Spores on Smoke Rings and Pain Free Vaccines
In this NewsFlash, we hear how Sphagnum moss spores travel on smoke rings, explore a new way to capture carbon, discover pain free vaccine patches and better solid batteries. Plus, the microbicidal gel that could block HIV.
In this episode

00:19 - Spores Travel on Mushroom Clouds
Spores Travel on Mushroom Clouds
Moss spores get extra lift from the same process that generates smoke rings and mushroom clouds, according to new research published in the journal Science this week.
Dwight Whitaker at Pomona College in California and Joan Edwards at Williams College, Massachusetts, realised that the spores of Sphagnum moss couldn't possibly reach as high as they do by ballistic propulsion alone, and set out to find the moss' trick.
 |
| Habit shot of the low-growing Sphagnum moss showing reproductive heads with capsules raised above the mat by pseudopodia. Some capsules are round and not yet exploded, and some have recently exploded (cylindrical). Sphagnum acts like a sponge - holding water by capillary action between its leaves and in specialized water-holding cells. It acidifies the water creating anoxic acidic areas where decomposition is slow. Over the millennia, Sphagnum has sequestered more carbon and any other plant genus - thus it plays a critical role in the global carbon cycle. © Image courtesy of Joan Edwards |
Sphagnum moss is thought to store more carbon than any other plant genus, and covers around 1% of the Earth's surface. In order to distribute its spores, it fires them upwards into a turbulent patch of air, where they can be picked up and transported on eddies and breezes. To take advantage of the breeze, the spores have to be very light, and have a terminal velocity of just 5 millimetres per second. This low terminal velocity creates a problem when getting through a layer of still air directly above the moss, where the spores will rapidly decelerate.
The spores develop in the top half of a 2mm spherical capsule, the bottom half of which is hollow. Each capsule contains from 20,000 to 250,000 spores. When the conditions are right, these capsules dehydrate and become cylindrical - in the process vastly increasing the air pressure inside. Eventually, the capsule fails, and the internal pressure fires both the spore, and a puff of air, upwards.
This puff of air creates a vortex - a self sustaining ring of rising air like a smoke ring or mushroom cloud - which is strong enough to carry the spore far further than it would otherwise go, and up high enough to catch the turbulent air and be carried away. High speed imaging confirmed their hypothesis.
We know of a few species that take advantages of vortices like this - bees and jellyfish for example - but this is the very first example of vortex use in plants.
 |
| Sequential still frames from a video filmed at 10,000fps. Each frame is 1/10,000 of a second. The mushroom cloud with a trailing wake is clearly visible. © Image courtesy of Clara Hard, Joan Edwards and Dwight Whitaker |

02:24 - Electrolysing carbon-dioxide
Electrolysing carbon-dioxide
Carbon capture is often suggested as a way to reduce carbon emissions, so far it has normally just meant collecting carbon-dioxide and hiding it somewhere - normally underground. But the ideal solution would be some way of converting the carbon-dioxide back to carbon, the natural way of doing this is of course to harvest sunlight with plants and then some of this biomass is sequestered. But the highest efficiency plants only convert a few percent of the sun's energy into biomass, and if you want to use it as fuel, the best conversion efficiency is less than 0.5%.
 A more radical rethinking of this problem has been to use solar electricity to electrolyse - or split - the carbon-dioxide into carbon and oxygen. Whilst in comparison to photosynthesis this is good, it still wastes large amounts of solar energy, as most solar cells can't use the huge amount of infra-red thermal energy in sunlight.
A more radical rethinking of this problem has been to use solar electricity to electrolyse - or split - the carbon-dioxide into carbon and oxygen. Whilst in comparison to photosynthesis this is good, it still wastes large amounts of solar energy, as most solar cells can't use the huge amount of infra-red thermal energy in sunlight.
Stuart Licht and collegues from washington DC have come up with a way of increasing this efficency. They are electrolysing carbon-dioxide dissolved in lithium carbonate, so still using electricity, but they have discovered that if you heat the system to 950C you need 40% less electricity to split the carbon-dioxide.
The really neat thing about this is that you can use the infra-red part of the solar spectrum to heat up the electrolysis cell while still using a solar cell at maximum efficiency - in fact slightly better than normal, as there are less overheating problems. Using the most efficient solar cells available, they think they can use between 30 and 55% of the sun's energy to either produce carbon or at a slightly higher temperature carbon-monoxide.
This could either be used to sequester carbon, or the carbon-monoxide could be used as a feedstock to produce hydrocarbon-fuels or hydrogen.
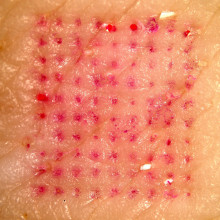
04:48 - Painless vaccine patches
Painless vaccine patches
For most of us, vaccinations are a normal, if not very pleasant, part of life. But what if there was an alternative? What if it was just as easy as putting on a sticking plaster? Researchers from Georgia Institute of Technology and Emory University reported in Nature Medicine this week that that might be closer to reality than we think.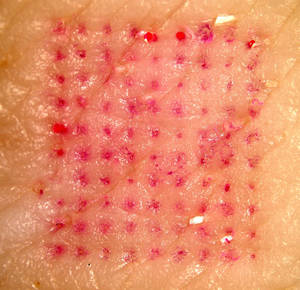
Sean Sullivan and his colleagues have developed a patch that delivered an influenza vaccine to mice. They compared the effectiveness of this patch with a more traditional intramuscular vaccine and found that it protected mice equally as well against the otherwise lethal flu virus. Even more exciting, 90 days after vaccination, the patch-vaccinated mice were found to activate the immune response to influenza more promptly and to clear the virus more quickly from lung cells.
The patches each held 100 tiny needles just 650 microns long that dissolve painlessly from the patch into the skin, taking with them their load of freeze-dried deactivated influenza virus. Vaccinating in this way takes advantage of the skin's special immunological properties and may explain why the patch seemed to provide even better protection than the intramuscular method by some measures. Some drug patches are already in widespread use, for example nicotine and contraceptive patches but what distinguishes these new microneedle patches is that they are able to deliver much larger particles such as viruses, as opposed to just small molecules like nicotine.
This work offers an exciting glimpse of what vaccines could be like in the future. If we no longer rely on traditional intramuscular vaccination, the risk of spreading disease through the re-use of contaminated needles, which is a common problem in some developing countries, would be eliminated. In addition, the patches allow the vaccine to be given with little or no medical training making large-scale vaccination programmes cheaper and easier. And on top of all that, getting life-saving protection against many infectious diseases could soon be pain-free.
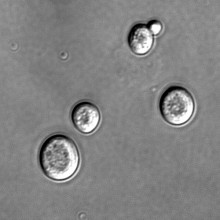
08:19 - What Makes A Cell Grow Old?
What Makes A Cell Grow Old?
A family of proteins has been identified that may limit the number of times any cell can divide, according to research published in the journal Nature Cell Biology this week.
Most cells seem to have an in-built limit to the number of times it can divide. From human cells down to single-celled yeast, they seem to limit themselves to only 20-30 divisions. Cell aging is thought to be related to the build up of toxic agents, but the mechanisms are not well understood.
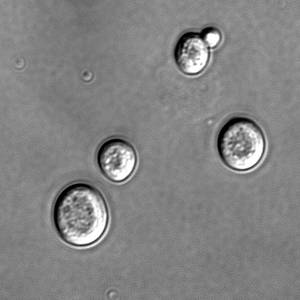 Now, Rong Li and colleagues in Kansas have identified a group of chemicals that may be responsible for limiting the reproductive lifespan of a cell.Yeast cells multiply by growing and dividing. A mother cell will create more of the components it requires, and then a daughter cell will bud off from the mother.
Now, Rong Li and colleagues in Kansas have identified a group of chemicals that may be responsible for limiting the reproductive lifespan of a cell.Yeast cells multiply by growing and dividing. A mother cell will create more of the components it requires, and then a daughter cell will bud off from the mother.
Each cell can only do this 20-30 times, and so far this has been put down to the build up of damage to DNA.
Multidrug resistance, or MDR, proteins are involved in transporting chemicals across the plasma membrane in yeast, and are inherited from the mother cell. Oddly, this inheritance is asymmetric - rather than a batch of new proteins being produced, then all the proteins split, the newly formed proteins preferentially move into the daughter cell.
Over subsequent divisions, the mother cell will keep hold of the 'old' MDR proteins, which may become damaged and lose function. As these proteins have crucial roles in metabolism and dealing with stress, this will lead to a gradual reduction in fitness of the mother cell, and a limit to how many times it can divide.
Supporting this hypothesis - yeast cells lacking certain MDR genes show a reduced replicative lifespan, while introducing extra MDR genes increases the lifespan.
It's now important to see if membrane transporter proteins show the same asymmetry in stem cells, which could limit the potential of stem cells, and to see if the same proteins play a role in aging in multicellular organisms - including us.

11:24 - Solid Li-ion batteries
Solid Li-ion batteries
Lithium ion batteries are now pretty much the standard high performance battery, they have a relatively high capacity and are becoming more affordable. They are used in most laptop and phone batteries and have recently been used in the Tesla Roadster sports car. However there are still major issues, the cathodes change shape and degrade over time, causing an electric car battery to only last 2-3 years. This is related to the batteries using a liquid electrolyte between the active electrodes, which also requires lots of infrastructure to hold everything apart, avoid short circuits, and keep the electrolyte in. One solution to this problem is to use solid electrolytes, which can allow ions to move whilst remaining solid, unfortunately these normally have to be made using vacuum deposition systems like computer chips, which are very expensive.
 A company called Planar Energy has developed a process for making these solid state batteries using printing techniques which are far cheaper than the deposition. They have also apparently increased the conductivity of the solid electrolyte to be comparable with the conventional electrolytes and made high quality electrolytes by using chemical self assembly processes.
A company called Planar Energy has developed a process for making these solid state batteries using printing techniques which are far cheaper than the deposition. They have also apparently increased the conductivity of the solid electrolyte to be comparable with the conventional electrolytes and made high quality electrolytes by using chemical self assembly processes.
Apparently the batteries have a capacity similar to the best high performance conventional batteries, but should survive longer, have three times the capacity, lose energy more slowly and be safer than conventional Li-ion batteries. Even if they don't manage the half the current price per kW hour, they do sound promising.
14:00 - Gel offers protection against HIV infection
Gel offers protection against HIV infection
Salim Abdool Karim, University of KwaZulu-Natal, Durban
Chris - Also in the news this week, a vaginal gel which contains the anti-AIDS drug called tenofovir has been found to reduce transmission rates of HIV amongst women by up to 50%. To explain a bit more about the study, which was carried out in South Africa, Salim Abdool Karim is from the University of KwaZulu-Natal is with us now to tell us what he did. Hello, Salim. Thank you for joining us. First of all can you set the scene for us; how big a problem worldwide is HIV? What's the scale of the problem?
Salim - Globally, we have a good idea of what's going on with the HIV epidemic. We know in 2009 that there are 33.4 million people living with HIV and that during 2008, there were 2.7 million new infections, and about 2 million deaths. So globally, the epidemic continues to grow, although it's growing more slowly now than it was some five years ago.
Chris - And just totting those numbers up in my head, that would mean something in the region of 7,000 people a day must be dying of HIV and 7,000 new infections every day. So we need to sort this out. A trial in Thailand suggests that vaccines are only at best 30% effective, so you've been taking a slightly different approach. These gels, how do they work?
Salim - There've been several gels that have been made, and foams and sponges as well, and they've been impregnated with different kinds of chemicals and called microbicides. And the underlying hypothesis is that these chemicals, when put into the genital tract, into the vagina, would prevent HIV from causing infections. Well up to now, in the past 15 years or so, there've been 11 trials of six candidates. None of which has been shown to work.
So, it's been a pretty difficult time in the field to find something that could prevent HIV infections. So we took a different approach. We decided to go with an anti-retroviral drug which is very widely used for treating AIDS and this drug is called tenofovir. It is a standard part of many cocktails of three drugs that are used to treat AIDS. So we put this drug into a gel formulation and we put it into single-use applicators and we did a study of 889 women in South Africa where they were asked to use this gel within 12 hours before sex and within 12 hours after sex. And what we found was that in the half of the women that used the tenofovir gel, there was 38 HIV infections, compared to 60 HIV infections in those women who were using an identical placebo gel. So that translates to a 39% protective effect of this gel.
Chris - And over what period of time were you studying? How long did you look at?
Salim - The first women were enrolled in May of 2007 and we completed the study in December of last year so 2 ½ years in total.
Chris - So if you extrapolated this to making this available to every exposed individual, let's just take Africa as an example, how many cases of HIV do you think you could prevent per year with this strategy?
Salim - We've done some mathematical modelling, creating a hypothetical scenario, as if we were implementing this in South Africa where we have very good data, and we emulated the kind of adherence we got within the study. So within the study, for those women who used the gel most consistently, they had 54% protection and those women who used it least consistently, less than 50% of their sex acts, we saw 28% protection. So what we did is we modelled that. We said, "Okay, what if 40% of the women used it in a consistent way as we observed in our study and 40% used it in an inconsistent way as we observed in our study." And if we did so, then over the next 20 years, we estimate we could prevent 1.3 million new HIV infections and avert just over 800,000 deaths, just in South Africa alone.
Chris - And what cost would that come out at? How much would it cost to implement that?
Salim - Now that's a bit more difficult to calculate. We know the actual cost of the gel is negligible. For the study where we only made a small quantity so we didn't benefit from scale, the actual gel costs about a cent or 2 in US cents. So the cost is not in the gel, but it's in the applicator, the wrapping, and the packaging, and so on, so that for the trial, each application costs us $0.32. If it's produced to scale, we would estimate that it will be substantially less and we worked out that even at the current price of $0.32, it is still cost-effective to implement because the cost of treating somebody who develops HIV infection is so high that even at this cost, to use the gel is more cost-effective than to allow women to get infected.
Chris - Well that brings me on to my last point, which is that the way we treat HIV is with triple therapy. We give people a combination of drugs so that the risk of the virus becoming resistant is reduced. You're using a single agent in this gel. Is there not a risk that we could end up eroding the ability of this agent to prevent HIV because people are being exposed to this as monotherapy?
Salim - Yes. It is a hypothetical concern and certainly, before the results of our study has been looked at in many different ways. These are the first data that have come out now where we can look at whether any of the 38 women who became infected while using tenofovir developed resistance, and the answer was no. We found no evidence of tenofovir resistance.
Chris - Indeed. Salim, thank you very much. That's Salim Abdool Karim who is from the University of KwaZulu-Natal in Durban, South Africa, and he is reporting there the study that he's just published in the journal Science, looking at the use of a vaginal gel which contains the agent tenofovir which seems to be able to reduce by a significant margin the rate of HIV transmission.










Comments
Add a comment