In this NewsFlash, we hear how scientists have created Malaria-free mosquitoes, how hostile bacteria in the Arctic can be tamed to create vaccines and how Gobies can fight their way through slime in our oceans. Plus, we discover why a transfer from high-heeled shoes to flat ones is so uncomfortable!
In this episode
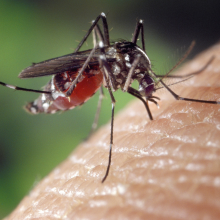
00:12 - Making Malaria-proof Mosquitoes
Making Malaria-proof Mosquitoes
There's been a study published this week in the journal PLoS Pathogens where scientists attempted and succeeded in creating a malaria-proof mosquito. And this is really exciting, because malaria kills around a million people every year, many of them children. It's caused by a parasite called Plasmodium, that infects and replicates inside female Anopheles mosquitoes, before being passed on to the next person. The incubation period in the mosquito is about 10 to 14 days.
So the group from the University of Arizona, led by Michael Riehle, genetically engineered Anopheles mosquitoes to increase the expression of a particular gene in the gut for a protein called AKT. This protein is what is known as 'highly conserved', meaning it is found in many animal groups - both vertebrates and invertebrates - to do the same thing - in this case involved in immune response and lifespan.
The researchers compared mosquito siblings with and without the engineered gene by feeding both groups an artificial blood meal containing Plasmodium. Ten days later, the team checked the guts of the mosquitoes to see if any of the Plasmodium had successfully formed oocysts, the next stage in their development.
Up to 99% of mosquitoes that were heterozygous for the engineered gene (so with one copy) were found to be parasite-free, and all of the homozygous mosquitoes (with two copies of the gene) were parasite free. There was also a 20% reduction in lifespan. This is an important point, because it takes time for the Plasmodium to develop in the mosquito to become infectious, so shortening the lifespan of the vector is an efficient way of preventing infection.
So what does this mean for the global fight against malaria? Well, Riehle's eventual aim is that engineered mosquitoes would be released, and breed with wild populations, introducing the engineered gene to reduce Plasmodium infection and shorten mosquito lifespan. This is obviously still quite a way off, but it's a really encouraging first step.
03:07 - Killer bugs tamed with a dose of the Arctic
Killer bugs tamed with a dose of the Arctic
Scientists have found a way to tame hostile bacteria and turn them into docile vaccines, by replacing key genes with those from the bugs Arctic counterparts!
Writing in PNAS, University of Victoria, Canada, researcher Barry Duplantis and his colleagues reasoned that some of the essential genes carried by bacteria that have evolved over billions of years to live in very cold climes will most likely be optimised to those temperatures. So exchanging some of these genes for their equivalents carried by warmth-favouring pathogenic bacteria could produce a temperature-intolerant, disabled bug that could function as an ideal vaccine.
 To find out, the team tested out nine genes known to be critical to survival in all known bacterium; the gene sequences were first "cloned" from bacteria that flourish in the Arctic and then inserted into the chromosomes of Salmonella and Francisella bacteria and also a Mycobacterial species related to TB, replacing the native gene.
To find out, the team tested out nine genes known to be critical to survival in all known bacterium; the gene sequences were first "cloned" from bacteria that flourish in the Arctic and then inserted into the chromosomes of Salmonella and Francisella bacteria and also a Mycobacterial species related to TB, replacing the native gene.
The modified bugs grew unhindered at low temperatures, but grown at higher temperatures the bacteria suffered stunted growth. Injected into a cool spot on the bodies of experimental rodents, the modified bugs, which are usually fatal, instead provoked a robust immune response allowing the animals to fend off a lethal dose of unmodified bacteria which were administered a few weeks later.
This suggests that the technique could be used to produce a host of safe, temperature-sensitive live vaccines capable of protecting people against significant pathogens including Salmonella and TB.

06:32 - Gobies fight against the 'Rise of Slime'
Gobies fight against the 'Rise of Slime'
Well it's been rare in recent times to find a positive story about life in the oceans, but this week, researchers from the University of Bern in Norway have shown that there is a species that is unexpectedly thriving.
The study, published in Science is about a fish called the bearded goby, Sufflogobius bibarbatus, and it seems to be an unexpected winner in an ecosystem that had been severely damaged - the Benguela upwelling system off the coast of Namibia. In the 1960s, aggressive overfishing, combined with environmental changes in the area, led to a collapse of the sardine population and a shift in the structure of the ecosystem. There was a massive rise in jellyfish numbers, and the sea bed was covered in an anoxic, sulphurous mud layer.
But the team led by Anne Utne-Palm found that the bearded gobies seemed to be thriving. They showed through a series of very elegant behavioural and physiological experiments that they were able to hide in the low oxygen mud on the sea floor from predators, and to feed on the bacteria in the mud. They turned down their metabolism to survive in the low oxygen sludge, ascending up into the more oxygen rich layers of the water under the safety of darkness to digest their food. They also munch away on the jellyfish, returning what were previously thought to be 'dead end' resources back to the food chain by then becoming prey themselves to larger fish.
The researchers see the goby population as a kind of stabilising factor, able to buffer the local fisheries against changes in environmental conditions in the future - if oxygen levels go down, goby predator numbers will go down, the goby population will increase, and will allow the larger fish populations to recover once conditions become more favourable.
It's really quite a promising finding, because it shows that it is possible for an apparently collapsed marine ecosystem to recover.

08:24 - Why high-heels are habit-forming
Why high-heels are habit-forming
High-heel tottering fashion slaves often complain that stepping down to normal footwear is extremely uncomfortable following the elevating effects of a six-inch height boost. Now scientists know why.
 In a paper in the Journal of Experimental Biology, Manchester Metropolitan University scientist Marco Narici explains how he used newspaper ads to recruit over 80 women with at least a 2 year, 5 cm high-heel history, 11 of whom reported discomfort when walking without their heels.
In a paper in the Journal of Experimental Biology, Manchester Metropolitan University scientist Marco Narici explains how he used newspaper ads to recruit over 80 women with at least a 2 year, 5 cm high-heel history, 11 of whom reported discomfort when walking without their heels.
Ultrasound measurements of the calf muscle lengths amongst these women showed significant shortening - by 13% - of the muscle fibres compared with non-heel wearing controls.
At the same time, MRI scans showed that the Achilles tendons of the two groups were the same lengths, indicating that the tendons of the heel wearers had not lengthened to compensate for the shorter muscle length, although the heel wearers tendons were nonetheless thicker and stiffer than those of the controls.
Consequently, walking on flat-feet becomes uncomfortable for these people because the tendon cannot stretch sufficiently to compensate for the altered posture.










Comments
Add a comment