Ben and Dave select their favourite bits of Naked Science: from taking an MRI of outer space to orange fireballs and chocolate teapots. We explore the boys' best Naked capers. Plus, we join Dr Hal for a gassy set of explosive experiments.
In this episode

01:37 - The Teslathon - High Voltage Fun!
The Teslathon - High Voltage Fun!
with David Woodroffe, Teslathon
Ben - This week also saw the annual Teslathon, held at the Cambridge Museum of Technology. The Teslasthon sees enthusiastic amateurs get together to show off their home made tesla coils - high voltage devices based on the same principal as an electric transformer.
 A transformer works because a current in one circuit - called the primary, induces a current of a different size in the secondary circuit. This is how mains electricity is scaled down from the high voltage in power lines into the safer voltage that gets to your house. I met up with Derek Woodroffe to find out more about what the Teslathon is...
A transformer works because a current in one circuit - called the primary, induces a current of a different size in the secondary circuit. This is how mains electricity is scaled down from the high voltage in power lines into the safer voltage that gets to your house. I met up with Derek Woodroffe to find out more about what the Teslathon is...
Derek - Teslathon is a group of people who are interested in high voltage electronics, Tesla coils and pretty much anything to do with high voltage, current, static electricity: all sorts of technology-related stuff like that.
Ben - So really anything that can make a nice big spark.
Derek - That's very much part of it. Some of us try and make the biggest spark possible. Some of us try and do it in more interesting ways. Of course we try and push the modern technology to do something that couldn't be done 18th century-wise by Tesla himself.
Ben - How do Tesla coils work? They seem a very simple principle.
Derek - They are a very simple principle. Effectively it's a standard transformer with a primary and a secondary. What Tesla did was he also introduced resonance so the primary has an associated capacitance. The secondary has an associated capacitance. The two synchronise with each other and form a resonant coupling, very much like a young child pushing somebody on a swing. You can get a very small movement that can be made into a very large movement just by the process of resonant rise or multiplication.
Ben - And this enables you to have huge voltages and this is what gives you these lightning-like forks that seem to be flying across the room behind us.
Derek - That's right. Some of the coils start at about 240 volts. They quite often cheat and go up to 10,000 volts or so into the primary of the coil. Then, due to resonant rise in the way the Tesla coil is constructed we'll get 100,000 volts or 200,000 volts from the top. But because it is high frequency AC that means we can then push quite a lot of power into a spark or an arc which will then grow much longer than the 100,000 volts sounds.
Ben - And that's why they do seem to be reaching out and fingering their way across the room. There are some really huge forks of lightning across here. Is it actually safe?
Derek - No. Is the simple answer. Like most things that are interesting or fun it isn't safe. You have to be very careful. Most of the people in this room have been doing it for very many years. They know their equipment because they've had to build it from scratch. It's not something you can just go out and buy. There is inherent safety: we all abide by a set of rules for the safe running of these sorts of events. People have to stand back from the equipment. The equipment has to be able to be made safe but obviously there is that inherent danger. Any high voltages, high currents, unpredictable equipment you've got to view with a degree of distrust.
Ben - I'd imagine that the element of distrust you have means you have to be fairly reserved in public. The people who come along to the Teslathon this weekend won't really see the full power of what your devices can do.
Derek - They will see a limited amount. There are some things certainly that we wouldn't do in a public environment that we would do in private. Obviously there's the safety of the public and the people who are watching the Tesla coils here today is absolutely paramount. We don't want to hurt anybody. It would really ruin the enjoyment of the whole event for everybody.
Ben - Cambridge Industrial Museum, where we are today, seems like a very appropriate setting for this. I understand you come back each year to do another Teslathon here. Does it feel like home?
Derek - Certainly for me. I've been doing this for about seven years now and the actual Teslathon has been here to my knowledge for 9 or 10. It's usually on the same weekend every year, which for some reason happens to be Halloween. I don't know whether that's by planning or by accident! We've always been very welcome here and obviously with the connection to 18th century technology we seem to fit in very well with the other machines and equipment at the pumping station. Of course, we all like to go and have a look round that sort of technology too.

How useless is a Chocolate Teapot?

12:46 - The Post Prandial Proceedings of the Cavendish Society
The Post Prandial Proceedings of the Cavendish Society
with Dr Jeff Hughes, Manchester University
Jeff - The post-prandial proceedings of the Cavendish Physical Society were a collection of after dinner songs that the research students sang at the annual Cavendish dinner every year. Some of the students were very good singers so they sang favourite songs of theirs. Some of them were very good aspiring lyricists and they re-wrote the words to some of the songs to reflect the events and personalities of the Cavendish Laboratory. They just told stories in their songs about what was going on in the lab.
 Ben - This was a student thing. Was this just a lot of fun by the students and researchers or did some of the more eminent people get involved as well?
Ben - This was a student thing. Was this just a lot of fun by the students and researchers or did some of the more eminent people get involved as well?
Jeff - At the annual dinner the professor would be there, the head of the laboratory. There would be guests who would have been former students of the laboratory who had gone on to jobs and scientific eminence elsewhere. They'd be invited back and that would create a very nice sense of tradition and continuity with the past with the current students. The former laboratory members could be held up as role models for them as to what they might aspire to.
Ben - A very good opportunity to meet some of the era-defining scientists of the time and, at the same time, have quite a lot of fun.
Jeff - Absolutely. Could you imagine being in a dinner where you would see your head of department and a well-known Nobel prize winner standing on their chairs, linked arms singing Auld Lang Syne at the top of their voices.
Ben - This sounds like a very casual thing. It happened at a yearly dinner but how do we know about it? This sort of thing usually would be a bit of an inside joke that would pass by unnoticed.
Jeff - This was a very serious informal tradition. The students were so pleased with their own songs that they kept them. In 1904 they published them in a pamphlet and that was republished in six editions up to 1926. That's how we know about these songs. We know something from diaries and letters and so on about how they were actually performed.
Ben - Could you give me an example of the sorts of lyrics that they were coming up with?
 Jeff - Yeah. A.A. Robb, the mathematical physicist wrote this one about 1905-06. It's to a cod Irish jig called Father O'Flynn. I like this one because I play the fiddle and I play Irish jigs quite a lot. Imagine an Irish jig rhythm. It's:
Jeff - Yeah. A.A. Robb, the mathematical physicist wrote this one about 1905-06. It's to a cod Irish jig called Father O'Flynn. I like this one because I play the fiddle and I play Irish jigs quite a lot. Imagine an Irish jig rhythm. It's:
Of dons we can offer a charming variety
All the big pots of the Royal Society
Still there is no one of more notoriety
Than our professor, the pride of us all.
Here's to the health of professor JJ
May he hunt lions for many a day
And take observations and work out equations
And find the relations which forces obey.
When the professor has solved a new riddle
Or found a fresh fact he's as fit as a fiddle.
He goes to the tea room and sits in the middle
And jokes about everything under the sun
Then if you try to look grey at his jest
You'll burst off the buttons that fasten your vest
For when he starts chaffing though tea you'll be quaffing
you cannot help laughing along with the rest
Ben - This evening, we've seen some of them performed by the HBS choir, do you think this might be the first time they've been performed in, maybe, 100 years?
Jeff - As far as I know, this is the first time that these, the three songs we've heard tonight, have been performed since probably the 1930s.
Ben - So really, it's quite an historic event that we've been involved in?
Jeff - Absolutely! Historians of science these days are really interested in re-creating historical experiments, what we've heard tonight is the recreation of historical songs, and I'm, absolutely thrilled.

18:52 - MRI Scanning the Stars
MRI Scanning the Stars
with Professor Alyssa Goodman, Harvard-Smithsonian Center for Astrophysics
Chris - That's exactly what Professor Alyssa Goodman from Harvard University is doing. She's taking some of the systems that have been geared up to do better body scans with MRI and applying them to images of the night sky to enhance those pictures and she's with us now. Hello Alyssa. Thank you for joining us. Tell us a bit more about this work.
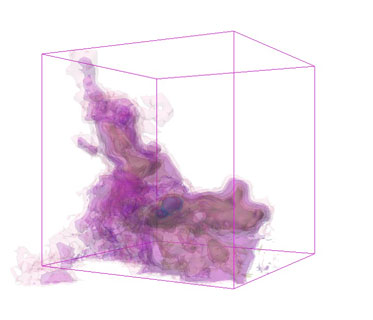 Alyssa - We have a challenge in astronomy that more and more often we're able to get a third dimension of data: something like distance, not always exactly distance. Instead of seeing just a flat sky we can see thing where we know what distance. We can see in my work, for instance, gas clouds that are busy forming new stars. We like to see what they look like the way that we could go around 3D clouds in the sky. Of course you can't do that in astronomy. We need a way of putting the images back together into something that looks more like a 3D picture that turns out that the computer software to do that we need is similar to the software that they use in medical imaging.
Alyssa - We have a challenge in astronomy that more and more often we're able to get a third dimension of data: something like distance, not always exactly distance. Instead of seeing just a flat sky we can see thing where we know what distance. We can see in my work, for instance, gas clouds that are busy forming new stars. We like to see what they look like the way that we could go around 3D clouds in the sky. Of course you can't do that in astronomy. We need a way of putting the images back together into something that looks more like a 3D picture that turns out that the computer software to do that we need is similar to the software that they use in medical imaging.
Chris - Why is it such a problem, compiling images into three-dimensions like that?
Alyssa - It turns out it's less of a problem in other fields and in astronomy people are just not used to having that kind of information. They were starting to try to build their own software and then we realised that a lot of other people have faced this ad done a good job of, for example, making animated movies. You may know that Pixar and companies like that have some of the most powerful computers around. It turns out that 3D animations, moving 3D pictures is rather computationally intensive to do high resolution. People in other fields, as I mentioned before - notably medical imaging and in film, movies and Hollywood - had figured that out quite well. We're trying to borrow on what they already learned.
Chris - When you start doing this do your images literally come alive? Can you see things that, can you identify details that have previously been overlooked?
Alyssa - One of the things that we're interested in, in my own work which has to do with star formation, is what the impact of jets of material and expanding shells from stars have on the clouds that the stars are forming in. Imagine terrestrial clouds and you set off a bomb in it and you want to see if the expansion wave, some kind of sphere expanding from that bomb looks like. You'd really love to be able to see a 3D image of that. When stars set off either supernova explosions of just powerful winds from stars the same kind of thing happens to these clouds that they're in. It's very important for us to understand what that looks like. In a lot off cases it's difficult to see that happening. It's important to view what happens to these clouds over millions of years as they evolve. This software has let us, among other things, see the outflows and shells that come from these stars in a 3D way that the human brain understands which was very hard to see when looking at just slices of the images before.
Chris - Can researchers begin to speculate that, in fact, our own solar system (in other words, the sun and our clutch of planets) actually get buffeted into existence by a big star nearby that was doing something similar to what you're describing? It was putting a jet of material out which pushed a cloud of gas to make it fall into itself, which then formed us?
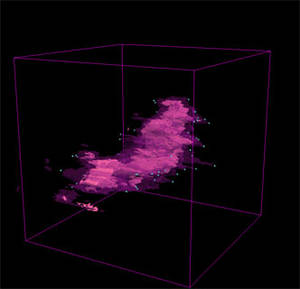 Alyssa - Absolutely. There's a theory called triggered star formations and the idea is there are these gas clouds out there which are marginally what's called self-gravitating. They're sort of held together by their own gravity but not quite. They might blow apart, they might collapse but if you come by and push them a little bit - sort of trigger - then they're more likely to collapse quickly and make something like our solar system. One way to do that is having a big blast wave either from one of these outflows or some sort of shell, possibly a supernova come by.
Alyssa - Absolutely. There's a theory called triggered star formations and the idea is there are these gas clouds out there which are marginally what's called self-gravitating. They're sort of held together by their own gravity but not quite. They might blow apart, they might collapse but if you come by and push them a little bit - sort of trigger - then they're more likely to collapse quickly and make something like our solar system. One way to do that is having a big blast wave either from one of these outflows or some sort of shell, possibly a supernova come by.
Chris - What are you looking at, at the moment? What's the prime focus of study?
Alyssa - Right now we have something we've been doing over the past five or six years called the COMPLETE Survey of Star-Forming Regions. That's a long, funny acronym you can look up online. What it does is it looks at some of the nearby star-forming regions using every technique we can use from the ground. Optical wavelength and radio wavelength. Radio is where we can make these three dimensional images. With the Spitzer Space telescope, which is the infrared part of the Hubble telescope, has also looked at these same regions. They're essentially targeted regions where we essentially want to understand the whole process of star formation. What we've been able to do with this 3D imaging project that we call astronomical medicine is to be able to give people 3D views of what these very large regions of space look like, to be able to put back together in our minds a picture of what's going on. From that picture we make hypotheses. Recently our work is about the details of the role of self gravity, how likely little bits of this gas are to collapse over time on themselves and to understand whether our theories are right. The best way to do that is to see a picture of what they mean. We've been able to convince people that we think we're on the right track.
Chris - And you have some spectacular pictures on your own website. If anyone listening on the radio wants to check it out, where's the web address so they can take a look at those pictures?
Alyssa - The best way to do it is to just type my name: Alyssa Goodman in Google and I think it's the first link that comes up.
Alyssa's website can be found
here, and there are some fantastic
pictures,
movies,
virtual reality objects and even a
3d pdf available on the
Harvard IIC Websites.

31:28 - Chemistry in its Element - Thallium
Chemistry in its Element - Thallium
with Henry Nicholls, Freelance Science Journalist
During World War I, Agatha Christie worked in a hospital and then a pharmacy, an experience that could explain the presence of poisons in many of her plots. In The Pale Horse, a thriller published in 1961, the star of the show was thallium, also known as "the poisoner's poison" because many salts of this soft, silvery metal is soluble in water, producing a colourless, odourless and tasteless liquid with a delayed effect on the victim. Here's an excerpt from the dramatic climax in which the novel's narrator Mark Easterbrook solves the mystery of several unexplained deaths.
 I slammed back the receiver, then took it off again. I dialed a number and was lucky enough this time to get Lejeune straight away.
I slammed back the receiver, then took it off again. I dialed a number and was lucky enough this time to get Lejeune straight away.
"Listen," I said, "is Ginger's hair coming out by the roots in handfuls?"
"Well - as a matter of fact I believe it is. High fever, I suppose."
"Fever my foot," I said. "What Ginger's suffering from, what they've all suffered from, is thallium poisoning. Please God, may we be in time..."
Christie may have got the idea for her plot a few years' earlier in 1957, when the KGB attempted to assassinate Nikolai Khokhlov, a former KGB assassin himself who had defected to the United States. In turn Christie's dramatic and detailed description of the symptoms of thallium poisoning in The Pale Horse is thought to have saved at least two lives and led to the arrest and conviction of a British factory worker who had used thallium to kill his stepmother, two work colleagues and nauseate around 70 others. It is so dangerous because thallium has similar biological properties to potassium ions, hijacking the ubiquitous sodium/potassium membrane pump to smuggle itself into cells throughout the body interfering with the important roles played by potassium.
Thallium is pretty abundant in the earth's crust, found in several selenium-containing minerals. Indeed, it was whilst cooking up one such compound in 1861 that British chemist William Crookes noted that "suddenly a bright green line flashed into view and quickly disappeared." He knew he was onto a new element and called it thallium after the Greek for green shoot or twig - thallos. The following year, he succeeded in isolating small quantities of the element, but nowhere near the quantities obtained by French chemist Claude-Auguste Lamy who was working away independently with a greater bulk of raw material. When, in 1862, Lamy was awarded a medal at the International Exhibition in London For the discovery of a new and abundant source of thallium, Crookes had a fit and it was only with his election to the Royal Society in 1863 - largely on the back of his thallium work - that the cross-channel spat for priority died down. Subsequent work on the chemistry of thallium showed it to have similar properties to several other elements, including silver, mercury and lead. So much so that French chemist Jean-Baptiste Dumas later dubbed it the "ornithorhyncus, or duck-billed platypus of the metals."
The raw material on which both Crookes and Lamy worked came from waste products deposited during the manufacture of sulphuric acid. The commercial production of thallium today is not dissimilar, with the metal mostly recovered as a by-product of smelting iron, zinc or lead sulphides to make sulphur dioxide. The resulting thallium contains the two naturally occurring stable isotopes, with around 30% of it made up of atomic mass 203 and the remaining 70% comprised of atomic mass 205.
 Owing to its toxic properties, thallium has been used as a rodenticide, though there are safer ways to kill rats and the use of this chemical in the environment is now banned in many countries. Today, thallium is of greatest use to the electronics industry. In particular, the conductivity of thallium sulphide alters on exposure to infrared light, making it an important compound in photocells. Thallium bromide-iodide crystals have also been used in infrared detectors. The addition of metals like thallium to glass can also reduce its melting point to as low as 150 degrees centigrade. As such low-melting point glasses do not shatter like normal glasses, they are particularly useful for the manufacture of electronic parts. Thallium is also being tested in high-temperature ceramic superconductors.
Owing to its toxic properties, thallium has been used as a rodenticide, though there are safer ways to kill rats and the use of this chemical in the environment is now banned in many countries. Today, thallium is of greatest use to the electronics industry. In particular, the conductivity of thallium sulphide alters on exposure to infrared light, making it an important compound in photocells. Thallium bromide-iodide crystals have also been used in infrared detectors. The addition of metals like thallium to glass can also reduce its melting point to as low as 150 degrees centigrade. As such low-melting point glasses do not shatter like normal glasses, they are particularly useful for the manufacture of electronic parts. Thallium is also being tested in high-temperature ceramic superconductors.
Alongside the two stable isotopes, there are a further 23 radioisotopes, though most of them with fleeting half lives. One of them, thallium 201, is useful in nuclear medicine. Its injected into the bloodstream and will find its way into all tissues with the help of the sodium/potassium membrane pump. This can then reveal to the clinician any part of the body not bathed in blood or where the membrane transporter is not working properly. In particular, it is used to image the blood flow to heart muscle in patients suspected of coronary artery disease. Thankfully, with a suitably short half-life of just 72.5 hours, Thallium 201 disappears from the body long before it can cause the lethal damage of the more stable isotopes.
In The Pale Horse, Agatha Christie was not as explicit about the treatment for thallium poisoning as she was about its symptoms. "Do they know how to treat thallium poisoning?" asks the narrator Mark Easterbrook when he reaches the hospital where the hair-shedding Ginger has been taken. "You don't often get a case of it," the investigating officer Inspector Lejeune tells him. "But everything possible will be tried." It was, and for those who like their happy endings you'll be pleased to know that Ginger makes a full recovery from the thallium poisoning that had stricken her down.
For more Chemistry in its Element, or the latest in Chemistry news from Chemistry World - visit the
Royal Society of Chemistry's Website.

44:30 - Printing Your Own House
Printing Your Own House
with Dr Rupert Soar, Freeform Engineering
Ben - Our next guest is building houses using a printer, or at least hoping to soon. Rupert Soar is from Freeform engineering where he uses computer aided design and rapid manufacture techniques to build walls and structures, essentially using a gigantic 3D printer! Hello Rupert, thanks ever so much for joining us...
Rupert - Hi Ben, Hi Helen...
 Ben - So, Freeform engineering - who are you and what do you do?
Ben - So, Freeform engineering - who are you and what do you do?
Rupert - Well, essentially we do rapid prototyping, with a slight speciality in that we focus on the construction industry and in particular on those large scale elements where we're really printing big things.
Ben - So, 'Rapid Prototyping' - I'm guessing the name gives away what it is? It's for making a prototype very quickly...
Rupert - Yeah, kind of. That's where it started. There's additive manufacturing, layer manufacturing, there's many different terms that are applied to this technology. All of them are essentially similar - taking your material, squirting it, pasting it down one layer at a time and building up a three dimensional object over time.
Ben - Now I would assume that normally this is used for things like new designs of mobile phones, or perhaps things that you hold in your hand.
Rupert - Absolutely, typically plastic things but, you know, small metal components, things that suit - mobile phones, automotive, aerospace, all very common.
Ben - But you're thinking of much, much bigger things?
Rupert - Yeah, in one level. Part of this is actually bringing awareness of what this technology is into the construction and architectural and design sector. And so trying to inform and bring about a news capability. Architecture itself is producing more complex designs and more interesting designs in the buildings that they see around them. They are able to generate amazing structures and forms in the computer and that challenge is then; how on earth do we actually fabricate those, how do we make them? They get stuck in the computer nowadays, these designs do. And so, these printing technologies just work layer by layer, printing one layer at a time and in fact there's no great magic involved - if you look at how any house is built its all bruit with layers of bricks. And the reason it's built with layers of bricks is that we can build the bits inside as well as the bits outside. So this technology is really cool because it allows us to print the really complicated stuff inside as well as everything outside.
 Ben - Of course, and the complicated stuff usually would mean greater expense because it takes more time and more expertise...
Ben - Of course, and the complicated stuff usually would mean greater expense because it takes more time and more expertise...
Rupert - If you were talking about traditional engineering and manufacturing and trying to machine something out of a solid block, trying to get inside it is damn near impossible at the best of times, but you can really get into these structures and make them really complicated and do quite remarkable things.
Ben - One of the key things in buildings nowadays is all about using the right materials, and we've talked on the show before about things like thin layers of wax inside little capsules that melt when you get to certain temperature and then solidify again when and using this as insulation. Are you limited with rapid prototyping as to what materials you can lay down?
Rupert - Yes and no, like all things. There's no one ubiquitous process that does everything but essentially if you take your DeskJet printer and literally scale it in your mind, and instead of ink you're putting through cement or gypsum, let's say, then you're somewhere close to where we're going with this. It seems almost strange to think that you could squirt cement out and it wouldn't slump all over the ground, but that's because cement is used in moulds at the moment and concrete and it's designed to be sloppy. You take out those retardants and things like that and it starts to set quite quickly. So very, very quickly you can start to build up three dimensional structures - very, very big ones, if that is indeed what you're into doing.
 Ben - I think you've given us a very good image of how it works, but I'm picturing a giant frame with an inkjet type head, effectively a bucket with a hole in the bottom that you control, that moves forwards and backward in three dimensions and releases your material as and where you need it.
Ben - I think you've given us a very good image of how it works, but I'm picturing a giant frame with an inkjet type head, effectively a bucket with a hole in the bottom that you control, that moves forwards and backward in three dimensions and releases your material as and where you need it.
Rupert - Yeah, if you've ever seen a large crane working in a shipyard, most people have got an image, that's a big crane and it's placing big things and that's kind of where this is going - large crane systems or gantry systems that can have deposition heads or squirty heads that are squirting stuff out and building them up; but that's a very simplistic level. As technology evolves then one very, very large machine very quickly becomes many, many small machines and then you're into autonomous robot swarms and all of that wonderful future that lies ahead for us.
Ben - It's a lovely idea that building sites might one day just consist of robots that get it over and done with really quickly, no wolf-whistling, no builders' bums...
Rupert - That's it. It's quite simple, you know, it's not hard to squirt things out and build things, ask anyone who has made a cake with icing sugar - it's dead straightforward. But getting it in swarms and connective agents and then you open up a whole new world of possibilities as to what you can build.
Ben - So effectively, because of the way that you can do this layer by layer; you can do the whole thing in one run. You don't have to print the outside walls and then take your machines inside and printing all the inside walls. You can actually just say - this is the design of house I want - go!
Rupert - Yes. The whole point of this is that traditional construction is a very hierarchical thing. You start by putting the superstructure and gradually with first, second, third fixing you come down in resolution, if you like, until you're literally fixing the screws and nuts and bolts into the structure and so its a very top-down approach. When you're printing a structure, and printing all the channels and ducts within the walls as well, you've essentially got to do the whole thing, all scales of resolution at the same time, so you've got to print fast to get your materials down but at the same time you've got to print fine to get all those little channels and ducts. And that's the key to it, as a friend of mine says - "The real estate of the future lies between the walls in our homes" those two surfaces. At the moment they're essentially solid, but very quickly we can engineer those walls and fabricate and essentially fold more functions into much, much smaller spaces. And that's the real key to where this drives forward.
 Ben - This is fantastic stuff. How is the cost likely to compare to traditional building?
Ben - This is fantastic stuff. How is the cost likely to compare to traditional building?
Rupert - Cost - you can never compare what is essentially something that's been going for 2000 years - you know, I'm not going to beat a brickie, I was one myself and we can work fast. What this does is it enables other abilities, things that a bricklayer can't imbue into a building. So if you're laying bricks, let's use that example, then you're good with straight lines and squares and fairly uniform shapes. A printer, if you ask it to print a squiggly line or a square it doesn't care, it makes no distinction. Essentially you can print complicated structures and forms. There's no cost involved in how complex the structure is. Now that's a fundamental difference between how existing construction is and what it could be. So we're not usurping traditional construction by any means, we're just going to add to those capabilities. So a lot of the key discussions are about sustainable construction. Natural ventilation I know you've got covering in this programme, and what we're able to do is actually fold the structures and channels and functions into tighter and tighter spaces within these walls, and actually make and design structures that truly can capture energy from the environment. People know me for termites, I'm a bit mad like that but that's where we're going with that - walls as membranes and not barriers if you want a quick sound bite distinction there.
Ben - This all sounds fantastic and I hope that we'll see it in building sites in the very near future. That was Rupert Soar, director or Freeform Engineering explaining how one day you might just select the house you want, and print it out!

52:55 - Cool Chemistry with Dr Hal
Cool Chemistry with Dr Hal
with Dr Hal, Brighton University
This week, Dr Hal showed us the following cool chemistry experiments:
 | Lightbulbs in Liquid Nitrogen |
 | Dry Ice Explosions |










Comments
Add a comment