eLife episode 29: Microbiome mind control, epilepsy and parastic worms
In this episode of the eLife podcast we hear about parasitic worms, dog tumours, epilepsy, DNA sequencing classes and social behaviour in mice.
In this episode
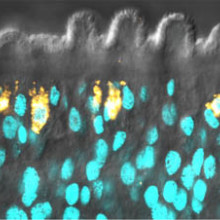
00:33 - Building barriers
Building barriers
with Jim Collins, University of Texas Southwestern Medical Center
At the University of Texas Southwestern Medical Center, Jim Collins studies schistosomiasis, the worm behind the disease Bilharzia. A population of stem cells inside the worm is critical to maintaining the organism's outer coat, which, he put to Chris Smith, is how it fends off the immune system...
Jim - Schistosomes infect 200 million worldwide, largely in sub-Saharan Africa. They kill about a quarter million of people every year. So, it's a serious problem. In fact, worldwide, it's almost as prevalent as diseases like malaria but receives far less attention.
Chris - How do people pick up the parasite in the first place?
Jim - People are exposed to the parasite when they go into water. People who were infected, they pass eggs through their urine or their faeces, and if that material reaches water, little critters come out which are called Miracidia that infect a snail. And then the snails inside the water, the parasites will propagate inside the snail and then they come out of the snail, and they're attracted by animals coming into the water namely humans. And then they burrow through your skin.
Chris - Once a person is carrying the parasite, where does it live in the body?
Jim - It depends on the schistosome. The schistosome we work on - Schistosoma mansoni - they live in the blood surrounding the intestine. That's where they start producing eggs. Once they're there, these parasites can live for an incredibly long period of time inside the blood. There's cases where people find that they move from endemic regions like Africa to places like the United Kingdom and they find 40 years later that they still have these parasites living inside them laying eggs.
Chris - But the blood must be a hostile environment for something to be able to persist with the full onslaught of the host's immune system potentially at its doorstep. How does it do that?
Jim - It's an open question. That's what this paper really tries to get at. What we knew is the parasites are capable of surviving inside the blood for decades. And so, we previously showed that they have these stem cells: basically cells that are able to rejuvenate tissues that are getting old. What we found in this paper was that the main job of these stem cells in these parasites is to generate the parasite's skin. This really unique structure, called the tegument. The tegument is what actually interfaces with the host immune system. It's not really known what features of the tegument allow the parasite to be able to invade the immune system. What we find is that the stem cells are continuously making new tegumental tissue. And so we think this doesn't necessarily answer the whole question about how the parasites are able to invade the host immune system, but we really think this is a very important clue as to what sorts of developmental tricks the parasite is using to be able to survive in the host blood.
Chris - And therefore, if one shuts down those stem cells, do you see a corresponding reduction in the production of this tegument and therefore a reduction in longevity or ability to fend off the host immune system in those worms?
Jim - Yes, we get rid of the stem cells - so we can kill the stem cells - and we completely blunt the ability to make new tegumental cells. But we don't know whether that has a consequence in the context of a natural infection. So these experiments that we do, we do them in a dish without a host immune system there.
Chris - How did you manage to show at this stage that those stem cells are producing that particular layer?
Jim - What we were able to do in the parasite is, we can kill these stem cells quite efficiently using genetic techniques, or also by just simply irradiating the parasites and that will kill all the stem cells. When we looked at the genes that were affected when the parasites didn't have stem cells for a really long time, we found that a number of these genes were genes previously shown to be associated with the schistosome's tegument. And so, that was a really big clue for us. So then what we did is we used a methodology to label the stem cells and ask, "what do they become?" It turns out what we were able to find is that a large fraction of the stem cells end up giving rise to cells that express things that are known to be associated with the tegument.
Chris - Is there particularly high turnover of these cells which would tell you that, in turn, there must be a very high turnover of the tegument - the skin of the parasite - which you could therefore deduce is because it is fending off actively some kind of host attack?
Jim - Exactly. So when we killed the stem cells, within a few days we lose progenitor cells to the tegument. And so, what we think is that stem cells are rejuvenating this tegumental tissue at a very high rate. The tegumental tissue is kind of a short-lived sort of surface.
Chris - So, does this give us some insights into how we might be able to attack schistosome in a new way? Is there a way of getting at those cells and effectively rendering it susceptible because it can't maintain this very high turnover of its outer layer?
Jim - Yeah. So there's two things. If we blunt the ability of the parasite to have stem cells that proliferate, that will ultimately lead to death of the parasite inside the host. The other real opportunity - the best thing for us - in treating schistosomiasis wouldn't necessarily be a drug but would be a vaccine. When we look at our data set, we basically have stumbled into a treasure trove of potential anti-schistosome vaccine candidates. In fact, some of the candidates that are being developed now as anti-schistosome vaccines are on the lists of genes that we identified. And so now we have potentially hundreds of molecules that we can follow up on or somebody else can follow up on to try to develop the next generation of anti-schistosome vaccines.

06:32 - Going global
Going global
with Andrea Strakova & Máire Ní Leathlobhair, The University of Cambridge
CTVT, or canine transmissible venereal tumour, is very unusual kind of cancer. The majority of cancers we see arise from an individual's own cells. But in this dog tumour, which emerged in one single dog 11,000 years ago, the cells from that animal's cancer are infectious and they can spread to other dogs, causing genital and facial tumours, when the animals mate. Now scientists at Cambridge University have studied dogs with the cancer from around the world, turning up a big surprise: the cancers have adopted DNA from the host dogs they've infected. Specifically this has happened in structures inside cells called mitochondria, which provide cells with their energy and this is how the tumour keeps fit. Georgia Mills went to see researchers Andrea Strakova and Máire Ní Leathlobhair at the department of veterinary medicine...
Andrea - We looked at CTVT tumours from different parts of the world. So we collected over 400 samples from 39 countries around the world and we looked at the mitochondrial DNA of these samples. So mitochondria are let's say, batteries of the cell which provide energy. They have a small piece of DNA which codes for the proteins needed by the mitochondria. We looked at the mutations or genetic changes in this mitochondrial DNA which gave us a unique opportunity to look at the ways that the disease spread around the world.
Georgia - Because these cancer cells are transferred directly from dog to dog, cells in each tumour are from the original dog to contract it 11,000 years ago. Meaning, it can be traced back to that time. But very occasionally throughout history, a tumour in a specific dog has done something a little unusual and grabbed mitochondrial DNA from cells of the host dog.
Máire - My name is Máire Ní Leathlobhair and I'm a second year PhD student in the Transmissible Cancer group. We were able to use the kind of phenomenon of horizontal transfer in CTVT cells as a kind of cellular tracking device. If you can imagine that usually, dogs would carry around their own normal mitochondria but these host mitochondria then swapped into the cancer cells and these were spread throughout global dog populations over hundreds to thousands of years. Using these and kind of the patterns we saw across our global population of samples, we're able to track how different groups of dogs afflicted with CTVT moved.
Georgia - This snatching of DNA happened at least five times in history. Meaning, the team could get a clearer idea of the movement of infected dogs throughout time. This pattern matched old trade routes across the ocean. So, it looks like people took their dogs with them to the high seas. Andrea...
Andrea - So, from the five different transfers of mitochondria, we were able to define five different clades. The timing of these clades was based on the mutations which we found in each of the mitochondria.
Georgia - By working out the just normal background rate of mutations, you can look at these, you can take the DNA from the dogs that are infected, look at the DNA in the mitochondria and then find out how old this cancer is.
Andrea - Exactly, because we used a so-called molecular clock so we can look at the number of mutations which we see in each of these different mitochondrial types. That helps us to look at the timing when each of them arise.
Georgia - And then these different clades are from when these big events, when mitochondrial DNA switched.
Andrea - Exactly. So, these are from the time when the mitochondrial DNA from the dog actually jumped into the tumour cell and this was the switch as you described.
Georgia - Why would this happen?
Andrea - Well, we think one of the reasons could be that the mitochondria in the tumour have so many mutations that they would be in a way less functional. And therefore, gaining mitochondria from the normal dog, that would provide a selective advantage for the cell.
Georgia - By grabbing this mitochondrial DNA from healthy dogs, they can reduce the number of harmful genetic changes that will have built up over time - a very clever trick. But the team came across something even more surprising.
Andrea - Well, we found one very, very special case in a dog in Nicaragua. What actually happened in this dog is that not only that the mitochondria jumped from the dog into the tumour cell but actually, we found that these two mitochondria mixed to create a single mitochondria formed both by the tumour and the dog DNA.
Georgia - Why is this unusual?
Andrea - This is something that has never been reported in cancers before. We believe this could be because it may be very difficult to detect. So we suspect that this process could actually be a lot more common than expected but we just don't have the tools to detect it. So, what we are planning to do in the future is to look more widely to see if this type of mitochondrial mixing is found in other tumours around the world. And also, it would be very interesting to see if we can see this type of mixing in human cancers as well.
Georgia - Do you think this would have any implications for cancer treatment?
Andrea - If mitochondrial recombination is indeed common in human cancers as well then certainly, they would be a potential for targeted treatment.
12:23 - New hope for epilepsy
New hope for epilepsy
with Catherine Woolley, Northwestern University
About 1% of adults in countries like the UK have epilepsy. This is a neurological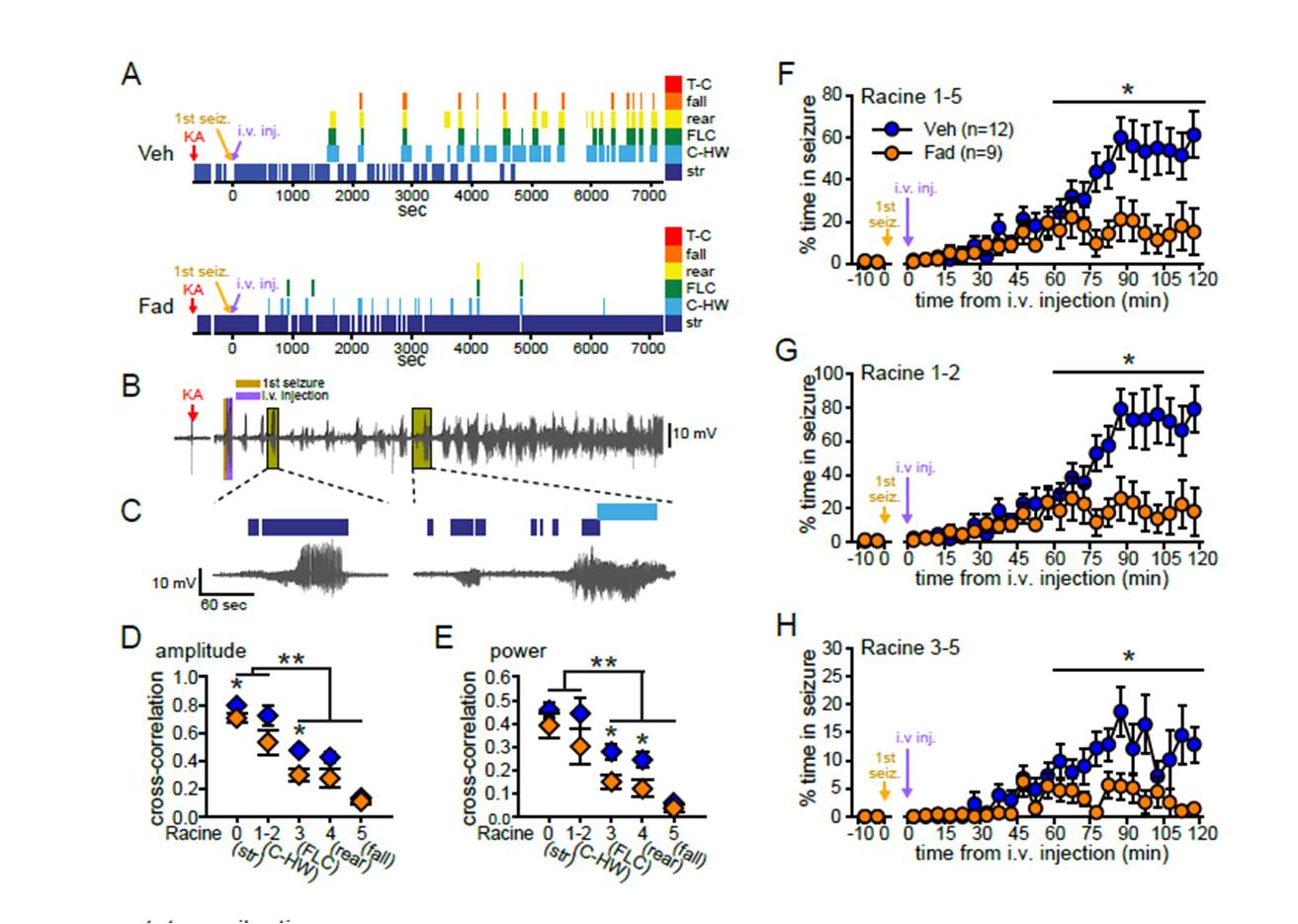 condition that occurs when groups of nerve cells adopt abnormal patterns of electrical activity that then become generalised across the brain causing the sufferer to lose consciousness. Usually the seizure is self-limiting and after a short time the brain resets itself to a normal neuronal firing pattern and the patient recovers. But sometimes a life-threatening condition called "status epilepticus" takes hold and the fit persists. Why wasn't known. But now Catherine Woolley has found that the hormone oestrogen might be to blame, as she explained to Chris Smith...
condition that occurs when groups of nerve cells adopt abnormal patterns of electrical activity that then become generalised across the brain causing the sufferer to lose consciousness. Usually the seizure is self-limiting and after a short time the brain resets itself to a normal neuronal firing pattern and the patient recovers. But sometimes a life-threatening condition called "status epilepticus" takes hold and the fit persists. Why wasn't known. But now Catherine Woolley has found that the hormone oestrogen might be to blame, as she explained to Chris Smith...
Catherine - We tested two new ideas. First is that seizures stimulate the synthesis or production of oestrogens in the brain and the second is that oestrogen synthesis in the brain during a seizure fuels that seizure, making it worse. If this were true, that would give us a novel handle that we could use to control that seizure activity.
Chris - Is this oestrogen which is coming from say, the ovaries in a female or is this oestrogen being made elsewhere in the body?
Catherine - This is not oestrogen coming from the ovaries. This is actually oestrogen that is synthesised directly within the brain.
Chris - What do we think those oestrogen molecules made in the brain do?
Catherine - It's actually been a longstanding question - what neurosteroid oestrogens might be doing in the brain. There has been until now, only circumstantial evidence that neurosteroid oestrogens actually are synthesised in the brain. And now we know that seizures are one of the circumstances in which they're synthesised and one of the roles that they play is to promote neural activity during that seizure. So, our hypothesis was that seizures initially stimulate oestrogen synthesis in the brain and that that sets up a feed forward loop that we then might be able to break by inhibiting the synthesis of oestrogens.
Chris - And so, that would be in keeping with this condition that's called status epilepticus where you see an individual who has a fit but then they keep fitting. The seizure does not extinguish itself.
Catherine - That's right. So status epilepticus is a condition that is considered to be a neurological emergency. It happens when a person has a prolonged episode of seizure activity that lasts for 5 minutes or longer, or when seizures occur in such quick succession that the person isn't able to regain consciousness between them.
Chris - So, how did you actually explore this? What did you do to try to find out whether it is down to oestrogen doing this?
Catherine - One experiment was to measure oestrogen levels in the brain before and during seizure activity using a technique called microdialysis and we found that when animals are having seizures, oestrogen levels increase in a part of the brain called the hippocampus which was very commonly involved in seizure activity.
Chris - So, that would fit in the first instance, if you excuse the pun, we've got a situation where in the context of seizure activity, levels of oestrogen go up. Does that prove that the oestrogen is causing the seizures to be maintained or merely that it's a bystander effect?
Catherine - That alone does not prove any causal relationship between the neurosteroid oestrogen synthesis and seizure activity. To investigate that question, we used a drug called an aromatase inhibitor that blocks the synthesis of oestrogens. We gave that aromatase inhibitor just at the beginning of a seizure and found that inhibiting oestrogen synthesis dramatically weakened seizures, made them much less severe. This drug was effective either if we gave it directly into the brain or if we gave it IV that is inhibiting oestrogen synthesis throughout the body.
Chris - Might it not be though that that molecule, as well as inhibiting the production of oestrogen happens to naturally be an anticonvulsant drug and it just stops the seizure for some other reason, nothing to do with oestrogen?
Catherine - One way that a person can assess whether a drug is working through the mechanism that one anticipates is to use two different drugs with different chemical structures they have in common but they do the same thing. In this case, inhibit the aromatase enzyme and we did find that two different aromatase inhibitors had the same effect of suppressing seizure activity.
Chris - Therefore, would this suggest that this could be quite a useful mechanism in the clinic if we can administer agents like this or exploit this mechanism to get better control of patients who have poorly controlled epilepsy?
Catherine - That's my hope. Aromatase inhibitors are clinically used now. In fact, we used one that is used now to treat breast cancer. And so, aromatase inhibitors are generally safe to use and I'm optimistic that aromatase inhibitors might indeed be a way to control neural activity during a seizure in a way that is targeted more toward the seizure activity than current anti-seizure medications are targeted.

18:27 - Training the next generation
Training the next generation
with Yaniv Erlich, Columbia University
A criticism often levelled at undergraduate science degrees is that a lot of spoon  feeding goes on and there's not enough emphasis placed on acquiring practical skills and the ability to think "outside the box". These are critical assets for a successful scientific career, particularly when it comes to the emerging and fast moving field of molecular biology. So why do we often wait until a student embarks on a PhD before giving them the chance to discover whether they really are cut out for this line of work? Yaniv Erlich has taken this DNA bull by the horns and set up a special class for his students at Columbia University where they were given a mobile DNA sequencing device and told to decode what was in a postdoc's dinner...
feeding goes on and there's not enough emphasis placed on acquiring practical skills and the ability to think "outside the box". These are critical assets for a successful scientific career, particularly when it comes to the emerging and fast moving field of molecular biology. So why do we often wait until a student embarks on a PhD before giving them the chance to discover whether they really are cut out for this line of work? Yaniv Erlich has taken this DNA bull by the horns and set up a special class for his students at Columbia University where they were given a mobile DNA sequencing device and told to decode what was in a postdoc's dinner...
Yaniv - Our idea was to let the students have hands-on experience with DNA sequencing technologies. Part of this class was that Sophie, my post-doc left a DNA from her food over a week. And she prepared DNA libraries and gave it to the students. They used these mobile DNA device to sequence the DNA from her food and they had to guess. They had to find what she ate. The whole concept here was to show them end to end how to generate, analyse, and infer results from DNA experiments.
Chris - Why did you decide to launch on this particular topic in the first place? Why do this?
Yaniv - The vision that we and others share is that we're going to see this era of ubiquitous DNA sequencing. So we want to have the students a glimpse to the future and also to train them in a way that they will help us to bring this future so they can think what they can do with this device. They can really realise the potential of this device by touching and playing with it writing some software to analyse its data and so on.
Chris - How did they respond? How did it go down with the class? Did the students take to it?
Yaniv - Yeah. So the students were highly enthusiastic about the class. I have to admit this is the first time that I'm teaching so I was a bit nervous before the class started in general. But the feedback was just fantastic. They love this class. They appreciated that we exposed them to an early on technology. One thing that was interesting to see, they are used to technologies that are near perfection. Let's say, cell phones or their operating systems on the computers. Now, this device is not yet perfected. There are still some gaps. They need to find work around, they need to tinker. It was interesting to see how they react to something that is not yet perfected, is not in a production grade I would say. But they're engineers and it's part of their training - is really to see things at an early stage and to think how they can evolve them, how they can help to mature this device, and how they can find work arounds.
Chris - I suppose in that respect, it's quite a nice prelude to what to expect from a career in science where people don't hand you a solution on a plate like they often are criticised for doing during an undergraduate degree.
Yaniv - Exactly. We got a feedback from some of the students that this is the first time that they feel in their undergraduate degree that they do science.
Chris - So that was what they learned. What did you learn and how does this inform what you do next with this project?
Yaniv - So, we learned how to design this experiment and how to engage the students. You can get quite a lot from a group of undergraduates and masters students that are highly enthusiastic about the devices. In the second Hackathon that was done around - the main topic was forensics, we in fact, started a research project in my group after the results of one of the Hackathon groups with the device. They came up with a very interesting algorithm and we said that this might be a research project so we should take on and really expand these preliminary leads that they found in the class.
Chris - Big trend now though to take things even lower and further down than that undergraduate level and people are talking about going into schools with this kind of thing. Do you think this is the kind of thing that could be used to encourage even younger learners, people before they've even gone to university to show them what they could be doing scientifically and then nurture more of that scientific spirit so we get more people going to university to study science?
Yaniv - Yeah. I think this device had a potential to go to schools. The main challenge is to create a curriculum around this device. We need to give the teachers a box with all their experimental setting, with the lesson plans that they can really conduct that in their class. This is something that we are actively thinking of - how we can take this device and build some curriculum around it to introduce it to high school students.
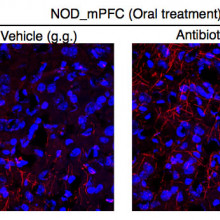
23:28 - Gut Feeling
Gut Feeling
with Mar Gacias, Icahn School of Medicine
People often talk about having a "gut feeling" for things, and now, thanks to 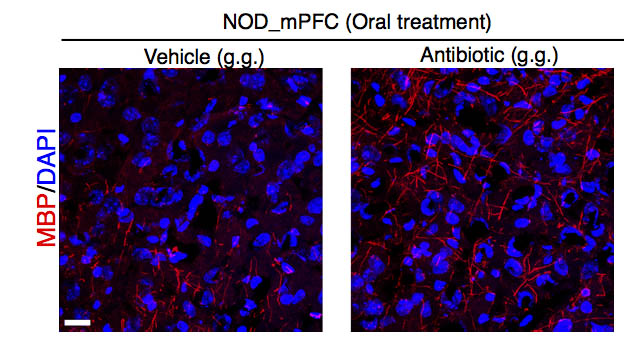 Icahn School of Medicine scientist Mar Gacias, we have new insights into why. The bugs that live in the intestine communicate chemically - via the bloodstream - with the brain, where they can control gene expression and myelination, the process that leads to the ensheathment and support of nerve fibres and ultimately affects their function... Mar spoke to Chris Smith about this new insight...
Icahn School of Medicine scientist Mar Gacias, we have new insights into why. The bugs that live in the intestine communicate chemically - via the bloodstream - with the brain, where they can control gene expression and myelination, the process that leads to the ensheathment and support of nerve fibres and ultimately affects their function... Mar spoke to Chris Smith about this new insight...
Mar - We have discovered how alterations in the bacterial communities that we have in our intestine are able to modulate the amount of myelin that we have in our brain. And these changes in the myelin are able to induce behavioural consequences depending on the genetic background of the mice.
Chris - There's been a number of studies recently showing that the microbiome, the makeup of bacteria living in us and on us can have effects on an individual's mood and their predisposition to experiencing symptoms of things like stress and anxiety. So, does this fit with what you're finding here?
Mar - Yes, I think it fits 100 per cent and our paper gives an extra level of confidence providing a strong evidence of the microbiota moderates brain function, and the mood.
Chris - To be clear, are the animals that are developing or are these adults or both?
Mar - In our study, we're using adult animals and I think that's an important feature of the paper because most of the papers that are out right now have been using experimental paradigms during development. So in our paper, we have used two different groups of mice with different genetic backgrounds. We have depleted the microbiota of these mice using a cocktail of antibiotics. And also, we have used water as an internal control, not to affect the microbiota. What we have seen is that when we depleted the microbiota of these mice, we are not able to see any depressive-like phenotypes after mild stressors. However, when these mice have the intact microbiota because they have been treated with water, they do.
Chris - In summary then you've got these groups of mice and you either deplete the microorganisms that live in them and on them with antibiotics or you don't do that depletion. You stress the mice and they only show signs of being stressed or anxious if they've got intestinal bacteria there that haven't been depleted.
Mar - Yes.
Chris - And now, you've gone a step further and found perhaps what's going on in the brain to account for why they have that abnormal or different response. So how did you do that?
Mar - After seeing the behavioural phenotype, we wanted to see what's going on in the brain and what we did, we did an analysis of the transcriptional changes that are occurring in terms of gene expression in a brain area called prefrontal cortex. We saw that in the mice displaying depressive-like phenotype, there were some clusters of genes that are associated to myelin or sleep disorders that were affected.
Chris - But how do you link the bacteria being there or not to those changes in gene expression? What's the connection between those two?
Mar - It's a great question. This is one of the key experiments of the paper where we were able to directly link the bacterial communities with these changes in the brain doing a transplantation experiment. Meaning that we took the faecal content of the mice that have displayed a behavioural phenotype and we transfer this faecal content into mice that were healthy. So what we saw is just by transplanting the microbacteria from one mice to the other ones, we were also able to transfer not only the gene expression profile but also their behaviour, showing a causality in this case because just change in the microbiota of the recipients was sufficient to change as well their behaviour and their genetic expression profile in the brain.
Chris - So, you take mice that are not showing any of these stressed features. You have a group of mice that are, you do a transfer of the bugs from the symptomatic mice into the asymptomatic, the normal behaving mice and they not only show a change in behaviour. They also show this change in gene expression.
Mar - Exactly.
Chris - Now, how does the brain know that those bugs are in the gut? What's the signal?
Mar - In our paper, we have studied the metabolites that are produced in the gut at the level of the intestine. We have seen that the gut is able to produce some molecules that they can travel to the brain and affect the function of the brain. We have found one molecule that it's highly expressed or highly produced in the mice with the depressive phenotype. That is able to impact directly on the brain affecting the cells that produce myelin.
- Previous A new way of keeping time?
- Next Reaching for the Stars










Comments
Add a comment