On this week's News Flash, we find out what Margaret Thatcher's face can tell us how monkeys recognize each other, what sharks have in common with serial killers and why dolphins are a bit like jet fighters. Plus, we look back to this Week in Science History...
In this episode

Margaret Thatcher and the Evolution of Face Recognition
Research from Emory University this week shows that we, and our ancestors, have been recognising faces in much the same way for 30 million years or more.
 By watching how Rhesus Macaques respond to a well known optical illusion called the Thatcher effect, professor Robert Hampton and colleagues were able to determine that these monkeys, like us, recognise faces not because we compare them, mentally, to a particular idealised image of a face, but because of the relationship between the different features.
By watching how Rhesus Macaques respond to a well known optical illusion called the Thatcher effect, professor Robert Hampton and colleagues were able to determine that these monkeys, like us, recognise faces not because we compare them, mentally, to a particular idealised image of a face, but because of the relationship between the different features.
The Thatcher effect is an optical illusion that many of you will have seen before - it's created by taking a picture of a face, cutting out the eyes and mouth, then turning them upside down. When you see this image completely upside down (so the eyes and mouth are actually the right way up, but the whole head is rotated), it's hard to see anything wrong with it. It's only when the head is the right way up (meaning the eyes and lips are upside down) that it looks grotesque, and clearly not right.
It's thought that this effect works in humans because we perceive faces as a configuration, rather than an image - this makes us more sensitive to differences in configuration, and makes us more able to recognise individuals.
If monkeys see faces in the same way, then the Thatcher effect should work on them as well.
To test this, the researchers showed some Rhesus macaques some pictures of other Rhesus monkeys' faces, some normal, some upside down and some Thatcherised, and monitored how much attention they paid to each set of images. Once the monkeys had habituated to the photos, they showed no more interest in upside down Thatcherised images than they did normal faces. But when the Thatcherised images were shown the right way up, they paid significantly more attention to them.
This suggests that the monkeys detect faces in the same way as us, as a configuration, and so that this may have evolved at least 30 million years ago, in our common ancestor with the macaques.
Previous studies have shown Thatcherised images to have no effect on pigeons or baboons, which may seem to contradict these results. However, in the baboon study, published in the American Journal of Primatology in 2008, the animals were extensively trained with a small set of images, and so may not indicate the same spontaneous effect as seen in humans.
"This direct evidence of configural face perception in monkeys, collected under testing conditions that closely parallel those used with humans, indicates that perceptual mechanisms for individual recognition have been conserved through primate cognitive evolution," said Professor Hampton.

Serial killer sharks
Scientists have shed light on how those iconic ocean predators, great white sharks, go about catching their prey. It turns out they have something in common with human serial killers.
 Great white sharks lurk in spots that don't just guarantee them the best chance of encountering seals but they try and minimise the chances that seals will spot them before they launch an attack. They also take into account where other sharks are.
Great white sharks lurk in spots that don't just guarantee them the best chance of encountering seals but they try and minimise the chances that seals will spot them before they launch an attack. They also take into account where other sharks are.
The shark research team, lead by PhD student Neil Hammerschlag from the University of Miami in the US, took advantage of the way great white sharks in False Bay in South Africa attack their favourite food, cape seals. They target young solitary seals at the sea surface when light levels are low. They lurk at the sea floor, stalking the seals before ambushing them in a sudden vertical rush that propels both shark and seal out of the water. This strategy gives the sharks the best chance sneaking up on the seals and delivering a single fatal bite.
The researchers recorded 340 shark attacks which can be seen from quite a distance across the water and pinpointed the location using GPS units.
To analyse the data the researchers teamed up with Kim Rossmo, a criminal justice expert, who developed computer software to study the geographic patterns of human crimes including terrorist attacks and serial killers - a process called geographic profiling. By looking at where serial crimes take place, the software narrows down an "anchor point" for the criminal, usually their home or place of work. So Hammerschlag and the shark team plugged their hunting data into the same software.
The sharks attacked in non-random patterns and, like human criminals, had their own anchor points. The larger sharks tended to hang out in areas about 100 metres from areas where seals congregate, a vantage point that meant they didn't scare the sea colony, but from where they could easily pick off any seals that stray from the main group.
Smaller, younger sharks hunted over a wider area and were less successful in killing seals than larger sharks. This suggests that as they get older, sharks gain experience and learn to refine their search patterns, concentrating on areas where they are most successful. And it could be that the larger sharks aggressively keep out smaller sharks from the best hunting spots.
Unveiling some of the mystery behind these incredible predators is not only providing fascinating insights into how they successfully catch prey, but perhaps it will also help us understand more about areas where people would do best to avoid, in case they are mistaken for a seal, hanging around on the sea surface.

09:34 - In-Flight Mind Reading
In-Flight Mind Reading
In a first study of its kind, researchers have recorded what's going on in a pigeon's brain during flight...
Writing in the journal Current Biology, researchers at the University of Zurich wanted to know if familiar landmarks could be associated with changes in brain activity.
The exact methods that homing pigeons use to find their way home are still unclear, though there is some evidence that they rely on their sense of smell, the position of the Sun, the Earth's magnetic field and familiar landmarks. This versatility makes them very difficult to study, because even if you're tracking their flight with GPS, it's hard to say what method they rely on at any particular part of the journey.
By fitting pigeons with a very small electroencephalogram, or EEG, which records electrical activity in the brain, and then combining this data with GPS data for flight paths, the researchers we're able to map brain activity onto geographical areas of interest. The pigeons were monitored as they flew from a release sight back to their loft.
They found 2 distinct levels of brain activity - a middle frequency response, which happened when the pigeons paid visual attention to something; and high-frequency activity which seemed to correspond to familiar objects or when additional cognitive processing is going on.
These results means that we can use the record of brain activity to identify areas of importance for pigeon navigation, that we never could have determined from GPS data alone. This gives us real insight into how birds navigate in the real world.
Interestingly, there were a few bits of the journey with unexplained brain activity - places so close to the loft that they weren't needed for navigation. The researchers called this a 'riddle' and one they only solved when they went to visit theses sights - one was a farm and the other a barn. Both contained colonies of feral pigeons, which clearly caught the study pigeons' attention!

Flying dolphins
Scientists have uncovered the secret behind some dolphin's amazing aquatic acrobatics: they have flippers that work in the same way as delta wing, those characteristic triangular-shaped wings on fighter jet planes, or that feat of 20th century engineering, Concord. So, dolphins can literally fly through the water.
 The research team, led by Laurens Howle and Paul Weber from Duke University, took flippers from 7 species of dead dolphins and whales, either from museum specimens or animals that were stranded on beaches. They produced detailed 3D pictures of the flippers by putting them into a computer tomography, or CT, scanner. Based on these scans, the team then made accurate models of the flippers and then placed them in a flow tunnel with water rushing through it.
The research team, led by Laurens Howle and Paul Weber from Duke University, took flippers from 7 species of dead dolphins and whales, either from museum specimens or animals that were stranded on beaches. They produced detailed 3D pictures of the flippers by putting them into a computer tomography, or CT, scanner. Based on these scans, the team then made accurate models of the flippers and then placed them in a flow tunnel with water rushing through it.
By mounting the model flippers on a set of special weighing scales the team measured two forces, the lift and drag, generated by each flipper at different angles of attack. The key to how wings and flippers work is their tear-shaped cross section. As air - or water - flows over the wing, it has further to travel over the top than across the bottom of the wing, so it speeds up and that results in a lift, pushing the wing upwards. To be stable in the water, the flipper needs to generate enough lift to overcome the drag caused by friction with water, which tries to force the fin backwards.
The different shaped flippers from different performed differently with some species doing better than others. The familiar bottle nosed dolphins generated greatest amount of lift, while the least efficient fins belonged to the harbour porpoise and Atlantic white-sided dolphin.
Some of the dolphins have flippers that act in a similar way to the triangular swept-back wings of fighter jets. They create additional lift with a "leading edge vortex", a swirl of air currents over the front edge of the wing.
This all goes to help explain how some dolphins can swim incredibly quickly, up to 20 miles an hour. These are speeds that until now were thought theoretically impossible because water creates too much friction and drag for these marine mammals to keep swimming against. Now we have a better idea how they do it.
17:25 - This week in science history - The birth of Hans Spemann
This week in science history - The birth of Hans Spemann
Sarah Castor-Perry
This Week in Science History saw, in 1869, the birth of Hans Spemann, a German embryologist who won a Nobel Prize for his work on embryonic development and discovery of 'organiser' areas in the embryo.
Spemann was born in Stuttgart to a publisher and his wife. After attending school and doing a year's military service, at the age of 22 he attended the University of Heidelberg to study medicine. Two years later he moved to the Zoological Institute of Wurtzburg, where he began his career in examining cell lineages, heredity and embryo formation.
In the first few years of the 20th century, as a Professor of Zoology at the Zoological Institute in Freiberg, Spemann's work began to focus on the formation of the embryo and the groups of cells in the embryo that would become particular tissues in the adult, and how to extract and study them.
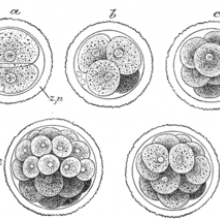
 In vertebrates, after an egg has been fertilised, the newly formed embryo begins to divide, until it forms a ball of cells. As these cells all migrate around the ball they form what is known as the 'gastrula' stage, with chemicals released at different parts of the embryo telling the cells where to go and what to become - muscles, nerve cells and so on.
In vertebrates, after an egg has been fertilised, the newly formed embryo begins to divide, until it forms a ball of cells. As these cells all migrate around the ball they form what is known as the 'gastrula' stage, with chemicals released at different parts of the embryo telling the cells where to go and what to become - muscles, nerve cells and so on.
The part of the embryo that Spemann worked on is known as the dorsal lip.
A group of cells on the dorsal lip (now known as Spemann's organiser, after Spemann's discovery) causes the formation of a structure called the notochord. This tells other cells around it to form the nerve cord (i.e. the spinal cord), and groups of cells called somites that will become muscles, parts of the vertebral column and the skin. So essentially this organiser determines which part of the embryo will become the 'back' or the dorsal side of the developed animal, and tells the cells around it what structures and tissues to form, a process known as 'dorsalisation'.
 In order to prove that it was indeed this area of the embryo that was causing the 'dorsalisation', Spemann and his PhD student Hilde Mangold transplanted a group of cells from the dorsal lip onto the opposite side of a second 'host' embryo. They used different species of salamander as the donor and the host, chosen as their colouration was different and the cells from the donor and the host could be told apart. What they found was extraordinary. Where they had implanted cells from the donor's dorsal lip, a second notochord formed, which told the host's cells to form a second spinal column and set of somites on the opposite side to its original ones - basically forming a Siamese salamander, with two spinal columns, two heads and two tails, but sharing a single 'belly'. This proved that something in that group of cells was responsible for organising the surrounding cells to form dorsal structures like the nerve cord and the somites, hence why Spemann termed the group an 'organiser'.
In order to prove that it was indeed this area of the embryo that was causing the 'dorsalisation', Spemann and his PhD student Hilde Mangold transplanted a group of cells from the dorsal lip onto the opposite side of a second 'host' embryo. They used different species of salamander as the donor and the host, chosen as their colouration was different and the cells from the donor and the host could be told apart. What they found was extraordinary. Where they had implanted cells from the donor's dorsal lip, a second notochord formed, which told the host's cells to form a second spinal column and set of somites on the opposite side to its original ones - basically forming a Siamese salamander, with two spinal columns, two heads and two tails, but sharing a single 'belly'. This proved that something in that group of cells was responsible for organising the surrounding cells to form dorsal structures like the nerve cord and the somites, hence why Spemann termed the group an 'organiser'.
This work took Spemann and Mangold the best part of 10 years to complete and it was published in full in late 1924. Tragically Hilde did not live to see the publication as she was killed in a domestic explosion in September of the same year. The work they had completed together led to Spemann receiving the Nobel Prize for Physiology or Medicine in 1935.
Spemann continued to study different organisers, as well as pioneering techniques that were the first steps towards cloning, right up until his death from heart failure in 1941. His discoveries led on to further investigations into 'organisers'. For example, we now know the exact chemicals that are released by Spemann's organiser that cause nerve and somite formation - chemicals with names like noggin, chordin and follistatin.
 All these chemicals are, evolutionarily speaking, pretty ancient. Similar versions are found in all vertebrates and in arthropods such as fruit flies. The ability of cells in an early embryo to know whether they should form 'back' or 'front' structures & tissues is clearly essential for the development of an animal and so the chemical mechanisms behind it are thought to have evolved in our common ancestor with arthropods over 500 million years ago.
All these chemicals are, evolutionarily speaking, pretty ancient. Similar versions are found in all vertebrates and in arthropods such as fruit flies. The ability of cells in an early embryo to know whether they should form 'back' or 'front' structures & tissues is clearly essential for the development of an animal and so the chemical mechanisms behind it are thought to have evolved in our common ancestor with arthropods over 500 million years ago.
Spemann's work throughout his life broke new ground and has been fundamental to our understanding of how dorsal structures of an embryo are formed. The Nobel Prize was a fitting reward for years of hard work into embryonic development, and it is tragic that his student and colleague Hilde Mangold, who helped him complete the work, did not live to see their success.
Related Content
- Previous Scrapping a Banger
- Next Driving into the Future










Comments
Add a comment