In this NewsFlash, how wind speeds over the oceans are changing, successful synthesis of sperm cells and getting under the skin of melanoma. Plus, the science of the blooming lily and how reprogrammed stem cells could restore sight for macular degeneration patients.
In this episode

00:28 - Wind speeds across the seas are increasing
Wind speeds across the seas are increasing
Researchers have reported this week that wind speeds across the oceans have increased, on average, by at least 0.25 per cent each year for the past 23 years.
We don't know whether this is linked to climate change, but Ian Young from the University of Technology in Melbourne claims that it's an important but often overlooked variable in climate change studies.

Publishing in the journal Science this week, the team used satellite data taken predominantly from GEOSAT altimeter readings, which they pieced together. They found that wind speeds are increasing more in the southern hemisphere than they are in the north and that the more extreme wind speeds are increasing by 0.75 per cent per year. Overall, a quarter of a percent each year doesn't sound like that much but it adds up to an increase, in the last 23 years, of between 5 and 10 per cent.
The effects of this could be bigger waves, but the team found no statistically significant growth in wave height over the past two decades. The heights of northern hemisphere waves seemed to be getting only very slightly smaller and the southern hemisphere waves were only getting slightly bigger. But neither of these changes were significant. And the researchers point out that this indicates wave height is determined by far more than just wind speeds, with other factors including swell and fetch.
On the other hand, the largest waves in higher latitudes do seem to be increasing in height. And the authors point out that, in more extreme conditions, this relationship between wind speed and wave height becomes more apparent. So you could argue that, if wind speeds across the oceans continue to increase, the largest waves would grow even bigger. This could potentially affect how humans are able to exploit the sea: affecting fishing and shipping. Or it could be a positive boon to our efforts in wind farming.
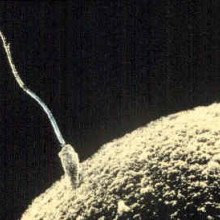
02:58 - How to Synthesise Sperm
How to Synthesise Sperm
For over 70 years scientists have been trying unsuccessfully to produce sperm in a petri dish. Now they've finally succeeded.
Writing in Nature, Takehiko Ogawa and his team at Yokohama City University reasoned that sperm probably depend for their development upon the complex three-dimensional environment provided by multiple nurturing cell types found within the testis. To recapitulate this sperm cell nursery, the team developed a system to culture small pieces of newborn mouse testicular tissue which they suspended in an agarose supporting gel and perfused with culture medium and a limited cocktail of growth factors.
 To make the job easier they also used tissue that had been genetically programmed to glow green when mature sperm cells were being produced. Using these tactics, and by carefully manipulating the chemical environment they eventually hit upon just the right conditions to persuade spermatogonial stem cells to turn first into spermatocytes, the first step in sperm maturation, and then to take the crucial step of halving their complement of genetic information and growing tails.
To make the job easier they also used tissue that had been genetically programmed to glow green when mature sperm cells were being produced. Using these tactics, and by carefully manipulating the chemical environment they eventually hit upon just the right conditions to persuade spermatogonial stem cells to turn first into spermatocytes, the first step in sperm maturation, and then to take the crucial step of halving their complement of genetic information and growing tails.
The ultimate proof, though, was when they were able to use these sperm to fertilise mouse eggs and produce viable - and fertile - offspring. This, they say, shows that testicular tissue can be triggered to produce mature, functional and fertile sperm under artificial conditions, which could have huge clinical implications for the treatment of infertility.
In particular, individuals facing chemotherapy for cancer could "bank" a sample of healthy testicular tissue prior to undergoing treatment. Kept in liquid nitrogen in the interim, this material could then be used to rear sperm for the patient at a later date, should the treatment render him infertile.

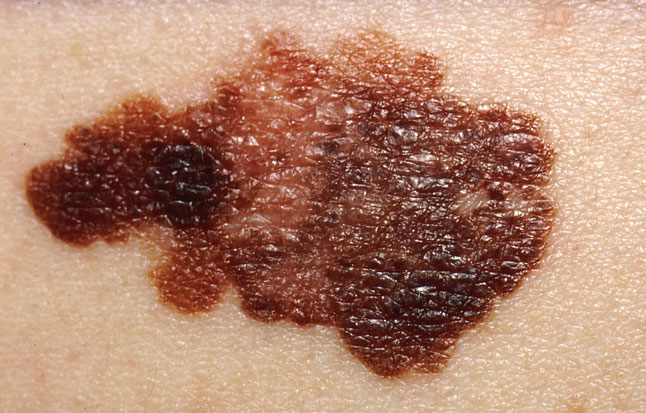
06:23 - Getting under the Skin of Melanoma
Getting under the Skin of Melanoma
Dr Leonard Zon, Harvard University
Diana - This week has also seen a breakthrough in the field of melanoma, a form of skin cancer that's becoming increasingly common; in fact, the incidence of the disease has doubled in the last ten years. But now there's some good news, because, with the help of a tankful of fish, scientists at Harvard University have discovered a key gene that drives the disease and therefore could hold the key to new ways to treat it. To explain more and talking to Chris, here's the author of the work, Leonard Zon.
 Leonard - Well, my laboratory has been focusing on melanoma which is a very deadly skin tumour and we had developed about 5 years ago a model of melanoma in the zebrafish. In this model, we took the human gene that's known to cause melanoma, a gene called BRAF and we over expressed it in the zebrafish, in combination with another gene, P53 which is the most common mutated form of a tumour suppressor gene in humans. That combination led to fish that develop melanoma. We were able to study those tumours and they really resemble very similar signatures of genes, to what you would see in a human tumour.
Leonard - Well, my laboratory has been focusing on melanoma which is a very deadly skin tumour and we had developed about 5 years ago a model of melanoma in the zebrafish. In this model, we took the human gene that's known to cause melanoma, a gene called BRAF and we over expressed it in the zebrafish, in combination with another gene, P53 which is the most common mutated form of a tumour suppressor gene in humans. That combination led to fish that develop melanoma. We were able to study those tumours and they really resemble very similar signatures of genes, to what you would see in a human tumour.
With that, we wanted to understand whether we could use this model to find new genes that cause cancer or find new therapies that might be used for the treatment of melanoma. We knew there was a region on human chromosome-1 that was amplified in about 30% of all human melanoma. We studied that region and found that there were 54 genes in that interval. We then looked at gene expression among 100 human melanomas and we found that 17 of those genes were expressed very highly. And so, we needed to figure out which was the driver gene, which was the most important to the cancer. And so, what we decided to do was to take each of these 17 genes and to inject them individually into our zebrafish embryos at the one cell stage, and then to grow up those fish and count how many fish developed a melanoma. What we found was that one of those genes, a gene called SETDB1, had the ability to greatly accelerate the melanoma, and this was likely to be the driver gene in this particular critical interval.
Chris - And is this representative of what you think goes on in humans? In other words, if you were to take human melanomas, real clinical tissue, do you see the same gene, this SETDB1 gene that you've now discovered to be involved also mutated in the human problem?
Leonard - Well that's right. So, 30 percent of human melanomas will have amplifications of SETDB1 and we went on to show in this paper that actually, 70 percent of melanomas will over express SETDB1. So it's something that's central to being a melanoma tumour is to over express this particular gene. So I think that over time, we'll be able to see that this gene also participates in other cancers too, and this region is also amplified in other tumours of humans such as lung tumours, and also breast tumours.
 Chris - We'll come on to what SETDB1 might be doing in a second, but first of all, if 70 percent have it, what about the 30 percent of human melanomas that don't? What's going on with them then?
Chris - We'll come on to what SETDB1 might be doing in a second, but first of all, if 70 percent have it, what about the 30 percent of human melanomas that don't? What's going on with them then?
Leonard - The way I think about this as an oncologist is, if I see a patient, I'll often describe their tumours as let's say, poorly differentiated or well-differentiated. What that means is that I can actually classify them by how the tumour looks under a microscope. We think that melanoma isn't a single disease, but there's actually different causes of melanoma. And so, there's different driver genes, depending on where the melanoma arises on your body and what types of exposures to carcinogens or light, sunlight is often thought as an instigator for the melanomas. So, with all these different options, the tumours could be heterogeneous and so, what we would say is that the 70 percent that over express SETDB1, that must be one category, and then the other 30 percent have a different classification and probably represent a different stage or a different location of those melanomas.
Chris - So what do you think SETDB1 is doing?
Leonard - Well, there's a new field that's blossomed over the past 5 years called epigenetics. Epigenetics deals with things that aren't inherited in a typical genetic manner. We're used to mutations, let's say, being inherited genetically, but in this particular case, DNA is actually wound around a spool and that spool is a set of proteins called histones. When DNA is wound too tightly, the genes are shut off. When DNA is wound loosely, the genes are on. This gene SETDB1 seems to wind the DNA a little bit too tightly and that shuts off particular types of genes that have an identity in the melanoma. So for instance, one class of genes that we found are a gene set called the hox genes which regulates the body plan normally of how embryos developed. But we think that somehow, this gene alteration in terms of expression leads to a change in the cell fate and that makes the tumours more invasive.
Chris - And does this in turn also give you new strategies for how to combat melanoma? The survival prospects for someone who's diagnosed with an advanced melanoma are really dismal at the moment, are we going to be able to do something about it with this discovery?
Leonard - So SETDB1 is an enzyme that actually methylates the histones and that regulates whether the DNA is tightly wound or not. And so, because it's an enzyme, it's possible to make inhibitors to this enzyme and so, we're in the process now of talking to drug companies to think about inhibiting this particular enzyme and we think this is a wonderful treatment for the patients, 70 percent of the patients who over expressed this gene who have melanoma.
Diana - Harvard Scientist Leonard Zon talking to Chris Smith there. He published that work he was discussing with me, this week in the journal Nature.

12:21 - How the lily blooms
How the lily blooms
This week scientists from Harvard and The University of Science and Technology of China have worked out how the lily pops open when it blooms.
They looked at the Asiatic lily, otherwise known as Lilium casablanca. Researchers Liang and Mahadevan marked the bud with dots along the inner petals and outer sepals; those are usually the green leaf-like parts underneath the petals. They then set up camera to track the dots as it grew.
 Publishing in PNAS, they found that the petal and sepal edges lengthened 40 per cent more than the midribs (these are the central veins in a petal or leaf). This disparity in growth created that characteristic wrinkling at the edge of the petal.
Publishing in PNAS, they found that the petal and sepal edges lengthened 40 per cent more than the midribs (these are the central veins in a petal or leaf). This disparity in growth created that characteristic wrinkling at the edge of the petal.
The authors say that this difference in growth causes stress to build up inside the bud and the forces eventually exceed those keeping the bud closed, causing the flower to burst open. Previous studies have argued that it is actually the midrib which causes the popping-open of the lily so, to test this, the researchers actually shaved it off the petal. Lo and behold, it still popped open as normal.
Why is this important? The authors argue that it can be mimicked in technology for designing thin film motors which would need blooming explosions on a small scale.

14:09 - Stem cells for Macular Degeneration
Stem cells for Macular Degeneration
Stem cells could hold the key to a future therapy for age-related macular degeneration (AMD), the leading cause of sight-loss among older adults.
The disease leads to the loss of retinal photoreceptor cells, the light-sensitive elements that convert light into nerve signals the brain can understand. This robs patients of their central, or macula, vision, the most acute part of the retina which is used to read, watch television or recognise peoples' faces.
| Vision without AMD: |
 |
| Vision with AMD: |
 |
Part of the pathology involves the degeneration of a tissue layer called the retinal pigment epithelium, a sheet of melanin-containing cells that nourish and maintain the photoreceptors. Replacing this lost tissue could therefore help to retard the progression of the condition.
To do this, Maria Kokkinaki and her colleagues at Georgetown University in Washington DC, writing in the journal Stem Cells, have developed a technique to transform mature skin cells into these specialised retinal cells. First the skin cells are "reprogrammed" by adding four genes (OCT4, SOX2, NANOG AND LIN28) which wipe the genetic slate of the cells and convert them into stem cells called hiPS cells (human induced pluripotential stem cells).
Grown under the correct culture environment, the team have found, these hiPS can be converted into retinal pigment epithelial cells that show identical morphology, gene activity, electrical, structural and biochemical behaviours to their naturally-encountered counterparts.
Crucially, once re-specialised, the cells showed no markers associated with still being stem cells, which is important from a safety perspective. This suggests that it should be possible to produce retinal spare-parts to tackle some of the commoner sight-loss syndromes using a similar technique.
However, the cells weren't perfect though. The telomeres, structures resembling end-caps on the chromosomes which shorten when cells divide, were shorter than they should have been, and the cells also showed other damage to their DNA which the team suspects might have been caused by the viruses used to deliver the reprogramming genes at the start of the procedure. This indicates that the technique, whilst an important proof of principle, has some way to go before it is ready for the clinic.
As study co-author Nady Golestaneh puts it, "This isn't ready for prime time though. We also identified some issues that need to be worked out before these cells are ready for transplantation but overall, this is a tremendous step forward in regenerative medicine."










Comments
Add a comment