The story of human evolution is long and complicated, but the simple truth is - you're only here because your ancestors got lucky. Plus, we wind the clock back to the very start of human life, and discover how new research is pushing back the frontiers of human embryology. Plus a suitably festive gene of the month. This is the Naked Genetics podcast for December 2016 with me, Dr Kat Arney, brought to you in association with The Genetics Society, online at genetics. org. uk.
In this episode
01:05 - Everyone who ever lived
Everyone who ever lived
with Adam Rutherford
It’s been roughly 200,000 years since the first anatomically modern humans, our species Homo sapiens, arose in Africa, and since then we’ve pretty much got everywhere. Our amazing story is written into our genes, mixed up with genes of the other early humans - such as Neanderthals - that we met and mated with along the way. This genetic journey is the subject of a new book - A Brief History of Everyone Who Ever Lived - by writer, broadcaster and geneticist Adam Rutherford. Kat Arney caught up with him to find out about the story behind his story of our story.
Adam - It’s got a certainly ambitious title. It is a brief history of everyone who ever lived. It is literally true in a sense that what it does is trying fuse older academic fields which is history and archaeology with a very much newer one which is genomics. So using DNA as a historical source to compare tests, verify, debunk what we think we know about history using the genome.
Kat - We hear a lot about all these things like 23andMe, you can get your genome done, people seem to be extracting DNA out of everything they can dig out of the ground. How has the science of DNA changed what we understand about our origins and how recent are we talking that these changes have come about?
Adam - It’s not an exaggeration to say that genomics has revolutionised palaeoanthropology, so the study of the evolution of humans. It’s all really happened in the last 10 years – mostly in the last 2 years. So, the first non-Homo sapiens human genome was only sequenced in 2009 and 2010 which was Neanderthal. From Neanderthal 1 – one of the first three Neanderthals found – this one being the one from the Neander Valley in Germany. We immediately began to make comparisons between us and them, and discovered a whole bunch of stuff which included the stuff that people now already know which is that we successfully interbred with Neanderthals. Now I argue in the book that that means that they were not a different species from us which I can defend…
Kat - Controversial, but yeah…
Adam - It is a little bit and I think the problem really is less to do with what the data says and more to do with our species definition. There isn’t one single species definition but the most widely used is that two organisms who are incapable of producing fertile offspring must be a different species. The fact that we can have our DNA tested – you and me as white skinned Europeans or largely white-skinned Europeans in my case – that we both have Neanderthal DNA in our genomes. We know that because we compared it with the Neanderthal DNA that was taken out of Neanderthals. The fact that we carry their DNA to this day means they had fertile offspring.
Kat - So they must've bonked each other basically.
Adam - Absolutely. I mean categorically, we can demonstrate that’s true. We know of – initially, it was only five but now, that number has gone up - gene flow events is what we refer to them as.
Kat - Euphemistically.
Adam - Euphemistically that. It does mean sex. It does mean sex in a sort of grand population scale rather than just two people getting it on. Many times that Homo sapiens met Homo neanderthalensis, we see gene flow events. We see it from males to females and females to males. We see it from Homo sapiens into Homo neanderthalensis and we see it from Homo neanderthalensis into Homo sapiens. So there isn’t a version of this which involves the production of infertile offspring.
Kat - A lot of us will have seen the image of the March of Progress, that diagram of evolution that starts with a knuckle-dragging monkey goes through various kind of heavy-browed knuckle-dragging ancestors, and the arrives at modern humans. So that very neat idea of a tree or a progression, that’s not right then, is it?
Adam - No. I think we’re ready to abandon that. I think we’ve been ready to abandon that for a few years now. That was an idea drawn in the 1930s and I think the fewer samples we had and the few data that we had, the more confident we were about – as you say, the March of Progress is what it’s known as. There are two things I argue are wrong with that. The first is the name and the direction of travel. So it suggests that ape-like ancestors on all fours were somehow simpler than us and that we have progressed into being bigger-brained and we used tools and that the final one is a sort of bearded man with spear. So, he is now Homo sapiens, an intelligent version of what had come before. That’s wrong. There is no direction to evolution. We are evolved, all organisms are evolved to exploit their environment at that time. So, we are no more or less evolved than any other organism on Earth. So there is no direction to the travel. There is no inevitability about tool use, and there's no inevitability about having bigger brains, and the intelligence that we enjoy. That’s the first thing. The second thing that’s wrong with it is with another path.
Kat - It seems to be more like not even a family tree but this enormous family bush, strangling thing all over the place.
Adam - Yeah, that’s right. I think maybe also, we need to think quite harder about abandoning the tree as a metaphor. I think it works on a broad scale, but of all the human species that we’ve discovered so far, it’s very difficult to put them on any sort of tree-like structure. We can't really draw the lines between any individual species as they’ve been traditionally known. We now can say because of genomics that Neanderthals did successfully mate with Homo sapiens. We know another new species, the Denisovans which was discovered in the last 5 years, very few specimens, a couple of teeth, and a finger bone. But we got the DNA out of that, got the genome out of that, and know that it’s not the same as Neanderthals and it’s not the same as us. And so, it’s another human species. We can position them on a geographical map and we can position them on a chronological map, and we can also look at the proportions of people living today, extant people and see how much DNA they carry of Denisovans. What we see is that the further east you go, the more Denisovan DNA there is, and the less Neanderthal DNA there is. If you go back into Africa, you see no Denisovan DNA and you see almost no Neanderthal DNA. What we do see is from modern Europeans migrating back into Africa. So we effectively have to redraw the map of how early humans migrated and spread all over the world over the past 250,000 years.
Kat - So that’s going way, way back in tens of thousands, hundreds of thousands of years back. What can our DNA tell us now about where someone like, you and I have come from? There's lots of services now. You can spit in a tube, you can get back this reading that says, “You're part German, part from here, part from there.” What can we know about where our populations today have come from?
Adam - It’s a great question. It’s a tricky one to answer because DNA will tell you such powerful things about immediate family, and more and more where people are discovering siblings and uncles, and aunts, and parents if they were adopted. Particularly in this era when there were a lot of war babies. We can use DNA with great accuracy to identify immediate family. So it’s very powerful in that regard. As in exactly the same way it is for identifying people, for being used in forensics. But the further back you go, the dimmer the past becomes and that applies to every bit of evidence that we use, and that includes DNA. The way I think about this is that the 2 parents, and 4 grandparents, and 8 great grandparents, and 16 great great grandparents, and so on, on a fully outbred family tree, the number of ancestors you have doubles every generation. So if you go back to 1066 which is almost a thousand years ago, then an outbred family tree has something like 100 trillion people.
Kat - Okay, that is more people that have ever lived on Earth. The math doesn’t work in this equation.
Adam - It doesn’t work at all. It’s a thousand times more than the number of people who’ve ever lived on Earth and we know that. That’s not controversial to say. So we figured that our family trees become pollarded and they overlap very soon a few generations up. And then if that’s true which has to be because of the maths, there's the concept called the isopoint where all family trees cross. The isopoint for Europeans is around about the 15th century. So there was hypothetically and actually – although we can't say who it was – a person who existed through which every single person’s family tree flows. Every single European person has a common ancestor of about 500 years ago, which I find just absolutely bonkers.
Kat - I found this mind bending when I was reading the book. There's so many amazing things in there. You're digging into the story of Richard III, you dig into the recent story of Jack the Ripper, you're looking at where are we going, what can our DNA tell us. If there was kind of one thing in a sentence that you could sum up about, what you want people to take away from this book, your take away points, what do you think that would be?
Adam - I think it would be that humans are much more complex than we thought they were. And that I think is something worthy of celebration. The second thing is that if people are trying to give you simple answers to complex problems, then they're probably selling something. It is the same answer. It’s the same answer. I'm not sure why we thought there would be simple answers to the questions of, what makes us us, how we came to be what we are when we are so beautifully complex. For a long time, we thought there were going to be simple answers because I don’t think we’d thought about it hard enough. And now, what we find is that we’re even more complex in terms of our genome than we are in terms of our behaviour and that’s exciting!
Kat - Science writer Adam Rutherford, and his book A brief history of everyone who ever lived is available in all good bookshops and online retailers. And if you're in need of another idea for a Christmas present for the genetics buff in your life, my own book, Herding Hemingway's Cats - Understanding How our Genes Work, is out now too.
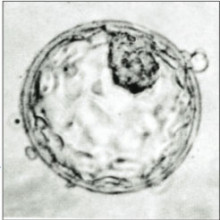
12:08 - Magdalena Zernicka-Goetz: Early embryos
Magdalena Zernicka-Goetz: Early embryos
with Magdalena Zernicka-Goetz, University of Cambridge
We've just wound the clock back to the beginning of human history, now let's tick back to the very start of each individual human life - the moment of fertilisation when egg and sperm meet. In 1978, the world welcomed Louise Brown, the first test-tube baby born by in vitro fertilisation, or IVF. Her birth sparked a heated debate about reproductive technology, and in particular, the use of human embryos for research purposes. This led to the so-called Warnock report in 1984, based on the deliberations of a committee chaired by Baroness Mary Warnock, which in turn led to 1990 legislation establishing the UK Human Fertilisation and Embryology Authority and a law banning research on human embryos grown in the lab for longer than 14 days. Until this year, this was thought to be quite a long period, as nobody was managing to grow embryos past 6 or so day, when they're a little football of cells known as a blastocyst. Then in May 2016, Cambridge University's Professor Magdalena Zernicka-Goetz and her team managed to keep embryos growing in the lab for a staggering 13 days. So should we change the law to go further and grow embryos for longer? Kat Arney met up with Magdalena at a recent (and rather noisy) conference run by the Progress Educational Trust to discuss exactly this question.
Magdalena - Well, when it’s successful, after fertilisation happens and activation of the egg. This is the time when you start to see the first divisions and they go on like that until the 6th day of development. So the big egg you can say is chopped up into smaller and smaller. And then when we have quite a few of those cells, they start to decide their fate and differentiate. Their decision at that stage of life is either to form the future foetus or the placenta, or the yolk sac. So they have three fate choices they have to make within these first few days. What we discussed today about is more focus on what happens in the next 6 days of their life which was a black box of development until very recently, when we now can directly look into these 6 extra days. So we doubled the time at which we can look at the development and discover what happens to those three distinct type of cells when they start to talk to each other and prepare the foundation for the future baby.
Kat - How do you do this? How do you go from this little football of cells that we can see suspended in a dish to making them start to go that extra few steps? What happens?
Magdalena - So essentially, the key was that there was no culture system developed that would allow this embryo to survive outside the body of the mother. So what we have done is to find the right environment, the right ingredients, the right hormones, and growth factors which we then, as a cocktail, put into this environment to support the growth of these embryos in a dish.
Kat - Because this would normally be a stage when the little embryo has started embedding in the womb and sorting itself out.
Magdalena - Yes. So this will be normally the time when the embryo will implant and to get into the physical contact with the body of the mother and it was thought that this physical contact might be absolutely essential for its further development. So we showed that it’s not so. These embryos can develop outside the body of the mother for these extra 6 days. I'm sure that at that point on, they would relying on the body of the mother for lots of different things, the most important, nutrition. but since we provided them with the nutrition, they were able to self-organise themselves and progressing through development. What we have shown, that they really go through these 5 critical steps of development in a correct way. So that was very important for us. Not only see whether they can grow or not, but whether they actually take those decisions in an expected way from what we know.
Kat - You’ve grown these embryos now for 13 days in this system. Can you go any further?
Magdalena - No, we can't go any further. So first of all, we don’t want to go any further because there is a recognised law right now that you cannot go beyond the day 14 of development. So we stopped this one day before, in the day 13. If it were to change one day, I don’t even know whether this culture system would be supporting development beyond day 13.
Kat - Why do you want to do this research? What would be the benefits of being able to grow embryos like this?
Magdalena - The first critical thing is to realise that we know so much about development of many other model organisms like fruit flies, worms, and we don’t know much about development of our own embryos. Actually, at this stage of development, we have nothing like that knowledge. So this is all really black box of development. So of course, we want to understand the principles which govern normal development at this stage to be able to understand what are the conditions that make those embryos developing correctly and what are the factors that we have to take into account when they do not develop correctly. So, a lot of it is to understand basic development and therefore, curiosity as each single scientist has. But also, it has a tremendous translational implication and we discussed a lot of those for IVF clinics but also for treating diseases that manifest themselves at this stage.
Kat - Magdalena Zernicka-Goetz from Cambridge University.
18:01 - Sarah Norcross: Embryo research
Sarah Norcross: Embryo research
with Sarah Norcross, Progress Educational Trust
The issues raised by Magdalena Zernicka-Goetz's work are controversial and challenging. Just because we might be able to grow embryos longer than 14 days - and with it discover important clues about early human development and what goes wrong in infertility, miscarriage and some types of cancer - does that mean we should? Kat Arney speaks to Sarah Norcross, director of the Progress Educational Trust, whose December meeting brought together scientists, philosophers and even an ex-archbishop of Canterbury to discuss this thorny question.
Sarah - We were thinking for our conference what would be really interesting? We always have to look for hot topics. Professor Azim Surani, who’s a geneticist, who has done a lot of embryo research at Cambridge, he was discussing at our conference in 2015 about the germ line in relation to mitochondrial donation. One of the questions that came out from the audience was, “What about if you could extend the amount of time you could do research on embryos for, would that be useful?” And he said, “Yes.” We thought this quite interesting. It did catch the media’s attention. But then when Magdalena Zernicka-Goetz, when her paper was published in Nature saying that she managed to develop embryos in vitro for up to 13 days, we thought, “Yes, this is a really interesting topic.” Because earlier this year in January 2016, that’s when Kathy Niakan was granted a license by the Human Fertilisation and Embryology Authority to do genome editing research in embryos. So because genome editing is a very new technology because we haven't started doing it in embryos in the UK yet because Kathy has just got her license. Well, if they did extend the period for which you could experiment on embryos, what would that mean in terms of genome editing technology? What could that possibly help us with? Would it help us just with understanding the basic science of something that is so fascinating and fundamental to us or the very early stages of human development – something that’s little understood and what the scientists call “The black box”. Would it allow changes to be made in embryos that can then in effect, eradicate disease because it will go from the germline? So, could we get rid of some conditions? There seem to be these possibilities and then of course, as soon as you start talking about those and people start thinking about enhancement, and how far should we go with this? Should we put checks and balances in place? And so, I think it was really important to start discussing genome editing and how long we should allow research in embryos to go on for before we call time on it?
Kat - What really struck me from today’s meeting was that this really dates back to almost a very specific point in time to the birth of the first test tube baby Louise Brown in 1978 and the public suddenly going, “Wow! We can do this now.” And then from that, so you can do IVF, you can put an egg and a sperm together in a test tube, it will form an embryo. Some of those will be viable, some of those will not be viable. You can implant some of them back in the womb, some of them will grow to make a baby, some of them won't. What are some of the issues around researching in this area? Should we even be doing this?
Sarah - I think there are many thousands of surplus embryos created through IVF. Some patients choose to donate those for research and that’s very laudable. When you think about all the research that needs to be done, it’s not just what the next “wow factor” scientific development may be.
Kat - I guess, just research into how to make IVF better and more successful.
Sarah - How to make IVF better is a real driver in this area because IVF, while it’s a successful treatment for many, it does fail a lot as well; the heartbreak that it causes people when it fails, and also because 60 per cent of people pay for their treatment in the UK, the expense when it fails as well is huge. So it’s an emotional expense as well as a financial expense.
Kat - When we’re thinking about genetic modification of human embryos whether that’s for research purposes or even potentially for human modification, how important is it that you think the public understands what is going on because I think there have been some examples for example with GM technology and food where it’s being presented as, “Here's this. Enjoy! Eat this stuff.” People go, “Uhmm, I don’t like that.” How can we educate and inform the public about what really is going on here?
Sarah - I think there are huge lessons to be learned, we’re determined to as the Progress Educational Trust to do something about that. So we’re starting to work on a project with small workshops to understand how people understand genome editing. So we’re taking people who are not scientists but who perhaps have got an interest in fertility. They perhaps work in a clinic or they represent a patient group, and to get them to give us their impressions about it, to have scientists explain it to them, and so that we can try and develop language of materials that will help a wider group of people understand the science behind it. So that we could explain to them in a way that’s accurate but not so heavily encumbered with difficult scientific terminology that they can get their brain around it, listening to it as a soundbite because unfortunately, I know for me, our time is so limited that we’re trying to get to grips with an issue by reading something really quickly. So we want to know about the paragraph at the most so they go, “Oh yes, I know about that now.” And then I can form my opinion on that because it’s similar to this or whatever else it is. And also because, I think it’s really important for scientists to speak to the public about their work, to get the public to understand what they're doing right from the beginning because I think otherwise, people think wrongly that perhaps they're hiding things, that there's something secretive going on, that there's a conspiracy. As soon as there's a sign there's a lack of communication, people project things onto that which is usually very negative. And so, I think by being open and transparent and say, “We’re looking at this. This is early stage. It’s not going to cure cancer. It’s not going to do this, but this is where we are and this is the start of something.” People can understand that and appreciate that that’s the way that science works. I think that’s going to be an important part of that, for people to understand that you don’t just come up with a light bulb moment that suddenly cures something.
Kat - Eureka!
Sarah - Exactly, eureka! That’s the word.
Kat - And finally, when we’re thinking about laws, we heard today from Baroness Mary Warnock who was one of the people who put together the legislation that says you can only go up to 14 days and the establishment of the Human Fertilisation Embryology Authority here in the UK that regulates human embryo work and human embryo research. is it really just down to scientists to tell us what they want to do and the public should just understand it and be okay with it?
Sarah - No. I don’t think scientists do or should have the authority, power, whatever you want to call it, to tell the public, “This is what we’re going to do and we’re going to do it, and you’ve got to go along with it because it’s science.” They have to make the arguments for while it’s important, I really do firmly believe that they have to build a case for why we should change the law. We shouldn’t just do it because we can or we might like to, but they have to make some sort of justification. Because as well as the sort of public engagement work that Progress Educational Trust does, we also campaigning work. I don’t want to campaign for something just because someone thinks it’s a bit of a whim.
Kat - This sounds like fun or my evil plan!
Sarah - Yeah, my evil plan! For me, it’s got to have some value to society at the end to improve the choices for people who are affected by infertility and genetic conditions.
Kat - Sarah Norcross from the Progress Educational trust, and you can find out more about their work and the conference at http://www.progress.org.uk/home

26:58 - Gene of the Month: Christmas disease
Gene of the Month: Christmas disease
with Kat Arney
And finally it's time for our Gene of the Month, which is the gene - or rather the gene fault - responsible for Christmas disease. This was first described in a paper in the British Medical Journal in 1952, and named after the unfortunate child suffering from it - a five-year-old boy called Stephen Christmas, who bled profusely in response to the slightest injury. It's now known to be a rare disease called haemophilia B, and is due to a fault in the gene encoding a protein called Factor IX, which is involved in blood clotting - hence the unstoppable bleeding in people who suffer from it. Factor IX is part of the cascade of chemical reactions that end up chopping up a large molecule called fibrinogen to make a sturdy, insoluble protein called fibrin. This then forms a sticky net at the site of a wound, trapping blood cells and starting the clotting process.
The gene for factor IX is carried on the X chromosome - one of the two sex chromosomes. This means that the disease usually only affects boys - who have one X chromosome carrying the faulty gene and one Y chromosome - as girls have two X chromosomes, so a fault in one can be balanced out by a functional gene on the other.
In 2009, scientists in the US showed that Queen Victoria actually, carried a faulty version of the factor IX gene on one of her X chromosomes, and three generations of boys in the British Royal family were affected by the condition, sometimes known as the 'royal disease'.
Today, Christmas disease is treated with injections of Factor IX produced in genetically modified bacteria, but previously was only treatable through blood transfusions. Sadly for Stephen Christmas, he contracted HIV through one of these transfusions, before they were regularly screened for the virus, and died of AIDS in 1993. So at this time of the year, spare a moment to remember the other meaning of Christmas.










Comments
Add a comment