How do we know that ants count their footsteps? We'll find out in this Naked Scientists Question and Answer show, as well as ask if rubber soles really protect you from electric shocks, if hair will clean itself when you don't, and why a layer of shaving foam stops the mirror from steaming up. Also, the spores that fly on smoke rings, new ways to capture carbon, pain free vaccine patches and the vaginal gel that could block HIV transmission. Plus, Meera investigates vintage computers and in Kitchen Science, Dave discovers how popping candy gets it's pop!
In this episode
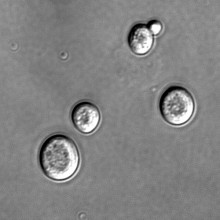
- What Makes A Cell Grow Old?
What Makes A Cell Grow Old?
A family of proteins has been identified that may limit the number of times any cell can divide, according to research published in the journal Nature Cell Biology this week.
Most cells seem to have an in-built limit to the number of times it can divide. From human cells down to single-celled yeast, they seem to limit themselves to only 20-30 divisions. Cell aging is thought to be related to the build up of toxic agents, but the mechanisms are not well understood.
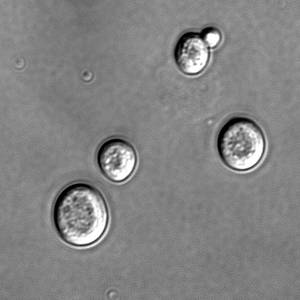 Now, Rong Li and colleagues in Kansas have identified a group of chemicals that may be responsible for limiting the reproductive lifespan of a cell.Yeast cells multiply by growing and dividing. A mother cell will create more of the components it requires, and then a daughter cell will bud off from the mother.
Now, Rong Li and colleagues in Kansas have identified a group of chemicals that may be responsible for limiting the reproductive lifespan of a cell.Yeast cells multiply by growing and dividing. A mother cell will create more of the components it requires, and then a daughter cell will bud off from the mother.
Each cell can only do this 20-30 times, and so far this has been put down to the build up of damage to DNA.
Multidrug resistance, or MDR, proteins are involved in transporting chemicals across the plasma membrane in yeast, and are inherited from the mother cell. Oddly, this inheritance is asymmetric - rather than a batch of new proteins being produced, then all the proteins split, the newly formed proteins preferentially move into the daughter cell.
Over subsequent divisions, the mother cell will keep hold of the 'old' MDR proteins, which may become damaged and lose function. As these proteins have crucial roles in metabolism and dealing with stress, this will lead to a gradual reduction in fitness of the mother cell, and a limit to how many times it can divide.
Supporting this hypothesis - yeast cells lacking certain MDR genes show a reduced replicative lifespan, while introducing extra MDR genes increases the lifespan.
It's now important to see if membrane transporter proteins show the same asymmetry in stem cells, which could limit the potential of stem cells, and to see if the same proteins play a role in aging in multicellular organisms - including us.

01:34 - Spores Travel on Mushroom Clouds
Spores Travel on Mushroom Clouds
Moss spores get extra lift from the same process that generates smoke rings and mushroom clouds, according to new research published in the journal Science this week.
Dwight Whitaker at Pomona College in California and Joan Edwards at Williams College, Massachusetts, realised that the spores of Sphagnum moss couldn't possibly reach as high as they do by ballistic propulsion alone, and set out to find the moss' trick.
 |
| Habit shot of the low-growing Sphagnum moss showing reproductive heads with capsules raised above the mat by pseudopodia. Some capsules are round and not yet exploded, and some have recently exploded (cylindrical). Sphagnum acts like a sponge - holding water by capillary action between its leaves and in specialized water-holding cells. It acidifies the water creating anoxic acidic areas where decomposition is slow. Over the millennia, Sphagnum has sequestered more carbon and any other plant genus - thus it plays a critical role in the global carbon cycle. © Image courtesy of Joan Edwards |
Sphagnum moss is thought to store more carbon than any other plant genus, and covers around 1% of the Earth's surface. In order to distribute its spores, it fires them upwards into a turbulent patch of air, where they can be picked up and transported on eddies and breezes. To take advantage of the breeze, the spores have to be very light, and have a terminal velocity of just 5 millimetres per second. This low terminal velocity creates a problem when getting through a layer of still air directly above the moss, where the spores will rapidly decelerate.
The spores develop in the top half of a 2mm spherical capsule, the bottom half of which is hollow. Each capsule contains from 20,000 to 250,000 spores. When the conditions are right, these capsules dehydrate and become cylindrical - in the process vastly increasing the air pressure inside. Eventually, the capsule fails, and the internal pressure fires both the spore, and a puff of air, upwards.
This puff of air creates a vortex - a self sustaining ring of rising air like a smoke ring or mushroom cloud - which is strong enough to carry the spore far further than it would otherwise go, and up high enough to catch the turbulent air and be carried away. High speed imaging confirmed their hypothesis.
We know of a few species that take advantages of vortices like this - bees and jellyfish for example - but this is the very first example of vortex use in plants.
 |
| Sequential still frames from a video filmed at 10,000fps. Each frame is 1/10,000 of a second. The mushroom cloud with a trailing wake is clearly visible. © Image courtesy of Clara Hard, Joan Edwards and Dwight Whitaker |

03:47 - Electrolysing carbon-dioxide
Electrolysing carbon-dioxide
Carbon capture is often suggested as a way to reduce carbon emissions, so far it has normally just meant collecting carbon-dioxide and hiding it somewhere - normally underground. But the ideal solution would be some way of converting the carbon-dioxide back to carbon, the natural way of doing this is of course to harvest sunlight with plants and then some of this biomass is sequestered. But the highest efficiency plants only convert a few percent of the sun's energy into biomass, and if you want to use it as fuel, the best conversion efficiency is less than 0.5%.
 A more radical rethinking of this problem has been to use solar electricity to electrolyse - or split - the carbon-dioxide into carbon and oxygen. Whilst in comparison to photosynthesis this is good, it still wastes large amounts of solar energy, as most solar cells can't use the huge amount of infra-red thermal energy in sunlight.
A more radical rethinking of this problem has been to use solar electricity to electrolyse - or split - the carbon-dioxide into carbon and oxygen. Whilst in comparison to photosynthesis this is good, it still wastes large amounts of solar energy, as most solar cells can't use the huge amount of infra-red thermal energy in sunlight.
Stuart Licht and collegues from washington DC have come up with a way of increasing this efficency. They are electrolysing carbon-dioxide dissolved in lithium carbonate, so still using electricity, but they have discovered that if you heat the system to 950C you need 40% less electricity to split the carbon-dioxide.
The really neat thing about this is that you can use the infra-red part of the solar spectrum to heat up the electrolysis cell while still using a solar cell at maximum efficiency - in fact slightly better than normal, as there are less overheating problems. Using the most efficient solar cells available, they think they can use between 30 and 55% of the sun's energy to either produce carbon or at a slightly higher temperature carbon-monoxide.
This could either be used to sequester carbon, or the carbon-monoxide could be used as a feedstock to produce hydrocarbon-fuels or hydrogen.
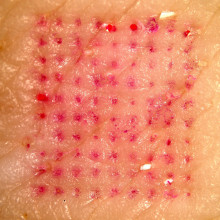
06:12 - Painless vaccine patches
Painless vaccine patches
For most of us, vaccinations are a normal, if not very pleasant, part of life. But what if there was an alternative? What if it was just as easy as putting on a sticking plaster? Researchers from Georgia Institute of Technology and Emory University reported in Nature Medicine this week that that might be closer to reality than we think.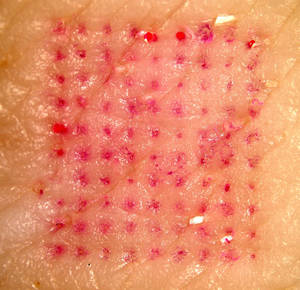
Sean Sullivan and his colleagues have developed a patch that delivered an influenza vaccine to mice. They compared the effectiveness of this patch with a more traditional intramuscular vaccine and found that it protected mice equally as well against the otherwise lethal flu virus. Even more exciting, 90 days after vaccination, the patch-vaccinated mice were found to activate the immune response to influenza more promptly and to clear the virus more quickly from lung cells.
The patches each held 100 tiny needles just 650 microns long that dissolve painlessly from the patch into the skin, taking with them their load of freeze-dried deactivated influenza virus. Vaccinating in this way takes advantage of the skin's special immunological properties and may explain why the patch seemed to provide even better protection than the intramuscular method by some measures. Some drug patches are already in widespread use, for example nicotine and contraceptive patches but what distinguishes these new microneedle patches is that they are able to deliver much larger particles such as viruses, as opposed to just small molecules like nicotine.
This work offers an exciting glimpse of what vaccines could be like in the future. If we no longer rely on traditional intramuscular vaccination, the risk of spreading disease through the re-use of contaminated needles, which is a common problem in some developing countries, would be eliminated. In addition, the patches allow the vaccine to be given with little or no medical training making large-scale vaccination programmes cheaper and easier. And on top of all that, getting life-saving protection against many infectious diseases could soon be pain-free.

09:42 - Solid Li-ion batteries
Solid Li-ion batteries
Lithium ion batteries are now pretty much the standard high performance battery, they have a relatively high capacity and are becoming more affordable. They are used in most laptop and phone batteries and have recently been used in the Tesla Roadster sports car. However there are still major issues, the cathodes change shape and degrade over time, causing an electric car battery to only last 2-3 years. This is related to the batteries using a liquid electrolyte between the active electrodes, which also requires lots of infrastructure to hold everything apart, avoid short circuits, and keep the electrolyte in. One solution to this problem is to use solid electrolytes, which can allow ions to move whilst remaining solid, unfortunately these normally have to be made using vacuum deposition systems like computer chips, which are very expensive.
 A company called Planar Energy has developed a process for making these solid state batteries using printing techniques which are far cheaper than the deposition. They have also apparently increased the conductivity of the solid electrolyte to be comparable with the conventional electrolytes and made high quality electrolytes by using chemical self assembly processes.
A company called Planar Energy has developed a process for making these solid state batteries using printing techniques which are far cheaper than the deposition. They have also apparently increased the conductivity of the solid electrolyte to be comparable with the conventional electrolytes and made high quality electrolytes by using chemical self assembly processes.
Apparently the batteries have a capacity similar to the best high performance conventional batteries, but should survive longer, have three times the capacity, lose energy more slowly and be safer than conventional Li-ion batteries. Even if they don't manage the half the current price per kW hour, they do sound promising.
12:16 - Gel offers protection against HIV infection
Gel offers protection against HIV infection
with Salim Abdool Karim, University of KwaZulu-Natal, Durban
Chris - Also in the news this week, a vaginal gel which contains the anti-AIDS drug called tenofovir has been found to reduce transmission rates of HIV amongst women by up to 50%. To explain a bit more about the study, which was carried out in South Africa, Salim Abdool Karim is from the University of KwaZulu-Natal is with us now to tell us what he did. Hello, Salim. Thank you for joining us. First of all can you set the scene for us; how big a problem worldwide is HIV? What's the scale of the problem?
Salim - Globally, we have a good idea of what's going on with the HIV epidemic. We know in 2009 that there are 33.4 million people living with HIV and that during 2008, there were 2.7 million new infections, and about 2 million deaths. So globally, the epidemic continues to grow, although it's growing more slowly now than it was some five years ago.
Chris - And just totting those numbers up in my head, that would mean something in the region of 7,000 people a day must be dying of HIV and 7,000 new infections every day. So we need to sort this out. A trial in Thailand suggests that vaccines are only at best 30% effective, so you've been taking a slightly different approach. These gels, how do they work?
Salim - There've been several gels that have been made, and foams and sponges as well, and they've been impregnated with different kinds of chemicals and called microbicides. And the underlying hypothesis is that these chemicals, when put into the genital tract, into the vagina, would prevent HIV from causing infections. Well up to now, in the past 15 years or so, there've been 11 trials of six candidates. None of which has been shown to work.
So, it's been a pretty difficult time in the field to find something that could prevent HIV infections. So we took a different approach. We decided to go with an anti-retroviral drug which is very widely used for treating AIDS and this drug is called tenofovir. It is a standard part of many cocktails of three drugs that are used to treat AIDS. So we put this drug into a gel formulation and we put it into single-use applicators and we did a study of 889 women in South Africa where they were asked to use this gel within 12 hours before sex and within 12 hours after sex. And what we found was that in the half of the women that used the tenofovir gel, there was 38 HIV infections, compared to 60 HIV infections in those women who were using an identical placebo gel. So that translates to a 39% protective effect of this gel.
Chris - And over what period of time were you studying? How long did you look at?
Salim - The first women were enrolled in May of 2007 and we completed the study in December of last year so 2 ½ years in total.
Chris - So if you extrapolated this to making this available to every exposed individual, let's just take Africa as an example, how many cases of HIV do you think you could prevent per year with this strategy?
Salim - We've done some mathematical modelling, creating a hypothetical scenario, as if we were implementing this in South Africa where we have very good data, and we emulated the kind of adherence we got within the study. So within the study, for those women who used the gel most consistently, they had 54% protection and those women who used it least consistently, less than 50% of their sex acts, we saw 28% protection. So what we did is we modelled that. We said, "Okay, what if 40% of the women used it in a consistent way as we observed in our study and 40% used it in an inconsistent way as we observed in our study." And if we did so, then over the next 20 years, we estimate we could prevent 1.3 million new HIV infections and avert just over 800,000 deaths, just in South Africa alone.
Chris - And what cost would that come out at? How much would it cost to implement that?
Salim - Now that's a bit more difficult to calculate. We know the actual cost of the gel is negligible. For the study where we only made a small quantity so we didn't benefit from scale, the actual gel costs about a cent or 2 in US cents. So the cost is not in the gel, but it's in the applicator, the wrapping, and the packaging, and so on, so that for the trial, each application costs us $0.32. If it's produced to scale, we would estimate that it will be substantially less and we worked out that even at the current price of $0.32, it is still cost-effective to implement because the cost of treating somebody who develops HIV infection is so high that even at this cost, to use the gel is more cost-effective than to allow women to get infected.
Chris - Well that brings me on to my last point, which is that the way we treat HIV is with triple therapy. We give people a combination of drugs so that the risk of the virus becoming resistant is reduced. You're using a single agent in this gel. Is there not a risk that we could end up eroding the ability of this agent to prevent HIV because people are being exposed to this as monotherapy?
Salim - Yes. It is a hypothetical concern and certainly, before the results of our study has been looked at in many different ways. These are the first data that have come out now where we can look at whether any of the 38 women who became infected while using tenofovir developed resistance, and the answer was no. We found no evidence of tenofovir resistance.
Chris - Indeed. Salim, thank you very much. That's Salim Abdool Karim who is from the University of KwaZulu-Natal in Durban, South Africa, and he is reporting there the study that he's just published in the journal Science, looking at the use of a vaginal gel which contains the agent tenofovir which seems to be able to reduce by a significant margin the rate of HIV transmission.
Why do my fingers go wrinkly in the bath?
Chris: - The reason that you get wrinkly fingers in the bath is because the skin on your fingers and hands and toes is quite thick, and it's there to protect you. You have a slight thickening of the skin because we tend to rub our fingers against things more often than we rub other bits of our body with things and the skin becomes thicker there to compensate. The top layers of that thickened skin are flat, dried-out, dead cells. In fact, they're falling off of you all the time. If you could total up the number of cells that you're losing, it's about 40,000 skin cells a minute that fall off of you. If you added them all up, that's 1 Ã?,½ stone in dead skin over a lifetime. Now that dead skin on your fingers which is still attached, despite being a flattened thick layer, is nonetheless slightly penetrable by water, and water can get into that layer, and it makes the thin flattened cells swell up a bit. And in the same way that if you make railway lines very, very warm, they can buckle and bend which is why you have to leave a gap between them, the skin cells to the same thing. They swell up, they press into each other, and as a result, they push each other out of the way, and they get thrown up into all these ridges and folds because the cells are bigger, because they've got water in them. After you get out of the bath or the sea, or the swimming pool, and you dry out again, that extra moisture that's got into the cells comes back out, the cells flatten out again, and they go back to their normal shape, and that's why you go wrinkly in the bath.
----UPDATE-----
In 2013 UK scientists discovered that fingers go wrinkly in water to improve our grip on wet or submerged object. They also showed that it is in fact constricting blood vessels, not osmosis, that drives this change.

Will rubber-soled shoes protect you from an electric shock?
Dave: - I think the simple answer is it is possible they would protect you from an electric shock, but it's not something I would ever recommend because it's only "possible". If you have a big, thick rubber sole then you're quite insulated from the ground, and if there's no path for electricity to flow through, you won't get a big current going through you, so you'll be fine. It's essentially a similar reason to why birds can sit on an electricity cable. They might be sitting on a very high voltage, but there's no path for electricity to get down to earth through them so they're absolutely fine, there's no current flowing.
So, as long as you're just standing on your shoes you're not touching anything else, your feet aren't wet so they're not creating a short path around the rubber soles to the ground, and you're not accidentally touching anything metallic - If you were touching the outside of a toaster which might be earthed then you might get a horrible current going through the knife, up your arm, then down the other arm, then you might be okay. But there's so many things that could possibly go wrong, so unplug the toaster first.
Ben: - I think that sounds like a sound advice, and that by and large, don't stick metal things in electrical appliances.

How do the filaments in a toaster work?
Dave: - It's basically just a very long piece of fairly high resistance wire. I've taken a toaster apart recently so I know this. It starts at one end and kind of zigzags up one side, then the current flows down and then zigzags up along the other side, and then it's attached to the neutral wire. So, one end of it is going to be at 240 volts. The other end is going to be at about earth. So, if you stick something metal anywhere near the 240 volts, even 100 volts, you're going to get a big shock.
Chris: - But if you were lucky and you got it to the neutral end, the potential there is a bit lower. So if you were making a toaster, hopefully, you'd design it with the most inaccessible bit of the high resistance filament, furthest away from where the knife's going to go.
Dave: - Looking at how the toaster worked, one of the holes is going to be more dangerous than the other one, but they didn't seem to have made any particular attempt to make one side more difficult to get at than the other.
Chris: - They assume people have got some common sense after all.
Ben: - Exactly. I guess if the general rule and the general understanding is, "Don't stick metal cutlery in your toaster" then the other safety features are probably obsolete!
Are there foods that flavour breast milk?
Chris: - Well we have looked at this as a news item on the Naked Scientists a couple of years ago. There was a paper we reported. I actually gave it the exciting title of "Fulsome flavours on offer at the "breastaurant". It was a piece of work done by a lady called Helene Hausner who is from the University of Copenhagen. It was a paper published in the Journal of Physiology and Behaviour in 2008. What she did was to investigate this very phenomenon because women, including my wife, who have had babies, often report that the certain things they eat will affect whether or not the baby is very enthusiastic to breastfeed or not.
To get to the bottom of this sort of transmission between food and breast milk flavourants, this group recruited 18 women who were breastfeeding at the time and they got them to give some milk samples before and then milk samples after they gave them some capsules containing various flavourants. The flavourants they tested were menthol - we all know what that is - and also a chemical called d-carvone. Carvone is the stuff that makes caraway seeds have that very aromatic flavour to them. They also tested a chemical called 3-methyl acetate which is a banana flavour, and trans-anethole which is a sort of a liquorice taste. It's the thing that makes star anise and Ouzo have the liquorice taste the way it does. By feeding the women these things and then taking breast milk samples from them for certain amounts of time after, up to 8 to 10 hours, and then measuring the volatiles (the smells) above the breast milk, they could work out roughly how much of these flavourants were getting into the breast milk. It wasn't trivial. In fact, they found that different times elapsed for different flavours. The menthol took about 4 to 6 hours to peak, d-carvone and the trans-anethole took about 2 hours to reach peak levels, but the banana flavour, the 3-methyl acetate, didn't come through at all.
The interesting thing was that there was an 80% difference in the levels of these different smells and taste between different groups of the women. So if you look at one woman and compare another woman for the same flavour, you might detect 80% more or less of that flavour in her breast milk compared with the other woman. And if you do the test more than once on the same woman, you might find more than 50% variation in the levels of these different flavourants in their breast milk. What this shows you, therefore, is that it makes a very big difference from one person to the next and it makes a very big difference even in the same person.
But the bottom line is that things that you eat definitely can end up going into your breast milk, and it's not just small molecules and flavourants; it's also whole proteins. Researchers back in the '70s did studies by radioactively labelling proteins and amino acids, sending them through in the diet, and tracing them out into breast milk, showing that they do then end up going out and going into the baby. So, people who say their babies do develop tastes for things based on what mum's been eating are absolutely right.
Ben: - How well developed is a baby's sense of taste? Do you think that babies can actually taste this?
Chris: - It's almost certain that they can. If you think about it, the baby, when it comes out, is very under-developed and tends to prioritise the development of the systems that enable it to succeed when it's little. So it tends to develop first the things that will be most essential to survival. That includes knowing when something is good for you and bad for you, and how to alert mum with a cry. And babies, if you want to encourage them to eat something, they've got to like what they're eating. It's a sort of reinforcing thing, isn't it? The brain has to say, "I like this. I want to do it more" which makes sure the baby feeds regularly. So it's almost certain that babies do get hooked on these flavours.
Can we justify the dangers of nuclear power?
Ben: - Some compelling arguments there, thank you very much, Beverly. It's a certainly one for you Dave, you're our physics guru. You know a lot more about nuclear power than I do. What are the arguments in its favour?
Dave: - It basically depends how bad you think burning coal is. Burning coal actually releases more radiation into the environment than nuclear power ever has, even including things such as Chernobyl, because there's a load of radioactive elements in coal. You burn it, they get released. There's no way of constraining it. Also, burning coal produces lots of carbon dioxide which is bad for all sorts of other reasons including the greenhouse effect, etc. There's also issues with acid rain which have been largely improved, but we're getting huge amounts of our energy at the moment from fossil fuels. Nuclear power is a very, very dependable form of power, you know it's going to work. If you replace it with something like wind power, I think it's going to be very, very difficult to supply the amount of energy we're using in this country using just renewable sources. There's a professor at Cambridge, David MacKay who worked out that in order to produce all the energy we use in this country using wind power, you'd have to completely cover the whole of the coast with miles of wind turbines to generate that amount of power. Nuclear power does have a lot of disadvantages, but it does have the advantage that you know it's there. It's predictable. You can store the energy. You're not dependent on foreign sources of energy. So, if someone stops supplying you with uranium, it's very easy to store 10 years worth of power.
Whilst in the long run, it might not be what we want to be using - it depends how dangerous you think radiation is. If you got a heavily radiated area like Chernobyl, it might not be particularly good for humans, but actually, if you look at things like the amounts of biodiversity there, how well the animals are doing, it's a lot better than anywhere humans are. Humans are a lot worse for the environment than radiation is!
There are all sorts of positives and negatives to it. On balance, I think it's probably worth using at least until we develop something better.
Ben: - Beverly, I appreciate that we may not have completely turned you into a fan of nuclear but does that help, hearing some of the positive arguments?
Beverly: - It's certainly interesting to see it that way round. I think your colleague was not addressing the issue of renewables as much as existing alternatives. Yes, we've got to look at how do we bridge for the next 30 years, and how we use electricity, you know, what sources are we going to use.
Ben: - Well it's certainly something that we'll have to keep an eye on. Thank you ever so much for your call.

33:00 - The Vintage Computer Festival
The Vintage Computer Festival
with Chris Vallance, Chris Serle, BBC
Meera: - Once again, I've come down to the BBC in White City in London to meet our resident technology expert, Chris Vallance. But this time, we're doing things a little bit differently because we're inside a pod inside one of the BBC cafes.
Chris V - Yes, we're in the cafeteria. We hurried out of the rain then we chanced upon this experimental pod for confidential meetings, which is really just a small glass or sort of Perspex room.
Meera: - So, this is a bit of an architectural trial, shall we say, but what have you been up to in terms of technology recently Chris?
Chris V - Well, I've been travelling back to the future. I went Bletchley Park. Now Bletchley Park is an interesting place to go visit if you've got an interest in technology. It's the home of Britain's World War II code breaking efforts and if you go there now, you can see a lot of the pioneering early electronic digital computers that we use to crack German codes, pioneering work by people like Alan Turing.
It's also home of the National Museum of Computing, but over the summer, it played host to Britain's First Vintage Computer Festival. What's a vintage computer festival? Well, if you're of a certain vintage yourself like me, a child of the '70s or a child of the '80s, you may remember BBC micros, LX 81s, VIC 20s, Commodore 64s. Perhaps you remember an Aquarius or an Acorn electron or Sinclair QL. All those machines and the enthusiasts who look after them and repair them were there.
Two thousand people came along to look at these things. People are doing really interesting things with the old bits of kit. They were pushing the machines to their limits, getting old games, repairing machines. There was even a ZX Spectrum which people connected up to the internet and you could Twitter from the Spectrum.
Other kinds of things where people really are just seeing how far they could push the graphics and the sound, and the processing power of these machines. For example, one group was showing off a ZX-81, running an image of a bouncing ball. That 3-dimensional bouncing ball doesn't sound very particularly impressive by today's standards when you consider how basic the machines are, you know, that's quite a challenge. So people doing things like that.
Some groups are actually sort of continuing to update the machines and to bring out new versions of them because if you like, they enjoy the way the machines work. So there's a big group of people who were very enthusiastic for the Commodore Amiga. You know, there are new versions of that machine coming out that were on display at the show. But it wasn't just the machines that were from the 1980s. I spoke to Chris Serle who used to present the BBC's computer show.
Chris S - In the early '80s, I co-presented the BBC's first ever television series dedicated to computers and it was the series which gave rise to the development of the BBC microcomputer which ended up in almost every school in the country.
Chris V - What's the importance or maybe just what's the joy of events like this?
Chris S - It's the first time this has happened and I think it's ridiculous that it's taken this long actually. I mean, arguably, the scientists in this country invented the computer.
...
Chris V - Tell me why you're here.
Vintage computer fan - Because I love computers. I mean, you go to see loads of other things that are really old. You go to see Spitfires and Lancasters, and stuff like that. It just makes a change to go and see something else.
Chris V - Commodore 64, I guess a 1980s machine?
Another vintage computer fan: - Yes, '80s. I can't quite remember. I play around my one, but yes.
Chris V - I just made you crash as well. I'm very sorry. You were flying a yellow spaceship, shooting lots of things and it's all gone wrong. Do you work in computers?
Another vintage computer fan - Yes. My dad brought one home and ever since then, yes, it hasn't really been a day gone by we're having played with the computer.
Chris V - What have you enjoyed most? What's the thing here that you've liked playing on those?
A young vintage computer fan - The games over there, because it's brought me a bit more to like computers now.
Chris V - And that the games over here, they're quite simple. I mean, the one over there that you're pointing out, that's Space Invaders which old people like me used to play, but you still quite like it?
Young vintage computer fan - Yeah, because it's quite a short game. If you're getting ready for bed and you just wanted to play a quick game on the computer, you could just choose Space Invaders. 
Chris V - So the old machine's were a hit even with those who'd never experienced them the first time around.
Meera: - Now Chris, did you have a go on any of the old machines and how were they functioning?
Chris V - Well, I mean, it was all great. I mean, I remember them from being a kid and I think - what did I have? I had an Acorn Electron. I think what's interesting is just that people were using the old machines to do new and interesting things, for people who enjoy programming and enjoy tinkering with electronics. There's something of a challenge to get an old machine to do things that it wasn't designed to do and having a limited amount of power, and resources at your disposal. I mean, millionths of the kind of memory that you have even in phones. If you think about it, a ZX81 had a 16k memory. If you bought an expansion pack! My phone's got 16 Gigs on it, so that's a millionth of the capacity. I think one of the interesting things is, you know, for the younger generation, the games are still fun, and the machines are still fun to play around with. And they really do sound like computers. There's something about that retro beeps and bloops, and the noises they make that's really endearing.
Chris - Making a machine do what it wasn't designed to do. I think Lada cars pioneered that concept with the heated rear windscreen that doubled up as a hand warmer to keep you from freezing when you are pushing. Anyway, that was Chris Vallance and he was showing Meera Senthilingam how to make the most of your old computers, and also revealing how computers, and not just clothing, are valuable when they're vintage.

How does hair clean itself?
Ben - Now that's a good question. I think he's probably referring to the idea that if you stop washing your hair for six weeks or so, then it ends up sort of auto-cleaning and you end up with clean hair despite it not being washed. I think the truth is a little bit more complicated than that. It's not that your hair is clean; it's that your hair has the right amount of oils. Normally, when you wash your hair with shampoo, it's a detergent and it will strip the oils off your hair. Hair is supposed to be a bit oily so your hair follicles produce additional oil to make up for the oil that's been washed away and you end up getting greasy hair. If you don't wash it away, there seems to be some kind of feedback mechanism that means that the follicles will produce the right amount of oil, and you won't end up with greasy hair.
Now I don't know if this really bears itself out in practice and from what I have seen and read about it, it seems to be much better for people with very short hair. It's not that they never wash their hair, it's just that they don't use soaps or shampoos - they just wash it with water. The water takes away the dirt and grime but the oils stay there to help protect the hair. Dave, you've got quite a lot of hair. How easy is it for you to manage?
Dave - A while ago, purely in the spirit of scientific inquiry, and much to the annoyance of my then girlfriend, I decided to see what would happen if I didn't wash my hair for three months. Basically, what happened is my hair sort of reached an equilibrium. It was much less greasy than it would be just before you'd wash your hair normally, but still greasier than just after it: a bit greasy, but not very greasy. So there does seem to be some kind of feedback loop. Also, interesting things happen when it rained because it appears that your natural grease from your hair is quite good at waterproofing because the outer layer got incredibly greasy and the rain would run off whilst the hair underneath would be entirely dry. So obviously, natural oils have advantages over conditioners.
Ben - That's very strange. It's like wearing a natural shower cap made only of hair - that's revolting.

47:51 - How do ants and spiders count?
How do ants and spiders count?
Chris Smith answered this...
Chris - Well, not just spiders but probably many insects can do this. The example I gave were spiders counting their steps in order to know how to build their webs but also, there was a very elegant paper that's published in Science a year or so ago by Matthias Wittlinger who's at Ulm University in Germany.
He and his colleagues were looking at how ants navigate. Ants use the sun and they also use a compass - they have in their brain a body clock and they're able to use this mental compass of the time of the sun moving across the sky to navigate by. But they also count their steps and the reason that the scientists know that for sure is that they performed the very delicate task of putting ants on stilts.
They cut hairs off the back of a pig and then glued those hairs onto the ants' legs to lengthen the ants legs, and when they did this, of course, the ants' strides became twice as long as they would normally be. When they followed the ants around, all of the ants overshot their nest because they walked twice as far as they should've done.
When they actually, paradoxically, cut the ant's legs off so they had much shorter legs, they didn't walk far enough. They're all circling around, thinking, "Where's my nest? I should be there." It was clear that they're actually counting the number of steps they've taken in order to find their way around.
In their brain and their nervous system, they must have some kind of neural integrator circuit that, every time they take a step, notches up another 1, 2, 3, and so on, so they can find their way around by counting steps. So there's the evidence: ants can do it, it's likely that other insects probably do the same.
Ben - So ants on stilts will overshoot their home! Poor ants!

Why does shaving foam stop the mirror from steaming up?
Dave - In some senses, it doesn't actually stop it steaming up. Steam is lots of little droplets of water. When the light hits it, the light gets bent and so you get a very distorted image which, when you move away from it, just looks like a kind of mist. What the shaving foam does: there are lots of detergents in it and those detergents reduce the surface tension of the film of water so it doesn't form lots of little droplets. It just forms a big flat sheet that you can see through much better, so you can see through it even though the water is still condensing.
Ben - So there's literally the same amount of water there; it's just a change in the structure of the water. So why do the droplets make it so hard to see things?
Dave - Because water has quite a high refractive index - when light hits it, it bends. If it hits this curved surface of a droplet, each one basically acts as a little tiny lens, light is bounced off in all sorts of different directions and makes it looks essentially white. This breaks up the image and makes it look misty.
Ben - Fantastic! Well very interesting. Not perhaps what you're supposed to use shaving foam for but if it works, it works.

50:39 - How much gas would I have to expel to achieve lift off?
How much gas would I have to expel to achieve lift off?
We put this question to Dr Mark Lewney, award-winning science communicator based in Cardiff...
Mark - If you weigh ten stone, or 63.5 kilograms, then the Earth pulls on you with a gravitational force of 622 Newton which you have to then overcome if you want to lift off for even a fraction of a second. You have to direct a force at least this strong towards the ground, which according to Newton's Second Law will be equal to the mass of the gas you expel multiplied by the acceleration given to it by your bowels. That means you could give an enormous amount of gas a small acceleration or give a tiny mass of gas a huge acceleration.
So how much does the average emission weigh? This is quite tricky to measure since methane is lighter than air. You actually weigh more after letting rip a ripe hum-dinger on boxing day due to a slight loss of buoyancy. Assuming that is that you don't also follow through with non-gaseous matter; an act commonly known as "sharting". Incidentally, pure methane is also completely odourless which proves that every real fart is actually a "shart" in disguise. Having asked Jeeves what the mass of the average human trump is, given the density of methane and the pressures and temperatures found in the bowel, he returned - admittedly with his nose turned up even higher than usual - with an answer of 0.037 grams or 0.000037 kilograms.
So, to generate the upward force required to oppose gravity, your bowel muscles must give this mass an acceleration of 622 divided by 0.000037, which equals 17 million metres per second per second, almost 2 million times the force of gravity. Assuming it's expelled in a 1 second toot, that yields a final exit velocity of 17,000 kilometres per second which is equal to 37.6 million miles an hour or 18% of the speed of light.
Anyone capable of achieving this feat is invited to contact the UK Space Agency with a view to highly profitable long term employment.











Comments
Add a comment