New and Emerging diseases go under the microscope in this week's Naked Scientists, as we discover how new diseases arise, cross species barriers and spread throughout the population. We hear about the origins of HIV, the conditions that create hotspots for new pathogens and a fatal new virus found in Africa. Also, we find out why some people are genetically pre-disposed to AIDS, how the giardia bug changes coats to trick the immune system and why captive elephants live shorter lives than in the wild. Plus, in Kitchen Science, Ben takes the Tabasco Temperature Test to see how mints and chilli affect the temperature on your tongue!
In this episode
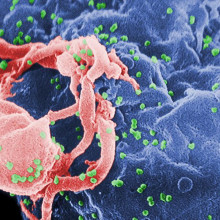
Genetic predisposition to AIDS
Some people might be genetically pre-disposed to developed more severe consequences of AIDS infections, new research has found.
 Writing in the journal AIDS, Stephen O'Brien and his colleagues at the National Cancer Institute, Maryland, sequenced the mitochondrial DNA from 1833 patients infected with HIV in the 1980s before anti-retroviral drugs became available. By comparing the genetic data with the time each of the patients took to develop AIDS the team were able to identify certain genetic signatures linked to progression of the disease. The results could be used prognostically in future to inform HIV-infected patients of their likely outcome.
Writing in the journal AIDS, Stephen O'Brien and his colleagues at the National Cancer Institute, Maryland, sequenced the mitochondrial DNA from 1833 patients infected with HIV in the 1980s before anti-retroviral drugs became available. By comparing the genetic data with the time each of the patients took to develop AIDS the team were able to identify certain genetic signatures linked to progression of the disease. The results could be used prognostically in future to inform HIV-infected patients of their likely outcome.
At the same time, understanding the mechanism that makes some people more susceptible than others may also help to highlight novel approaches to treatment.
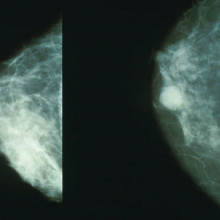
Getting personal with breast cancer treatment
It's becoming increasingly clear that the old saying "one size fits all" doesn't work when it comes to cancer treatment. The more scientists discover about the genetic faults that underpin different tumours, the more it's possible to divide them into sub-types, which may in turn respond very differently to treatment. A good example of this is the breast cancer drug Herceptin, which only works for patients whose tumours have high levels of a molecule called HER2.
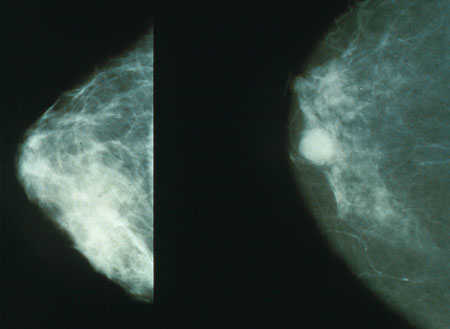 Now scientists in Edinburgh funded by Cancer Research UK have made an important discovery which could help to improve tailored treatment for breast cancer. They've found that a genetic test that's already used in breast cancer patients can predict whether an individual will benefit from certain drugs or not. And we could see it become standard clinical practice within a couple of years.
Now scientists in Edinburgh funded by Cancer Research UK have made an important discovery which could help to improve tailored treatment for breast cancer. They've found that a genetic test that's already used in breast cancer patients can predict whether an individual will benefit from certain drugs or not. And we could see it become standard clinical practice within a couple of years.
The drugs in question are anthracyclines, including one called epirubicin. Epirubicin as an effective drug for breast cancer, but it only works in a certain proportion of patients. But because it can cause quite bad side effects, and it doesn't work for all patients who get it, its use in the clinic has been limited.
So Professor John Bartlett and his team looked at tumour samples from more than 2,500 women who were on clinical trials of epirubicin, to find out if they could identify genetic markers in their cancers that would predict whether they did or didn't respond to the treatment.
In the end, they found that women who had extra copies of chromosome 17 were likely to respond to the treatment. But women with the normal number - that's two copies - were less likely to respond.
And here's where it gets interesting. This extra dosage of chromosome 17 is already known to be involved in breast cancer, because this is the region of the genome that harbour HER2. So there's already an effective test to look for duplications of the chromosome, which is routinely used by doctors to find out if a woman will benefit from Herceptin treatment.
The team are still waiting for the results of one final clinical trial to confirm the accuracy of the test. But if all goes well, we should see this becoming clinical practice within a year or two.
Giardia Gene Shuffling Masks Bug from Immunity
A common gut bug has evolved an ingenious strategy to outwit the immune system, helping to conceal the parasite from immune attack.
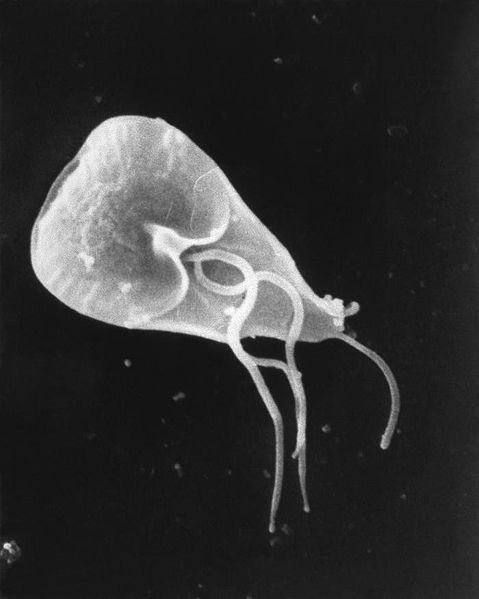 Writing in this week's Nature an Argentinian research team led by Hugo Lujan at the Catholic University at Cardoba found that Giardia lamblia, a major cause of intestinal infections worldwide, has a repertoire of 190 different coat proteins with which it can disguise itself. The organism selects a different coat every few generations, returning the previous outfit to the genomic wardrobe to wear again in the future.
Writing in this week's Nature an Argentinian research team led by Hugo Lujan at the Catholic University at Cardoba found that Giardia lamblia, a major cause of intestinal infections worldwide, has a repertoire of 190 different coat proteins with which it can disguise itself. The organism selects a different coat every few generations, returning the previous outfit to the genomic wardrobe to wear again in the future.
By studying which genes were being turned on in Giardia the researchers found that the parasite uses a mechanism called RNA interference to suppress the action of the other 189 coat genes it's not using. The bug makes short sequences of the DNA-relative RNA, which are the genetic mirror-image of the un-used coat genes. These mirror-image RNAs lock on to their respective coat genes and prevent them from being expressed, keeping them in the closet.
Exactly how the outfit subsequently gets reshuffled to select a different coat remains a mystery, but the team suspect that chemical tags applied to the DNA, called epigenetic markers, could be responsible. Either way, understanding this system will inevitably lead to new ways to combat the bug, which spreads via contaminated water and infects millions of people, worldwide, each year.

Zoo life not so good for elephants
Everybody loves elephants, and they're often a big attraction in any zoo. But some rather sad news in this week's edition of the journal Science suggests that zoo life my not be all that good for them.
 Researchers from the RSPCA, along with other colleagues, did a survey of nearly 800 elephants living in zoos, and compared them to around 3,000 elephnts who either lived wild in Amboseli National Park in Kenya, or were working with loggers in Burma.
Researchers from the RSPCA, along with other colleagues, did a survey of nearly 800 elephants living in zoos, and compared them to around 3,000 elephnts who either lived wild in Amboseli National Park in Kenya, or were working with loggers in Burma.
The results of the -ahem - mammoth survey were shocking. The African elephants in the National Park lived for around 56 years, and even those that were poached or killed by humans lived and average of 36. But African elephants in zoos only made it to an average of 17. And we know this is because adults are dying younger, and the death rates among baby and young elephants were the same across the populations.
Looking at Asian elephants, the team found the same thing. The logging elephants in Burma made it to an average of 42 years old, compared with Asian elephants in zoos that lived to an average of just 19. But in this case, most of the difference was due to higher death rates among newborn baby elephants in zoos, rather than adults dying younger. The researchers think this my be due to traumatic events while the babies are still in the womb, as Asian elephants that are born wild but then captured for zoos live longer on average than those born in captivity.
So what's causing the difference? The RSPCA believe that zoo life causes the elephants stress and obesity, which affects their lifespan. And diseases such as herpes, and tuberculosis, as well as lameness and infertility play a part. The researchers also point the finger at the stress that happens when animals are transferred between zoos - especially when this involves separating mothers and calves.
Although it's causing controversy in the world of zookeepers, this study highlights the fact that more needs to be done for the welfare of zoo elephants. And maybe we should ask if they should be kept in zoos at all.

Paws for thought - dogs demonstrate envy!
Scientists have found that just like humans and other primates, dogs get jealous too!
 The Austrian team paired up pooches and asked the dogs to hold out a paw on demand from the handlers. One of the dogs was given a treat whenever it cooperated, the other dog went unrewarded. Quite quickly the un-treated dogs detected the inequality and refused to cooperate further. That this was down to jealousy was suggested by the fact that when the dogs were tested on their own - i.e. without a second dog being rewarded alongside them - they continued to cooperate despite the absence of any treat.
The Austrian team paired up pooches and asked the dogs to hold out a paw on demand from the handlers. One of the dogs was given a treat whenever it cooperated, the other dog went unrewarded. Quite quickly the un-treated dogs detected the inequality and refused to cooperate further. That this was down to jealousy was suggested by the fact that when the dogs were tested on their own - i.e. without a second dog being rewarded alongside them - they continued to cooperate despite the absence of any treat.
"This shows that dogs have the same basic brain programming for inequity that we do," says study author Friedericke Range, "although it is probably better developed in higher animals like primates that also gauge the size rather than just the sheer physical presences of a reward."
The team suggest that the result is an evolutionary mechanism for fostering cooperation between pack and social animals like dogs, wolves and monkeys. "When you see that someone doing the same work as you is getting more reward it makes sense to refuse to cooperate until equality is restored," says Range.
Does a mint make your mouth cold?
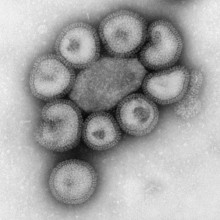
How do viruses cause disease?
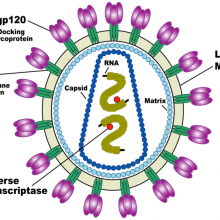
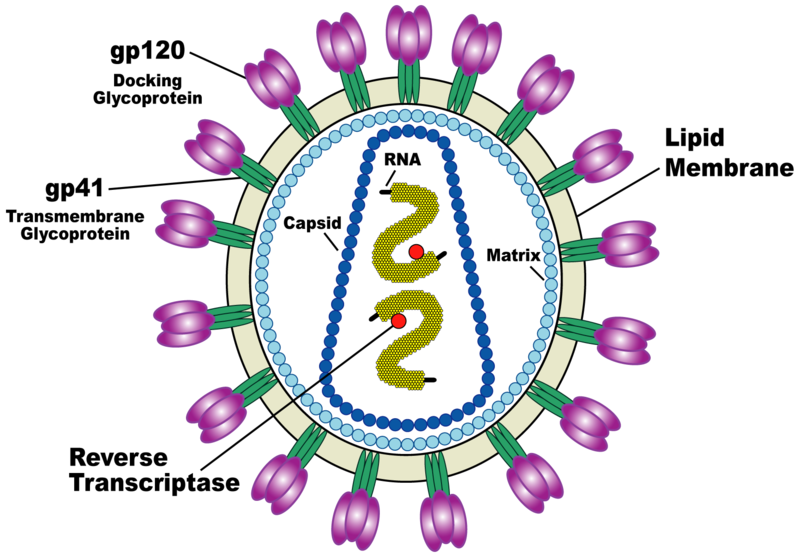
Chris - Viruses are not actually living - they're just an infectious bag of genes; in other words, they are some kind of coat that surrounds infectious material. This can be DNA, which we've all heard of - we've all got that - or its genetic relative called RNA.
Viruses are also tiny: a flu virus is about 1/10,000'th of a millimetre across. That's too small for them to have any of the machinery they need to make new viruses inside themselves, so they need to hijack a cell to do that. There are viruses for plants; there are viruses for animals, and there are even viruses for bacteria [called bacteriophages] - so bacteria can catch a cold too!
The 'flu virus has "receptors", which are like viral Velcro, on the surface of the virus particle. They will lock onto a target cell using these chemical receptors on the surface, which docks onto the cell surface. They then go into the cell. Once they're in the cell they use it like a factory: they take it over and make it produce thousands - or in some cases millions - of new copies of the virus, which come streaming out of the cell. They infect other cells to make more viruses, or they escape from the body and infect a new victim. When they're damaging cells - when they're infecting cells - they can potentially kill them - that's called lytic infection. When they kill a cell that has a consequence for us because if it's a cell in your airway, for example, it might damage the mucosa (the lining of your airway). This means you get inflammation, a rather blocked-up, sniffy nose. Plus, because you've got damage to the lining of the nose, you might get a bacterial infection on top, so they can cause secondary infections.
Kat - Also, something like Ebola. Ebola's a virus where it just breaks down the tissues of the body...?
Chris - Yes, it depends on the "tropism". It depends on what sort of cell the virus targets, because if the virus goes into the cells in your respiratory tract then it can damage the respiratory tract. 'Flu can damage the lungs, and this can cause respiratory failure. Other viruses have a tropism towards other tissues. For example, HIV has a tropism towards cells which have a "CD4" chemical marker on them.
Kat - Those are immune cells?
Chris - Those are immune cells, and so the virus goes into those cells. It can loiter in the cells for a long time before it actually does infect them, but often it can damage the cells and make them die. If you lose those cells, your immune system is disabled.
So there's a whole host of ways in which the viruses can damage different parts of the body. When you've got polio virus, this comes out of your intestines, goes into your blood, and then goes to your spinal cord. It then invades motor neurones, which are the nerves which supply your muscles, and the virus grows in the motor neurones, killing them in the process. This paralyses you.
In summary, it depends what sort of cells the virus is targeted at to determine how likely it is to cause damage to that tissue and how likely it is to have consequences that are clinically manifest...
Why are cold sores so painful?
Chris - Cold sores are the herpes virus - this is herpes simplex. There are two types of herpes simplex: type 1 and type 2. Cold sores traditionally are caused by type 1 herpes. This is a virus that gets into your body, usually by the age of 3 most of us have picked it up by kissing a parent.
In the first manifestation it goes into cells in the mouth and throat and infects those cells to amplify the virus many times over and increasing the infectious dose. The first presentation is you get a sore throat and high temperature; your glands come up around your throat. It then appears to go away for a long time, some people never see it again. What actually has happened when you had a sore throat was the virus was also infecting sensory nerve endings that supply your mouth and throat. These nerve endings then transmit the virus back to the spinal cord or to what's called the dorsal ganglion which is where the cell body for those nerves lives; adjacent to your spinal cord. In the case of the head and neck it goes up to the trigeminal ganglion which is underneath your brain. This is where the virus hangs out just as a small circular piece of DNA for the rest of your life. If you go to the post mortem room and you study people you've died you can find evidence of the virus living in 80% of the population's nervous system.
Periodically and in response to poorly defined stimuli (these can include menstruation, they can include sunburn and tissue trauma - if you get a cigarette burn this can sometimes make it happen) some signal goes back up the nerve, says to the virus, "you're threatened, you need to come out." It reactivates and the DNA turns on again, makes fresh virus particles inside the cell. They come back down the nerve cell to a patch of skin that nerve cells supplies, the virus comes out of the nerve, onto the skin, raising the skin cells producing an infectious lesion. That's a cold sore and the point of this is the virus uses the cold sore to infect another person - when you kiss someone you're infectious. That's how the virus gets around but the rest of it is hiding inside your nervous system. When it reactivates in that way it can damage the nerve it's in and those nerves very often are pain nerve fibres. They get stimulated by the activation of the virus and that is excruciatingly painful and it can persist for a very long time.
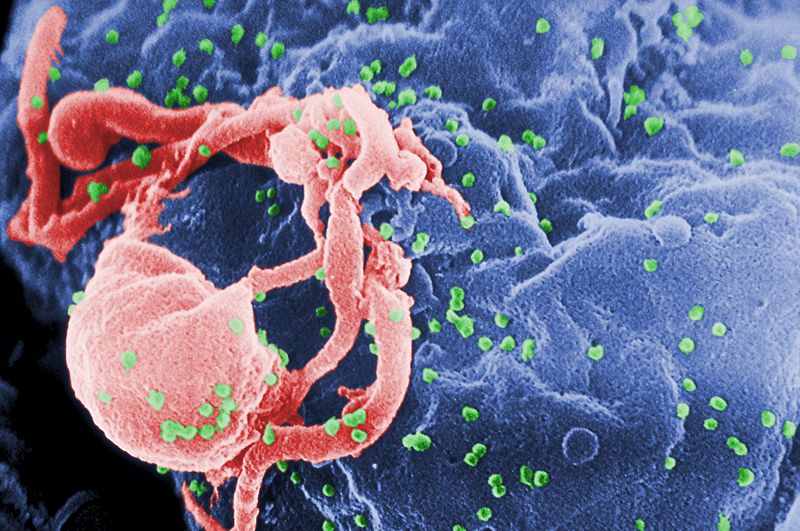
24:48 - The Origins of HIV
The Origins of HIV
with Mike Worobey, University of Arizona
Chris - When did HIV first pop up in humans?
Mike - Well this study that we recently published we used some archival samples. We went into a time machine back to 1960 and sequenced a variant of HIV from that time period. From that vantage point it became possible to home in on when we think the pandemic strain first started circulating in humans. It's pretty early. The virus may be celebrating its hundredth anniversary in humans. Our best guess is about 1908 + or - 20 years.
 Chris - First of all, let's take a look at how you came by these samples. What was the work that led up to being able to say I've got samples from 1960?
Chris - First of all, let's take a look at how you came by these samples. What was the work that led up to being able to say I've got samples from 1960?
Mike - Actually it kind of traces back to last time we talked four years ago. It was regarding some work I'd done collecting virus samples from chimpanzees in the Congo so that's one way you can go about investigating the origins of the virus. You can look at the modern counterparts in the reservoir species. At that time, actually even before that, I started thinking about ways that you could get at it. The obvious thing was to try to get human samples that go really far back in time and the state of the situation at that time was we had one little glimpse. A fragmentary sequence from one HIV patient from 1959 and then there was no sequence at all until the late 1970s, early 80s.
Chris - That 1959 sample was a blood sample that someone had luckily stored in a freezer for years, wasn't it?
Mike - Exactly, it was something that was collected for other reasons. It was genetic research to do with malaria but someone had the foresight to save those samples. Central African blood samples that go back that far in time are hard to find. I think most people just kind of gave up. They thought when we got the 1959 one we're not going to find any more, that this is all we have. What I thought about was are there other kinds of samples that aren't in freezers and aren't so delicate? I turned to paraffin embedded, wax tissue specimens, biopsies and autopsy specimens. They're routinely taken in hospitals and clinics around the world and put in a cellar and archived. It turns out it's kind of painstaking but you can get gene sequences out of these things.
Chris - Who happened to have some of the right sorts of paraffin-embedded tissue you could study?
 Mike - The one that yielded this sample from 1960 was actually stored in Kinshasa, the capital of the Congo at the University of Kinshasa. What i did for quite a few years actually was just put out feelers and do some work and talk to a lot of old Belgian scientists and physicians who had connections who had either done work in the Congo back in the day or knew people who did. Eventually turned up many old samples like this and, like I said, they were samples that had sat in Kinshasa all this time that yielded this sequence.
Mike - The one that yielded this sample from 1960 was actually stored in Kinshasa, the capital of the Congo at the University of Kinshasa. What i did for quite a few years actually was just put out feelers and do some work and talk to a lot of old Belgian scientists and physicians who had connections who had either done work in the Congo back in the day or knew people who did. Eventually turned up many old samples like this and, like I said, they were samples that had sat in Kinshasa all this time that yielded this sequence.
Chris - So you go to old human tissue, you go to 40+ years ago and you can find HIV in that. What does that tell you about where HIV came from and how do you wind back the genetic clock to find out when it first popped up in humans?
Mike - The first thing that tells us is you can compare the 1960 Kinshasa sequence to the 1959 Kinshasa sequence. It's like having a human genome, it's great and tells you lots of things. As soon as you have a chimp genome to compare against it there's a whole lot of other things you can tell by comparing. Once we have these two viruses side by side we could see that they were pretty surprisingly divergent. Even at that early date in 1960 a direct comparison of that early sequence indicated these things had spent a long time evolving away from their common ancestor: decades and decades. The other thing that we could do now is plug those early sequences into a more sophisticated analysis using more that 100 sequences worldwide. They really helped calibrate the molecular clock of these viruses and allowed us to estimate with a lot of precision when the virus started spreading in humans. Like I said, it's many decades before 1960.
Chris - but something must have happened 100 years ago in that part of Africa to make that HIV appear. The closest relative of HIV is SIV: the virus which you find in chimpanzees. What was going on in that part of Africa that meant this chimpanzee virus got into people?
 Mike - This is one of the coolest things about the finding. There are all sorts of SIVs from different primates that are circulating out there. The big question is if these things have been around why didn't they cross in earlier or later? Why now, at this point? When we matched up the timing of these genetic analyses of the viruses against what was happening in that epicentral region of Africa for me it was a surprise. I didn't realise how little urbanisation there was in the region until that time. Basically, in the whole area around southeast Cameroon where we think the chimp virus produced the pandemic strain of AIDS entered humans there were basically no cities at the turn of the 19th century. It wasn't until that time that places like Kinshasa were established as colonial administrative trading centres.
Mike - This is one of the coolest things about the finding. There are all sorts of SIVs from different primates that are circulating out there. The big question is if these things have been around why didn't they cross in earlier or later? Why now, at this point? When we matched up the timing of these genetic analyses of the viruses against what was happening in that epicentral region of Africa for me it was a surprise. I didn't realise how little urbanisation there was in the region until that time. Basically, in the whole area around southeast Cameroon where we think the chimp virus produced the pandemic strain of AIDS entered humans there were basically no cities at the turn of the 19th century. It wasn't until that time that places like Kinshasa were established as colonial administrative trading centres.
Chris - So you think it was people moving in and establishing cities that got the thing going?
Mike - I think it's almost too much of a coincidence to dismiss that what I think happened was, HIV is a fairly poorly transmitted virus human-to-humans. You have to set the stage for it to survive and create a chain of transmission otherwise viruses entering from chimps might get into one or two people but then die out.
Chris - So what do you think happened?
Mike - Well, with the advent of cities in the region cities make life a lot easier for HIV. They bring people into high densities. You often have high-risk behaviours like prostitution that facilitate the movement of the virus from person to person. We talk about this magic number of viruses: a basic reproductive number of one. This means a virus which your average infected person infects one other person will persist in the population. If it's below that number it will become extinct. The advent of urbanisation may have pushed that magic number above one for HIV.
Chris - Briefly, I think one of the other things to mention might be people immunising people. When they set up cities weren't they exploring vaccines in those early days? Could that also have been how it happened because people weren't so rigorous with sterilising needles as we are now?
Mike - It's definitely one of the things that have been discussed as a possible player in the establishment of the virus. My thinking is if you actually look at the timing of those events people like Preston Marx have talked about the spread of disposable needles. That's actually in a large part after the timing that we're pointing to. The bulk of the transmission of the virus in sub-Saharan Africa today is through sexual transmission. I don't see any reason to think that the establishment of the virus had things to do with things like dirty needles. They probably play a minor role in transmission but I don't think they're the key.
33:44 - Hotspots for Emerging Disease
Hotspots for Emerging Disease
with Kate Jones, Zoological Society of London
Kate - Emerging infectious diseases are diseases that have basically increased in incidence, impact or range, newly-evolved like multi drug-resistant TB or malaria or entered the human population for the first time like SARS or Ebola or HIV/AIDS.
Meera - What exactly are the factors that cause these diseases to emerge in the first place?
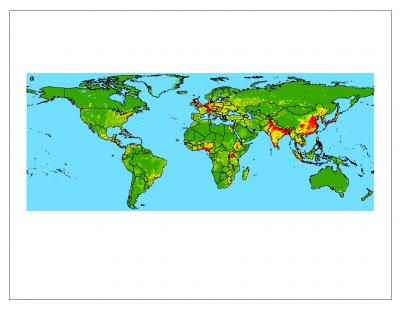 |
| Zoonotic pathogens passed from wildlife to people, from lowest occurrence (green) to highest (red). © Nature |
Kate - It's usually because there's a change in the transgression dynamics between and within host populations. You have populations of humans,wildlife and domestic animals all interacting. Changes which change those dynamics between these populations and within host populations cause these diseases to enter the human population for the first time.
Meera - What are the actual changes that happen?
Kate - It could be increase in intensification of agriculture. You get more contact between domestic animals and humans. It could be going to ranges of wild animals that people didn't do before because of bush meat hunting. You're exposed to a different disease. Diseases and pathogens don't want to kill their host because they're evolving with them. It's when you get a change in these transmission dynamics and suddenly it's new for the pathogen. That's where it causes the problem because pathogens don't actually want you to die because they're living in you.
Meera - When it comes to emerging infectious diseases there are obviously lots of different types. What are the factors that influence these different types?
Kate - We thought originally about emerging infectious diseases all as one thing. Then it because obvious that there were different drivers for the different things. We cut it up into broadly different types: drug-resistant emerging infectious diseases like drug-resistant TB, vector-borne diseases such as malaria and then we thought about zoonotic. This is diseases from wildlife that entered the human population such as Ebola, AIDS or SARS. Those are the broad categories that we use.
Meera - So what were the different drivers for the different disease playing more crucial roles in them?
Kate - Human population density is absolutely critical for predicting the probability of an emerging infectious disease because you've got more people, more chance of infection. For drug resistant ones human growth is actually more important. The rate of population growth is more important or, for example, zoonotic pathogens we were finding that human population density is really important but the other driver that was equally important was the distribution of possible hosts. In this case we used wildlife biodiversity.
Meera - Earlier this year you managed to create this map that basically showed hotspots on a global scale of where these diseases are occurring more often.
Kate - We took a really innovative, multi-disciplinary approach. I study broad-scale, by diversity pattern so I'm not an epidemiologist at all. I was working with people who were but the problem with the field as it stood before we tried to do this is that people were looking at single diseases and not taking a really broad-scale approach to think well, what are the processes behind those patterns?What we did was we tried to think about an emerging infectious disease and not just the disease but the event. The first time in space and time that that pathogen entered the human population. We tried to model the human event because we wanted to work out what were the factors that were affecting that event happening. Maybe we could predict that event in the future.
Meera - Are there particular examples of diseases that you managed to think about the factors for then?
Kate - Yeah so we tried to do this in a general approach. A few examples would be Nipah/Hendra virus in Malaysia. This is a zoonotic, a disease from wildlife. What happened was an intensification of pig farming in the region and so people put pigs natural forest for the first time and there was an interaction between the bats and the pigs in the forest. The pigs were eating fruit that the bats had eaten. Bats have Nipah virus naturally and they cope with it in their populations. Pigs, however, have never been exposed to that. The humans which were handling the pigs had never been exposed either. You had an emerging infectious disease event of this really nasty disease.
Meera - Is it safe to say that we're moving closer and closer to animals, it's causing a big change in the emergence of these diseases?
Kate - That's a really good question. That's what we were trying to try and answer. Our first result coming out of this huge database was that the incidence of emerging infectious diseases has actually increased over time. What we then did was plot it spatially. Normally in my biodiversity studies you find a lot more biodiversity, a lot more things, in the tropics. Now with emerging infectious disease events the opposite is true. You get more in the north and more in the south. We were thinking that possibly that shows you that biology is important but there are other socio-economic factors involved that we needed to model. Quite frighteningly since 1940 the hotspots of diseases have been in the really big capital cities of the world: London, New York and Tokyo. Maybe one possible explanation is that you put more money into antibiotics and you're getting a lot more emergence of these diseases which are drug-resistant. The other interesting factor was that if you look at the zoonotic patterns, they're affected by human population density but also the density of other wildlife in the region which makes perfect sense really. It's not just that you've got high population densities of humans or wildlife. It's the interaction of those two things. In a way it gives added value to preserving wild areas, to keeping them wild. We're preventing horrible emerging infectious diseases coming and entering the human populations.

45:27 - What is tryptophan? Does eating turkey really make you sleepy?
What is tryptophan? Does eating turkey really make you sleepy?
We put this to John Fry, consultant in food science, nutrition and dietetics.
There's been a question about sleepiness caused by tryptophan in turkey and this is a popular myth in the United States that a feeling of sleepiness arises after the Thanksgiving meal and it's caused by the Thanksgiving turkey having a high content of a substance called tryptophan. Tryptophan is an amino acid, one of the building blocks of protein which means that pretty much all proteins contain some Tryptophan but turkey's not unusual in its tryptophan content. It has about the same amount as chicken or beef. Tryptophan is involved in the desire to sleep after a heavy meal but only indirectly. The root cause of the drowsiness but only indirectly. The root cause is the large carbohydrate intake that usually accompanies a celebratory festive meal. All those roast potatoes, the stuffing, not to mention sugar-rich puddings. They all result in a burst of insulin in the blood stream as the body tries to cope with this influx of sugars. One of the side-effects of this secretion of insulin is that tryptophan gets into the brain more easily and once there part of the tryptophan is transformed into a substance that's called melatonin. Melatonin is a hormone involved in sleep regulation and it can encourage sleep. But really it's the carbohydrate in a heavy meal that triggers drowsiness and tryptophan is just a bit player in the biochemical consequences of over-indulgence in carbohydrates.
Why were colds so bad for indigenous inhabitants?
Kat - Presumably this is when you get stories of explorers who go to isolated communities and then all the people get really bad illnesses.
Chris - That's true and the reason is that isolated populations in distant geographical areas which are, in other words, when we say isolated were cut off from the mainstream viruses circulating and other infections circulating where there were big populations in Europe. Those individuals didn't have the same selective genetic pressure to have a more powerful immune system. If you compare the number of genes in people who are native American Indians and South Americans had for presenting information from the immune system to itself - so when a cells gets infection it presents various aspects of what it seeing washing round the body to the immune system in these things called HLA genes - well the native populations had far fewer of these than the Europeans did. Viruses that breed in Europe are much more virulent under those circumstances because they have to have all these immune evasion strategies built-in in order to outwit the much more powerful immune presenting abilities of the European population.
Kat - So they were really nasty.
Chris - They tend to be more powerful, yeah. If you take them to a population that are more vulnerable they haven't seen them before. Therefore there's no pressure on that population to evolve these defence mechanisms. Those populations are like a souped-up virus and it's overkill. They don't need to be that powerful and it makes people much iller than it would otherwise be. Earlier on in this programme we talked about how diseases spread from animals into people like the rats giving arena virus-like things to people in South Africa. It can work two ways because scientists published a paper in Current Biology where they showed that chimpanzees are dying of colds that we give them. There's a fairly recently uncovered cold virus called metapneunomvirus that we diagnose quite often in people, in humans. It causes a heavy cold and if you have children it causes wheeziness. It tends to be self-limiting and goes away. When people started studying chimpanzee populations in Africa and noticing that they were periodically getting sick and having mass die-offs they found metapneumovirus. Maybe the chimps were giving the metapneumovirus to the humans. Actually, there was in South America a few years ago there was a form of metapneumovirus with a genetic signature in it that was very specific to that strain and this then started cropping up later in Africa. This told researchers that in fact we're giving our viruses to chimps. When you put our viruses that give us a cold into a chimpanzee very often it can kill it. There's the evidence that viruses are programmed to cooperate with the immune system of the host they naturally affect. If you put them in a different context they can be much worse because they are over-optimised for that particular host.
Can we target the unchanging portion of virus’ genetic material?
Chris - What he's getting at is that some viruses change bits of themselves like the flu, changes its H genes (haemagglutinin genes) on the surface coat of the virus looks a little bit different. This is called genetic drift and this is down to genetic mutation when the RNA copies itself it makes mistakes and this translates into a slightly different structure for the virus. This is useful to the virus because it makes it look different so the immune system struggles to recognise it a second time.
Some bits of the virus do such a crucial job that they can't afford to change because if they did they would impair themselves. There are some un-variable bits of the virus that don't change. What he's asking is could we exploit that to make better treatments for viruses?
The answer is yes.
A good example is in flu itself. There was a recent study that was published in PNAS where researchers looked at the way in which a flu vaccine worked. Flu vaccines are based on the haemagglutinin coats on the virus. You make a coat on the virus, put it in a egg, you get some virus shrapnel and you can inject that into people. If you watch how the flu changes over time a vaccine that works against one type of flu might not work against another. If you get lots of examples of flu virus, compare the genetically you can find elements of the surface coat of the virus that have never changed over that time. If you make a vaccine out of that then in the case of the paper I'm referring to these people did it with a DNA vaccine. They just injected the DNA from that little bit of the virus coat. That hadn't changed and it was very effective against a broad repertoire of viruses so that might be a better way of doing it.
Another good example is HIV which has to bind onto CD4 receptors on our immune cells. If the virus were to mutate that bit of itself too much then it wouldn't be able to infect anymore because it wouldn't be able to recognise the target. Researchers are now looking at ways to specifically target the structure on the virus which the virus keeps very hidden but which doesn't change in order to block HIV.
Where do sexually-transmitted diseases originate?
Chris - Wherever there is an ecological niche something will spring up to occupy that niche. When you've got an opportunity for spread to occur and you've got humans coming into close contact - it doesn't matter if you're kissing someone and spreading Epstein-Barr virus that causes glandular fever or you're talking about other viruses and bacteria that can spread sexually - there is an opportunity for spread to occur and you have a certain environment which is coming into contact with another environment. That means there's an opportunity for anything that can tolerate those environments or exploited to spread. Because you have those opportunities and that contact anything that becomes able to exploit that environment will do so. That's just nature. If as viruses and bacteria evolve and change to exploit those environments so they become specialised to do just that. If you look at Neisseria meningitides, the bacterium that causes meningitis, it's got a relative called Neisseria gonorrhoeae. In other words the same kind of family, the same species of bacteria can cause bacteria down there and meningitis up there but if you move one to the other location they can cause a sort of similar infection and similar manifestation in both places. If someone gets Neisseria meningitides down there then instead of meningitis they can get a bit of an infection. Kat - Do you think we are winning against bugs or they're winning? It seems to be an evolutionary arms race. Chris - Ever since life got started there's been a furious arms race going on with each organism on the planet trying to secure its own patch of turf because there's only so much energy to go round. There's only so much chemistry to go round and if you've got it someone else hasn't. As populations increase you will see competition between one group of organisms or one entity trying to rob energy or exploit the system better than anyone else. In order to keep up everyone has to change. It's a bit like radio station changing to digital. You found people buying very expensive equipment that could make your sound a bit louder than the person who was running the radio station next door. The radio station then bought a bigger box that would make them louder than everyone else. Before you know if=t you've got this compression war going on where people are seeking to be louder than everyone else. Now people are saying now we've got the audio so loud we're going to make our station distinctive by being quieter than everyone else. It's the same with microorganisms. They're going to change and adapt in order to shift and exploit the circumstances as they exploit themselves. We're never going to have an end to this arms race.
Kat - Do you think we're due another massive flu pandemic or Black Death plague?
Chris - History has a habit of repeating itself there's absolutely no doubt about that. We know that flu pandemics come roughly every 30 or 40 years. There's no doubt that there will be another one. Whether there will be a major manifestation or not we don't know because the Earth is a very different place now than it was in the past. We've got half a million people airborne in aeroplanes at any instant in time. No city is more than 24 hours from any other city which means that's less than the incubation period for most infectious diseases. At the same time we have more people on Earth - nearly 7 billion than we've ever had before and living in higher density than we've ever had before. This is the time, if there's ever going to be one, that we're going to see some kind of infectious outbreak.
- Previous Jealous Dogs and Lost in Space
- Next Neon: Chemistry in its element










Comments
Add a comment