Could we one day be popping pills that tweak our metabolism and make us live longer? It's not as far off as you might think. Plus, the cow genome goes large, bat flu flies into focus, and an untidy gene of the month.
In this episode

00:56 - Jennifer Tullett - Ageing & metabolism
Jennifer Tullett - Ageing & metabolism
with Jennifer Tullett, University of Ken
Kat - Last month we took a look at the researchers hunting for the genes behind ageing, and now we're returning to the topic with a closer look at the links between genes, ageing and metabolism - that's how we use food to generate energy in the body, and how animals respond to changes in the environment. To start with, I spoke to Dr Jennifer Tullett, from the University of Kent, to find out more about her work in this area.
Jennifer - I try and understand the function of individual genes that affect lifespan and healthspan. We're very interested in that here. I also look at how you can alter the environment of an organism. For example, you can alter its dietary intake and extend its lifespan. So, we're interested in that and also, how these dietary interventions for example impact on the expression of genes and the function of genes in these processes.
Kat - So, how the things around us control whether our genes are switched on or off at different times.
Jennifer - Yes, exactly.
Kat - Because this might seem strange because you think, "Well, I've got my genes and they just do their thing." Actually, the genome is changing all the time in terms of which genes are on and which genes are off.
Jennifer - Yes, it's very important that genes are switched on and off but it's probably much more intricate than that and there's a lot of different levels of gene expression. And it's a coordination expression of lots of different things at once.
Kat - So, it's about the kind of the orchestra, how it plays these different genes.
Jennifer - Yeah, that's a very nice analogy.
Kat - So tell me, what are you studying? How are you trying to home in on these genes and how they interact with our environment?
Jennifer - So, I guess my main focus is, I look at different transcription factors that are involved in ageing. Transcription factors are proteins that are responsible for controlling the expression of a whole range of other genes. So, they're key regulatory molecules.
Kat - Kind of master switches I guess.
Jennifer - Master switches, yeah. And I'm interested in two of those specifically. One is called DAF-16 and the other one is called SKN-1. So, I look at the activity of these genes. I look at these other genes that they regulate and I try and understand how they function in different scenarios and which one of their target genes that they're regulating are important for lifespan.
Kat - So, it could be doing all sorts of things from just keeping the worms going, to actually controlling how long they're going to live for.
Jennifer - They live for about 3 weeks normally, but if you mutate either DAF-16 or SKN-1, then you'll dramatically shorten that lifespan by about 30%.
Kat - So, like two weeks.
Jennifer - Two weeks, yeah.
Kat - What kind of things are you finding so far that seem to be important that these genes do?
Jennifer - One gene that seems to be very interesting is AMPK which is AMP - activated protein kinase.
Kat - Sounds complicated.
Jennifer - And that's a very important metabolic enzyme and that seems to be very important for regulating lifespan as well.
Kat - It seems to me that there seems to be a lot about the metabolism, how our bodies use energy that is linked to ageing. Is it something to do with like, 'the faster you burn, the shorter you live'?
Jennifer - That's the rate of living hypothesis type idea. That doesn't seem to be quite as fashionable any more in the ageing field because there are examples of small animals that have high metabolic rates that live for quite a long time. So, it doesn't necessarily correlate the two things.
Kat - What do we know about how metabolism is affecting these worms in their lifespan and how does it all seem to fit together?
Jennifer - In C. elegans, actually, one of the most amazing lifespan extending mutations is a mutation in a gene called DAF-2. And if you make one single point mutation in this gene then you can double and even sometimes triple the lifespan of a worm.
Kat - That's pretty incredible.
Jennifer - It's very incredible and it is also conserved so you get the same effect in flies, you get it in mice, and there's also a small amount of evidence that the same thing could happen in humans as well. So, if you have this massive lifespan extension with mutation of DAF-2 - DAF-2 is the insulin receptor. So, these worms must have altered metabolism. In fact, if this was a human, they would be severely diabetic.
Kat - Because insulin is how your body controls the level of sugar that goes into a cell and stuff like that.
Jennifer - Yes, exactly. So, this lifespan extension caused by mutation of DAF-2 is dependent on those two transcription factors that I mentioned which are DAF-16 and SKN-1. These transcription factors are really important because they are regulating a whole variety of other genes. One of the genes that I've recently discovered that does contribute to this massive lifespan extension of DAF-2 worms is this gene called AMPK. This is a critical metabolic enzyme and it is responsible for regulating the energy homeostasis in the cell. So, it can sense when the body has low amounts of energy available and that switches it on.
Kat - So, it'll kind of stop you burning so much energy, just keeps you going.
Jennifer - Yes, keeps everything under control. It seems that if you make mutations in this enzyme, then the worms can't live as long.
Kat - Where do you want to see this go? I mean obviously, this is in worms and that could be quite useful thing to do for the worm population I guess. But where do you see the applications of this work going?
Jennifer - Well, something that we do a lot of here is, we try and look between species. Worms are really great to do ageing studies in because they're so short-lived. That's why so many people do it. Drosophila are also very good. Their short-lived but still, it's still a lot longer than a worm. So, what we try and do here is, if we find something in a worm, we then try and move it into a fly and then we can also try and move up to a mammalian model as well with the hope that if this research holds true in a mammal then it could also be relevant to humans.
Kat - And it does seem to me that there's a lot of interest in the field of ageing in metabolism and tweaking metabolism, and people having very, very low calorie diets or taking all these kind of supplements and things. Are there things that we definitely know at the moment will work in humans? If I wanted to live longer and tweak my metabolism, is there anything I know that I should definitely be trying to do?
Jennifer - I think in terms of an anti-ageing pill, if that's what you're asking, I don't think we're quite there yet. We do know in lower organisms that there are certain drugs that can be given to the animals to extend their lifespan. There are a number of those including rapamycin. That's a very good one. But in terms of humans, I wouldn't like to recommend that anyone takes any of these things in the very near future. I think the best thing people can do for now is just maintain a healthy diet, healthy lifestyle...
Kat - the boring stuff!
Jennifer - Basically the usual boring stuff, yes, and you're probably in with a good chance of living a healthy existence.
Kat - How long would you want to live for?
Jennifer - I would want to live for as long as I can be healthy and independent. I think that's probably the same for a lot of people. If I'm healthy and independent then I'm likely to be very happy and I can just carry on. But that is what our research is geared towards. We're not geared towards making people live forever in an awful decrepit state. That's nothing. That's not what we want at all. What we really are interested in is increasing the healthy lifespan of an individual.
That's what we really want to achieve. And the amazing thing to me as a researcher, the thing that's really kept me going in this field is the fact that if you have these point mutations that extend the lifespan of a worm or a fly or a mouse, not only do they live longer, but they are actually an awful lot healthier. That's the amazing thing about them. So, I think the fact that we study ageing and lifespan as an endpoint is one thing, but the really important thing to bear in mind is that we're really interested in health span.
Kat - That was Jennifer Tullett from the University of Kent.
08:32 - Dominic Kwiatkowski - Big malaria study
Dominic Kwiatkowski - Big malaria study
with Prof Dominic Kwiatkowski, Wellcome Trust Sanger Institute/MRC Centre for genomics and global health
Kat - This month, researchers at the University of Oxford and the Wellcome Trust Sanger Institute published results from the biggest ever study looking at how genetic variation in people affects how they respond to malaria infection. To explain more about the study, and why it was urgently needed to push forward the frontiers in the battle against this killer disease, I spoke to project leader Professor Dominic Kwiatkowski, director of the Medical Research Council's Centre for Genomics and Global Health.
Dominic - Well, it's been known for over half a century that human genetic factors influence the outcome of malaria and indeed that malaria has been a major source of evolutionary pressure on the human genome. And there have being a very large number of papers, hundreds of papers, published on different genetic factors that seem to influence susceptibility to malaria and in particular, influence the likelihood that a child who's living in a malaria endemic region who is repeatedly infected with malaria, the chance that the child will die or suffer life threatening complications of that infection.
Because one of the curious things is that in areas where malaria is endemic, whilst everyone has malaria for much over time, only a certain proportion of malaria attacks are fatal. Although a lot of studies have been published on the genetic factors, they've tended to come from relatively small studies. And so, when you look at the literature, you find lots of reported associations. It's very difficult to know which of them whether when you differences, whether those differences are like true biological differences or alternatively, just due to small sample size or to various factors that cause noise or to differences in assay design.
Kat - So, with this study, how many people did you manage to get into it and what did you find?
Dominic - Well, we managed to recruit over 12,000 cases of severe malaria - life threatening forms of malaria - from about 10 or 11 countries, together with our 17,000 population controls. So, the total size of the study was 30,000 individuals. What we found when we tested about 50 known loci that had been reported to determine resistance from malaria, we found that some of them were undoubtedly authentic and gave very strong and consistent effects. It was not any surprise at all that we found very strong effects - protective effects.
Kat - Would this be things like the sickle cell gene that we know about?
Dominic - Exactly. Sickle, it wasn't at all a surprise that it gave a strong effect. I think what did pleasantly surprise us was that that effect was very strong. It gives about 90% protection. It was very consistent across sites and also importantly, it was consistent protection against different sorts of severe malaria.
So children can die of malaria for multiple reasons. One is profound coma, a syndrome known as cerebral malaria. Another is profound anaemia. It turned out that sickle cell gave exactly the same protection against those different outcomes. And that, I think tells us that sickle cell is not protecting against those outcomes per se, but it's doing something for that. It's sort of helping to reduce the infection per se and that in turn is reducing risk of its complications.
The take-home message would be three things. Firstly, there was a bunch of things that we knew about that replicated, I mentioned sickle, also blood group O, also a calcium channel - a gene called ATP2B4 - another thing called G6PD. But then a whole bunch of other things that have been reported before just didn't replicate in a multi-centre analysis. And that really showed the power of doing large studies. It allows you to sort the wheat from the chaff.
The third thing we found which was really important was that some of the authentic loci gave very consistent effects, but others were undoubtedly authentic, but actually get quite heterogeneous effects. Either heterogeneous between different locations, different geographical regions or heterogeneous in their effects on different forms of severe malaria. And the most striking example that was this genetic factor called G6PD deficiency - that's a blood disorder - which gives protection against cerebral malaria. That's to say, coma due to malaria. But actually, makes individuals liable to profound anaemia. So, that was a bit of a surprise to us.
Kat - When you think about the battle between the human genome and the malaria genome, it is a bit of an evolutionary arms race. That tells us that it's a complicated battle going on, doesn't it?
Dominic - Well, it does and I think that this study gives us a foundation for looking at those issues because one of the most important outcomes of that evolutionary arms race that you're talking about is going on in real time. It means that actually, things are changing a lot in real time and so, genetic effects that may operate today in one population may not be operating in a different population at a different time because the parasite population may have changed. But what we have here as a result of this study is a framework that we can study those heterogeneous effects with some level of scientific certainty.
But the problem is that when we just do patchy isolated small studies. We never know whether those differences are due to true biological heterogeneity or whether they're just the sort of noise of doing small studies which aren't particularly well-standardised. Now, we have a framework to study that heterogeneity in considerable detail, and that's an extraordinary advance for us and for the field, we believe.
Kat - That was Dominic Kwiatkowski, from the MRC Centre for Genomics and Global Health.

14:08 - Cattle genome goes large
Cattle genome goes large
The cattle genome has gone large, as an international consortium of researchers announces a global database of cow gene sequences. In a new paper in the journal Nature Genetics, the scientists describe the genomes of 232 bulls and two cows from four different breeds, including Angus beef cows and Jersey milkers. In particular, they focused on key ancestor bulls, which have produced millions of descendants between them. With these new additions, the cattle genome database now comprises more than 1,200 animals of different breeds. And the data can be linked to data on key characteristics such as health, fertility, milk yield and growth, so researchers can spot genetic variations that lead to differences between breeds.
The scientists hope that the database will become the 'gold top' standard reference manual for cow genetics, and will open the door for further research into breeding and farming. Sounds udderly wonderful.

15:05 - Bat flu genes pose low risk
Bat flu genes pose low risk
Bats are well-known carriers of human viruses, such as rabies and the headline-grabbing Ebola. But could they be harbouring other infections too? Researchers recently discovered flu virus-like genes in bats raising concerns that these nocturnal creatures might carry flu viruses, and could transmit them to people. Fortunately, a new study from US researchers published in the journal PloS Pathogens suggests that bats may be in the clear for now. Because they couldn't find any trace of live flu virus particles in the animals, the researchers looked at the flu-like genes in depth, to find out if they came from active, infectious viruses or were merely relics of defunct ancestors.
Next, they synthesised the entire DNA of one of the bat viruses, called Bat09, and put it into human cells growing in the lab. Surprisingly, the virus DNA led to the creation of flu-like virus particles, but these were unable to infect any other types of cells, including human, dog, monkey, pig or bats themselves. This is because the virus DNA doesn't encode crucial proteins in the virus coat that enables it to get into cells. And the scientists think that the bat viruses are unlikely to be able to pick up this ability by recombining with other flu viruses inside bats, due to genetic incompatibility. So for now we only have to worry about rabies and ebola...

16:24 - Dog gene cancer clue
Dog gene cancer clue
Dogs share many aspects of their lives with humans - such as loving a warm fire, a good dinner and regular belly rubs. But man's best friend also shares a darker trait with its owners - the ability to spontaneously develop cancer, which is the most common cause of doggy death. While the genomes of several breeds of dogs have now been sequenced, helping to shed light on many diseases that affect both humans and canines, less is known about the epigenome - the molecular switches that control when genes are turned on or off.
Writing in the journal Cancer Research this month, Spanish researchers have characterised the dog epigenome, in this case from the cocker spaniel, in unprecedented depth. They also found that when the dog cells undergo a change that is critical in the development of cancer, known as the epithelial to mesenchymal transition, there are key epigenetic changes. Importantly, similar changes are seen in human cells when they undergo this transformation, suggesting pathways that are conserved during evolution. Researchers around the world are currently developing drugs that can manipulate the epigenome, which may hold promise for treating cancer not only in ourselves, but in our four-legged friends too.
17:45 - Nazif Alic - Manipulating metabolism
Nazif Alic - Manipulating metabolism
with Nazif Alic, UCL Institute for Healthy Ageing
Kat - We return to our age-old topic, with Dr Nazif Alic at the UCL Institute of Healthy Ageing. He's figuring out if tweaking some of the genes involved in metabolic signalling could help to prolong healthy lifespan - but could it help us live forever?
Nazif - I don't think we'd really be able to live forever, but what we might be able to do is to have a lifespan that's free of chronic and debilitating diseases that occur with age.
Kat - So obviously, this is the big problem in our society. We all live longer. We don't have as many infectious diseases, but there's a long period at the end of life when it's very debilitating.
Nazif - The final outcome of the research that we are doing or we hope the final outcome would be, to actually reduce the severity or reduce the duration of that very unhealthy period that certain people have at the end of their life.
Kat - So, this is about health span rather than lifespan.
Nazif - Yes, hopefully it is about health span rather than lifespan. But lifespan does keep on increasing in our society. So, there's a certain fear when people talk about extending lifespan, but actually, most of the medical advances that we've seen recently as well as advances in hygiene and the way the society functions have resulted in an extension of human lifespan. It's about making that lifespan better.
Kat - So, tell me about the work that you're doing? How are you trying to increase health span?
Nazif - A big part of what I'm doing, or what I have been doing recently, is trying to understand how manipulation of a single signalling pathway can extend this healthy lifespan.
Kat - These are the messages sent inside cells that tell them to do stuff.
Nazif - So, this is the wiring that tells you how to respond to your environment. These signalling pathways tend to be nutrient sensing signalling pathways and the one that we are working on is the insulin/IGF signalling pathway. So basically, the pathway exists so that your body can adapt to changing environmental conditions and in particular, to changing nutritional conditions.
Kat - So, you eat a meal, insulin helps you use the nutrients from that meal - the sugar.
Nazif - Insulin tells our cells what to do with that. The sugar is present and what to do with that sugar. Essentially, it can dictate the metabolism of the whole animal. What the community has recently found is that if you modulate that pathway - I mean if you slightly inhibit the insulin/IGF signalling pathway, in a number of animals, you can extend their healthy lifespan.
Kat - So, you can kind of turn it down and they'll live longer, healthier.
Nazif - Yeah. So you turn it down, they will live longer, they will live healthier, and also, it seems to help with a lot of diseases. When we look at model organisms and we try to mimic human diseases in model organism, reduction insulin signalling can ameliorate those diseases as well.
Kat - You mention model organisms - what organisms are you looking at?
Nazif - So, I'm basically working on the fruit fly, Drosophila melanogaster.
Kat - That's not something I'd associate with being tremendously long-lived.
Nazif - So, they're not very long-lived which helps us do the experiment so we can finish them quite quickly. They do live for some hundred days. We can get them to live 200 days or even longer.
Kat - Presumably then you can actually find flies that do live at particularly longer or healthier lives even within that timescale?
Nazif - Yes, so what's the biggest advantage of Drosophila is that its genetics have been studied for such a long time. There are a lot of very neat tools that we can use to alter their genetic makeup or to alter signalling within these pathways in various specific moments in time during the adulthood of the fly and in very specific tissues. So, we can try to understand in minute detail of what kind of manipulation we need to achieve a healthy long lifespan.
Kat - How do you tell if a fruit fly is kind of feeling old. "My back! I forgot where I've put my car keys." How do you actually look at these flies and work out how they're ageing?
Nazif - So, one of the main outputs that we've been using is really just how long they live. So simply, we can see when a fly is dead or alive. But more recently, we've been trying to look at more detail about their lifespan. So, we can for example look at how well they move. So similar to humans, as flies get older, their movement becomes slower and they find it harder to move.
Kat - Little flies in a Zimmer frame kind of thing.
Nazif - Yes, little flies not walking as fast as they could 20 days before. So, that is a very simple measure of their health and one that we've been using a lot.
Kat - So, you can find that manipulating this pathway, this IGF pathway affects their lifespan and their health span. What now? Where do you want to go with this work?
Nazif - So, the most recent work that we've done is looking at the effector of these pathways. So, we know in modern organisms again that the beneficial effects of this pathway require a transcription factor called FOXO. And this transcription factor essentially rewires the way the genetic information is used, remodels the way the genetic information is used so that the animal lives longer. And we've been trying to work out exactly what genes are controlled by FOXO in which tissues and how they can result in an extended period in life and extended lifespan.
Kat - If I was to have a child, how long would it be before I could give them some kind of FOXO manipulating thing that might make them live a much, much longer life?
Nazif - I don't think we really know how long it's going to be. Hopefully, it might actually be within your lifetime, that you might be able to take something that will prevent dementia or that might prevent type 2 diabetes or any chronic condition that will increase in prevalence as you age. But we don't know. What's quite exciting about this field is now, we've come to the point where we do have drugs that can be administered to people. So, they're FDA approved. They're approved for use on humans. And those drugs in model organisms can extend a healthy lifespan.
Kat - So, this seems to be much closer than perhaps we might think.
Nazif - Possibly, yes. I think the next step will be on one hand, people like me working on model organisms, we need to refine the approaches that can be used, and the other thing that needs to happen in parallel is, for people to find means to see if those manipulations actually do work in humans.
Kat - How long would you like to live for?
Nazif - I think the younger you are, the less you care, but I'm starting to care more and more. I do know that I have a very good amount of risk for cardiovascular disease and I would really like to avoid having a stroke or a very debilitating heart attack. So for me, it may not be that much about how long I live, but just the quality of life as I get older.
Kat - That was Nazif Alic from the UCL Institute of Healthy Ageing.
25:06 - Matt Jones - From genes to brains
Matt Jones - From genes to brains
with Matt Jones, University of Bristol
Kat - On the 27th and 28th of November, top genetics researchers from the UK and around the world will gather at the Royal Society in London for the Genetics Society's autumn meeting, entitled "Genetic Approaches to Study the Neurobiology of Learning and Memory". I spoke to co-organiser Dr Matt Jones from Bristol University to get a flavour of what we can expect.
Matt - Genetics and neurobiology are intertwined on so many levels and across so many timescales that this was a great opportunity to really emphasise how closely linked the fields are. In particular, to emphasise how technology that allows us to manipulate genetics is informing our understanding of the neurobiology of learning and memory. And we also wanted to focus a little bit on uniting the efforts of scientists who work in model organisms like Drosophila, the fruit fly, which are famous for being very genetically attractable, and the other extreme, people who work on mammalian systems, in rodents and in humans. And really, take the opportunity for the two parallel methodologies to inform each other explicitly because often, those researchers do move in parallel and that's a shame. We're not taking advantage of each other as well as we should.
I guess it's particularly timely in light of this year's Nobel Prize of Physiology and Medicine which was awarded for work, understanding in large part, the nature of spatial memory encoding in the brain. And there'll be some good examples of talks that relates genes and gene function and use of genetic technology to that spatial navigation system in the brain. So, that Nobel Prize does a good job of advertising the importance of this meeting.
Kat - Are there any particular talks you're really excited about or techniques you're really keen to hear about?
Matt - So, a good example is the reward system. A lot of our memories are stored because they relate to important or exciting events in our lives and that raises a lot of questions about what's in the brain that takes these events as important or exciting. There's been some great work done in Drosophila dissecting apart their system using optogenetics for example. So, a technique that allows you to control neuronal activity with light which is easily applied in Drosophila because they're small and some are translucent depending on their life stage. But there are people beginning to apply these technologies in mammals now as well. So, the speakers span the globe. Some from the US, some from Europe, some from Japan are really the forefront of driving forward these techniques and understanding of learning and memory.
Kat - If anyone is listening to this and thinks, "Yup! I'm in this field or I'm really interested in this field." How can they come along to the meeting? When and where is it and how can they get to come along?
Matt - That's the easy bit. So, the meeting is at the Royal Society in London from the 27th to 28th of November. You can head along to the genetics.org.uk website and register. We're particularly keen for younger, early career stage scientists to come along, hear from these flashy big wigs in the field and get the chance to chat with them directly, which can be really important in inspiring people in their future careers and experiments.
Kat - And if you're keen to come along, head over to the Genetics Society website and register now.
http://www.genetics.org.uk/Conferences/tabid/84/Filter/0%20GS/MeetingNo/...
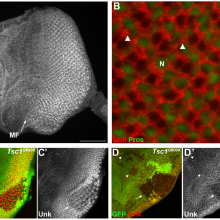
28:18 - Gene of the month - Unkempt
Gene of the month - Unkempt
with Kat Arney
Kat - And finally it's time for our gene of the month, and this time it's looking a bit scruffy - it's Unkempt. Discovered in September this year in the developing photoreceptor nerves in the fruit fly's eye, Unkempt - along with another newly identified gene called Headcase - help to control how the developing nerve cells become specialised into particular roles in vision. Problems with the timing of this process can lead to brain disorders such as epilepsy or autism, but until recently very little was known about the genes that control it. Unkempt and headcase are the first parts of this pathway to be discovered, but there are undoubtedly more. And it's not just flies - the researchers also found that the mammalian version of Unkept is active in the developing eye of a mouse pup in the womb, as well as in developing brain cells, suggesting it might be doing a similar job there too.
- Previous Detection dogs
- Next Will Climate Change Cost the Earth?










Comments
Add a comment