - So we take the gene that encodes for the protein that seems to be important in causing Alzheimer's disease in humans, and we introduce it into our fruit flies. Then, when that gene is expressed in the brain this protein, which is called amyloid, clumps together in the brains of our fruit flies, just like it does in the brains of humans with Alzheimer's disease. Then the fruit fly's brain starts to degenerate, and we can see that they can't move properly, they die much sooner, and their memory is impaired as well, just like people with Alzheimer's disease.
Meera - So, is it the case where all people that have the gene will develop Alzheimer's?
Leila - No. Actually, the amyloid protein that damages the brain in Alzheimer's disease is present in all of us. But under normal circumstances, it's present in small enough quantities, and in a form which doesn't seem to cause us any problems. In fact, what seems to happen in most patients with Alzheimer's disease that you produce a little bit more maybe of a slightly stickier than usual version of this protein. That starts to stick together in the brain, and then that starts the damage of the neurons.
Meera - So having given these flies Alzheimer's, you've set about finding a new molecule that should hopefully treat Alzheimer's in these flies, and hopefully in humans?
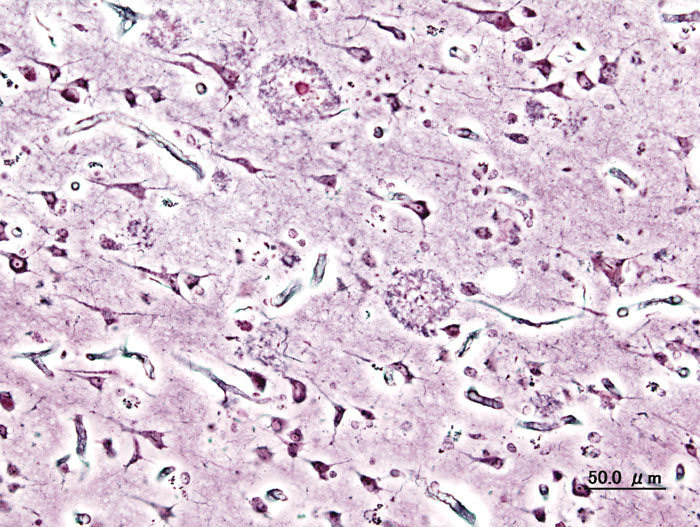 Leila - Yes. So what we thought was, well, if we know there's a protein, this protein called amyloid, which clumps together in the brains, then if we can prevent this protein from clumping together, then hopefully the cells in the brain won't die anymore. And so, what our colleagues in Sweden did was they designed a new molecule which is called an affibody which binds to the amyloid protein that is important in Alzheimer's disease, and prevents it from clumping together. They first tested this in a test tube and they found this was very effective, and then they came to us and said, "Well, can you test this in your fruit fly model of Alzheimer's disease?" When we put this new molecule, this affibody, into the brains of our fruit flies with Alzheimer's, we found that essentially, the fruit flies were cured of the Alzheimer's disease, and in fact the protein which clumps together, this amyloid protein, was now completely cleared from the brain.
Leila - Yes. So what we thought was, well, if we know there's a protein, this protein called amyloid, which clumps together in the brains, then if we can prevent this protein from clumping together, then hopefully the cells in the brain won't die anymore. And so, what our colleagues in Sweden did was they designed a new molecule which is called an affibody which binds to the amyloid protein that is important in Alzheimer's disease, and prevents it from clumping together. They first tested this in a test tube and they found this was very effective, and then they came to us and said, "Well, can you test this in your fruit fly model of Alzheimer's disease?" When we put this new molecule, this affibody, into the brains of our fruit flies with Alzheimer's, we found that essentially, the fruit flies were cured of the Alzheimer's disease, and in fact the protein which clumps together, this amyloid protein, was now completely cleared from the brain.
Meera - How was this affibody created?
Leila - It's based on a protein that is found naturally inside bacteria. What our colleagues in Sweden did was they made mutations in this protein, then they screened thousands, in fact, millions of different mutations in this protein until they found a mutant form of this protein that bound very specifically to the amyloid protein that's important in Alzheimer's disease.
Meera - And how did you set about putting this affibody into the Drosophila then as well?
Leila - So we essentially did the same thing that we did to make our model of Alzheimer's disease in the first place. We made a gene for this affibody protein and we put the gene into the brain of our fruit fly, and then that gene expresses the affibody protein, so then we have one fruit fly that expresses the affibody, one fruit fly that has Alzheimer's disease, and when we breed those two fruit flies together their offspring, their children, have both proteins. They have the Alzheimer's related protein and they also have the affibody, and then we could see that when you have those two proteins together, the flies didn't get Alzheimer's disease anymore.
Meera - So, its' not the case where the fly had Alzheimer's and then you gave it this protein, and it actually treated the Alzheimer's that it had previously?
Leila - No. So right now, we have to do it with both proteins there from the beginning together so we prevent the amyloid protein that's important in Alzheimer's disease from ever clumping together. But what we would like to do in the future is to make our flies so that they actually develop Alzheimer's disease, so we let the protein stick together in the brains of the flies, and then put in affibody protein afterwards, and see if we can either help the symptoms or reverse them. But we haven't done that experiment yet. First of all, we have to go through some more tests in slightly more complicated models of Alzheimer's disease in our fruit fly to see if this kind of protein - for instance, if we inject it into the bloodstream, can even get into the brain? It's got to be able to get to the brain, really, to have any effect. So, we're still somewhere off seeing whether this kind of therapy would ever work in humans, but at least in principle, we know that if we can clear this amyloid protein from the brain that it might help the Alzheimer's patients. Assuming we get to them early enough in their disease.
Kat - That was Leila Luheshi from the University of Cambridge, talking to Meera Senthilingam there about a work she's just published in the journal PLoS Biology.
Related Content
- Previous Are all the calories in food absorbed?
- Next The Science of Farming










Comments
Add a comment