Hundreds of times stronger than steel, transparent, an excellent electrical conductor, and weighing next to nothing, graphene is hailed as a wonder material. But what is it doing for us now? And where will it take us in future? This week graphene goes under the microscope. We hear how industry can mass produce it, we uncover how it can clean up air in cities, produce the world's fastest lasers, revolutionise communications and boost the power of computers. Plus, news of how Earth's earliest life reproduced, how to regenerate human organs, and why animals have different shaped pupils...
In this episode
01:02 - Organ regeneration trigger found
Organ regeneration trigger found
with Luis Garza, John Hopkins University
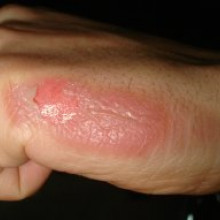
Researchers are saying that they are on the brink of being able to kickstart the  regrowth of hair follicles and even other bodily organs. Scientists have discovered a trigger - in the form of short pieces of genetic material called double stranded RNA - which switches on an immune pathway that induces stem cells to rebuild organs like they do in a developing baby. Done in the skin, it could help victims of severe burns. Luis Garza made the discovery at Johns Hopkins University in the USA, and explains to Chris Smith...
regrowth of hair follicles and even other bodily organs. Scientists have discovered a trigger - in the form of short pieces of genetic material called double stranded RNA - which switches on an immune pathway that induces stem cells to rebuild organs like they do in a developing baby. Done in the skin, it could help victims of severe burns. Luis Garza made the discovery at Johns Hopkins University in the USA, and explains to Chris Smith...
Kat - First this week. Researchers are saying they're on the brink of being able to kick start the regrowth of hair follicles and even other bodily organs. Scientists have discovered a trigger in the form of short pieces of genetic material, called double-stranded RNA, which switches on an immune pathway inducing stem cells to rebuild organs, just like they would do in a developing baby. Done in the skin, it could help victims of severe burns. Louis Gaza made the discovery at Johns-Hopkins University in the US.
Louis - When I was a resident studying medicine. All of my teachers taught that it was impossible to grow a new hair follicle if you destroy the entire thing in an adult. And yet, other people before us, and we in this paper show that you indeed can do that. And so, it does create hope. In the same way that I was taught that you can't grow a new hair follicle, now people are taught we can't grow a new limb. But maybe, that might become a possibility.
Chris - How did you do this then, to regrow hair follicles? What was the actual experimental model that you set up?
Louis - We had two sets of mice. We do these very large wounds down to the depth of muscle. So, we remove all of the skin. One set of mice have poor ability to regenerate which is kind of currently the way we think a lot of humans are. And then the second strain of mice was a very good regenerator, so we would do the exact type of wound, and instead of having a pure scar, they would have a lot of hair follicle regeneration. So, we did these gene arrays where we query, "What's the difference between the ones that do regenerate very well and the ones that don't?"
Chris - And was there a unique gene signature that appeared to be responsible for the difference in the healing?
Louis - Yeah. When we did statistics, we found a very significant signature to show that this pathway which is part of our innate immune pathway that I can explain. There are these receptors that were originally evolved to sense invading organisms that might try to cause an infection. And we found one of the elements of this ancient immune system in our mice that regenerate very well.
Chris - What is turning on that ancient arm of the immune system then in these regenerating mice? Is that the damage that then triggers that immune response that then in turn makes the skin grow better and makes new hair follicles come along?
Louis - Yeah. It's just as you said - It is damage. This is a really fun intellectual question to say - how does your body know it when it's been hurt? It turns out this ancient immune system also senses damage and that we find a strong signature for that.
Chris - Now the killer question then. If you take that pathway and you induce that effect in the mice that don't normally regenerate very well; in other words, you super load them with more of that immune response that the good regenerators have. Can you turn the bad regenerators into good regenerators?
Louis - Yeah. We were able to do that. Yes. We could significantly increase the amount of regeneration in our bad regenerating mice by giving them these compounds that activate their innate immune system.
Chris - What are those compounds?
Louis - The compound is double stranded RNA. Scientists used to believe that double stranded RNA was only present in viruses, for example, like the measles virus. And that this innate immune pathway is the receptor to tell your body, "Hey! A virus has attacked!" But now, we know that it's very likely that double stranded RNA is also formed during damage and that the double stranded RNA then is activating it's receptor, and then that's what's turning on the regeneration and turning on stem cells.
Chris - Given you've made this discovery then, and you can supply these chemicals, including this double stranded RNA signal that is the stimulus for this. Do you think then we are on the path now to being able to provision a human with these signals to make good damage and could you, for instance, make a bald person hairy again?
Louis - Yeah. We're really excited about this. And the very interesting part of this story is that we think that it's, like a lot of discoveries, it's already being used in ways we didn't predict. For example, men and women who want to look younger and go for rejuvenation and visit their local cosmetic dermatologist, and receive laser treatment or receive dermabrasion, or receive micro-needles for rejuvenation of their facial skin. It's very likely that the one thing that unifies all these very different methods of rejuvenation is damage, and that it's by activating the same tall three double-stranded RNA pathway that people are receiving benefits when they go to their cosmetic dermatologist even now. So the exciting question that our work raises is whether we can reduce the damage we have to do by instead just directly giving these agents like double-stranded RNA and save people the suffering of having to go through these treatments. The other main application of this could be in scar victims. So, people who've had burns, for example, where we think by creating new hair follicles, we'll be able to restore the skin to the way it normally is which is dense with hair follicles - even where we can't see them, for example, on the face.
Kat - That was Louis Gaza, and he's just published that work in the journal, Stem Cell.
06:11 - Huntington's gene links
Huntington's gene links
with Lesley Jones, Cardiff University
Huntington's disease is a rare but devastating genetic illness, affecting up to ten in every hundred thousand people. Sufferers carry a faulty version of a gene containing a region with three letters of DNA repeated over and over again. Fewer than 35 of these repeats and you are probably fine. But 36 or more and you'll develop the disease.
But although researchers know that the number of repeats is broadly linked to how early someone will develop Huntington's - the more you have, the earlier you'll get it - it's still highly variable, so there must be other factors involved. Now, thanks to the largest ever study of the genetics of Huntington's disease, Lesley Jones from Cardiff University and her colleagues have tracked down a number of genetic variations that affect the age at which people develop the condition, opening up exciting new avenues for future therapies. She explains to Kat Arney...
Leslie - So, there's been a huge amount of work in the biology of huntington's disease, and essentially, pretty well every biological pathway that you might be interested in appears to be altered in cells that carry the mutation that causes the disease. So, it's been really hard to pick out which pathways you should address in order to provide a treatment for the disease.
Kat - And what do you find? Who are your prime suspects?
Leslie - So, our prime suspects are a set of proteins that appear to be contributing to things like DNA repair. So they're proteins that actually operate on the DNA itself.
Kat - What sort of level of change are we talking about? What sort of influence do these variations have on when someone is likely to get the disease?
Leslie - So, we found a particular locus on chromosome 15. And there's several genes that are very close to this variant and actually, interestingly, there are two variants in this particular part of the genome. One of which, would tend to give people up to six years earlier onset of the disease. This is relatively uncommon, and another variant which is much more common, which appears to give a protection against the onset of the disease of perhaps, between one and one and a half years. But the fact that they occur close together makes us more confident that this is a real effect we're looking at here and that it's important in the disease.
Kat - And of course, the big question is, how can we then use this knowledge? The fact that DNA repair kind of molecules are important - how can we use this to find ways to treat the disease?
Leslie - The way that we can use this is to focus our attention on this pathway, rather than many of the other pathways that we've been looking at over the years. Because essentially, if you like, nature's done a natural clinical trial as one of my colleagues always says, for us. It's lets us know which of the pathways we need to intervene in in order to alter the age of onset of the disease. In other words, this gives us the potential to delay the disease. So, we think that if we try to address DNA repair, we might be able to alter the age at which people get the disease and to push it backwards. Now, altering DNA repair - it's a pretty fundamental process - and I think it's going to... it won't be straight-forward, I don't think. But people are already looking at these processes because they're important in cancers. There are one or two drugs potentially that, while they might not work immediately, they would provide a basis for further work to see if we can generate new compounds that might be suitable for manipulating this system in Huntington's disease. But essentially, we need to understand the biology better and this gives us a chance to investigate that biology in much more detail because we know exactly what we should be looking for, and that always gives you the chance to look at molecules that might alter those particular pathways because you know exactly what you're looking for.
Kat - Huntington's disease is caused by these triplets, these three letters of DNA being repeated and repeated. Are there other diseases that are similar that might also be affected in the same way by these same DNA repair processes?
Leslie - There are indeed. So, there's a series of diseases that are caused by expanded repeats. Most of them are triplets but not all of them. So, there are other diseases that could very well be affected by this same mechanism. Yes. And we're actually investigating this at the moment.
11:28 - How the first life reproduced
How the first life reproduced
with Emily Mitchell, Cambridge University
For billions of years, life on Earth was dominated by simple, single-celled microbes. Then, quite abruptly, about 565 million years ago, something dramatic happened and groups of cells began working together as multicellular organisms. Bur how did this complex early life reproduce? Scientists at Cambridge University have discovered that one of these animals, a seafloor dweller known as Fractofusus, which looked a bit like a fern leaf, appears to have had a dual method of reproducing itself. It could send out runners like a strawberry or spider plant to clone itself locally, and it could send out pieces of itself that could float off and set up new colonies of the organism somewhere downstream. Emily Mitchell explains to Chris Smith...
Chris - Ah! So, you're looking at the individual and you're looking at the community distribution, a sort of spatial distribution on what would have been the sea floor for this big community.
Emily - That's correct. Yes. So, we're looking at all different species, where they lived, and what's the position of them relative to the other fossils, the other species, and also within the species, what that can tell us.
Chris - And how does that give you an insight into how they might reproduce?
Emily - We compared its spatial distribution to what you might expect by random. We knew that if we got a non-random distribution then there's some interesting processes going on. So, we found that actually fractofusus was much, much closer together than you might expect by random.
Chris - In other words, you've got an organism here surrounded by other organisms at too high a density to be explained by - it's just a nice patch of sea floor to live on. So, you're assuming that there must be some kind of reproductive phenomenon going on that's lead to that density that you're observing in that particular area.
Emily - Yes. It's also about the spatial positions of the fossils within the cluster.
Chris - Are they, therefore, spawning a new one off the side of themselves in the same way that my strawberry plant in my strawberry patch will send out a runner and plant another plant next to itself?
Emily - We actually found that when we were looking at the reproduction of fractofusus, that it had a much more complicated reproductive mode than we were expecting. While the vast majority of fractofusus specimens were actually clinging to their parents, produced via stolons or runners of the type you see in strawberry or spider plants. It also had a waterborne propagule stage. So, that is there are little bits of it were released by the parent organisms into the water. And we used the phrase, propagules to describe them, because While they could have been sexually produced seeds or spores, they also might have been very, very tiny fragments or buds. So, what was going on as you had the grandparent specimens, if you will, were producing runners with little clones off the runners. The daughters of the grandparents were then going on to produce more baby fractofususes themselves. And what was really quite a nice part of the study is that when we broke down the sizes of the fractofusus to distinguish between the grandparents, the parents, and the daughters. The distribution of the largest, the grandparent fractofususes, were actually very different. They were randomly distributed on the bedding plane and they also showed current directionality. So, what this tells us is that they were actually formed not as clones but, instead, via waterborne propagules.

17:18 - Why cats have vertical slit pupils
Why cats have vertical slit pupils
with Martin Banks, University of California Berkeley
Your eyes are your 'windows to the world', and it is your pupils that control how much light comes through those 'windows'. But why do some animals - like cats - have their pupils stretched vertically, whereas others - such as sheep - have horizonal pupils. James Farr spoke to Martin Banks from the University of California, Berkely, who has been looking into how these different pupil shapes came about...
Martin - There's a class of animals that tend to have vertical slit pupils, and a class that tends to have horizontally elongated pupils. The former are very likely to be ambush predators, meaning that they hide and jump out at prey, and the latter, the ones with horizontal pupils, they're extremely likely to be prey animals that other animals predate upon. And our argument is that the orientation of the pupil in both cases is advantageous for these particular animals.
James - And why would that be the case? Why would, for example, the prey need these horizontal slits?
Martin - They are on the ground, and they need to see panoramically along the ground to detect prey that might be approaching from some unknown direction. And what we showed is by having a horizontally elongated pupil, it maximizes the light input along the horizontal plane. So, we think that's the right way to orient the pupil to help them see panoramically along the ground and detect predators. The other thing about these animals is their strategy for not being captured is also to run. And that's a really interesting problem because they run with their heads forward but their eyes are on the side of their head. And so, in effect, they have to see out the side of their eye to run effectively. And by restricting the pupil vertically making it short that helps decrease blur that would otherwise occur in the corner of the eye. So, we think that's the right way to orient the pupil for these animals.
James - What about the vertical slits? Why would an ambush predator, why would they not want to see a wide area? Is there something else they need to focus on?
Martin - They're not so worried about being predated upon, but what they typically do is hide and wait to lurch at an unsuspecting prey animal. So, their task is not to see panoramically so much as to gauge distance accurately. So, this is a really clever adaptation to open the pupil up vertically while narrowing it horizontally in order to maximize their distance estimation capability.
James - One potential problem is if an animal, say a sheep, still wants to make sure that it's not going to get attacked by a wolf from its side. But if it leans its head down to eat, is that not going to cause problems at all?
Martin - Yeah. That's a great question. We worried about that. We thought that might be fatal to our idea. So, we went to a zoo and to a farm, and we recorded video of goats, sheep, deer, and horses. And we found that the eye rotates in the head so the pupil remains roughly parallel with the ground even as they pitch their head down or pull their head upright. That movement is opposite in the direction in the two eyes as the head pitches down, the left eye has to rotate clockwise relative to the head and the right eye has to rotate counterclockwise relative to the head. And that's a movement humans can make but we make tiny ones and these animals are making much bigger ones. It's a really pretty remarkable capability.
James - Humans don't fit into either category of having vertical slits or horizontal slits. Why is that?
Martin - Well. There are multiple demands on the eyes. They're important for pattern recognition, in humans we read text and do fine recognition of objects. Having a vertical slit pupil or a horizontal slit pupil creates an astigmatic-like effect that could hurt some pattern recognition capabilities. So, we think humans just reached a different balance point in their goal to be good pattern recognizers.

21:47 - Bonobo language
Bonobo language
with Zanna Clay, University of Birmingham
From just a few months of age, human babies are able to produce sounds that are the building blocks for the language that we later develop. Scientists had believed that this is a uniquely human trait. But now, Dr Zanna Clay from the University of Birmingham, has found that our closest living relatives - the bonobos - also communicate with each other using "peep" sounds, the meanings of which are determined by context. Amy Goodfellow took a peep at the paper...
building blocks for the language that we later develop. Scientists had believed that this is a uniquely human trait. But now, Dr Zanna Clay from the University of Birmingham, has found that our closest living relatives - the bonobos - also communicate with each other using "peep" sounds, the meanings of which are determined by context. Amy Goodfellow took a peep at the paper...
Zanna - Although speech is unique to humans, the building blocks of speech were probably already present before we became human. And actually, our close relatives, the bonobos, show an ability that is essential for speech to develop.
Amy - Would that mean that maybe bonobos might go on to have more complex conversation or more complex speech like humans do?
Zanna - It's difficult to answer that. We know that there's probably a lot more complexity already existing in the way that these peeps are used. And for example, we've already found in another study that the peeps are combined with other calls and that the combination of the calls in sequences actually creates new meaning for the peeps. And this is something that's really interesting when we're thinking about language and speech evolution because we have what's called syntax in human speech - which is complex combinations of different sounds to create new meanings such as the speech sounds in words. And so, I think, we need to first look at how complex the peep system is before we can make any sort of assessments of what might happen in the future. But it does seem that the peep probably became more flexible and more complex in its use over time, and it probably started out more like a traditional fixed call.
Amy - What does the bonobo's peep actually sound like?
Zanna - So, bonobo peeps sound often like what you might think of as very high pitched bird or a squeaky door. They're really, really high pitched, very short little calls. In fact, in the jungle, sometimes I can't distinguish them very well from bird calls. So, they really are very "peepy" ... and actually high pitched calls require a bit more vocal control, so that's another thing that we think is interesting about peeps. It's harder to control high pitched vocalizations compared to them.
Amy - And from all of your experience in watching the bonobos, are you able to tell what they're saying explicitly?
Zanna - Well, I mean, I have a much better sense of it. Yes. Because they're not just producing peeps, they're producing many, many other different calls. And from my many years of working with bonobos in different situations, I've become a lot more connected to what they're doing and what they might be experiencing. So, by hearing and understanding vocalizations, you can get a really good window into what's going on in there. Sort of, you know, their social lives actually. And so, I find that by listening to calls as a sort of a specialist now, I can often work out roughly what's going on in their experience.
Amy - So, what kind of things do they use these peeps to mean?
Zanna - Well. I mean, they peep in really every context that you can imagine a bonobo experiencing. But they particularly peep in socially important events. So, they peep a lot during feeding and during food discovery, they peep actually when they're grooming, and when they're preparing their nests at night, they peep when they're just about to start travelling, or sometimes when they've stopped travelling, and they peep also during play and aggression, and even during sex. So, it seems any sort of relevant event for them, they like to peep about it.
Amy - Does this give us any information about speech in humans? For example, when we're babies and they make sounds that seem to be similar to peeps in my mind.
Zanna - Yes. I think it does provide us some interesting connections actually with speech development in babies because even before babies can speak or babble, actually. The study was done on babies that are less than four months old. Babies are actually able to produce these signals across all across different emotional situations. And although people often think of babies as just crying and responding to emotions all of the time, when the researchers actually looked at what babies are doing, they realized that the babies, even at this young age, can actually produce these very flexible calls that don't tie so strongly to emotion and probably form the really important foundations and building blocks of their future speech. And the ability for these babies to produce sounds that are freed up from their emotional content.

26:42 - What is graphene?
What is graphene?
with James Baker, National Graphene Centre
5 years ago, Manchester University scientists Andre Geim and Konstantin Novoselov  won the Nobel prize for extracting graphene. They did this using nothing more complicated than sticky tape and a lump of graphite - exactly as you get in your pencil. What they found was certainly a suprise. Graphene is 300 times stronger than steel; so thin you can see through it, and if you covered an entire football field with it, it would still weigh less than a gram. But what actually is this remarkable material, and why is it so special? James Baker explained to Chris Smith...
won the Nobel prize for extracting graphene. They did this using nothing more complicated than sticky tape and a lump of graphite - exactly as you get in your pencil. What they found was certainly a suprise. Graphene is 300 times stronger than steel; so thin you can see through it, and if you covered an entire football field with it, it would still weigh less than a gram. But what actually is this remarkable material, and why is it so special? James Baker explained to Chris Smith...
Chris - I've painted a picture of graphene, so far, as resembling chicken wire in some respects. But if we were to go in with a really, really powerful microscope, what would we see? What does it look like?
James - Graphene is a single atomic layer of carbon, two-dimensional material. So, occasionally, people talk about 2D material and actually graphene now is one of a number of over 2-dimensional materials that are now being looked at by the scientists. So, it is, if you like, a hexagonal structure. The chicken wire is a very good description and at that level as a single layer, I often think, if you like, of graphite as a pack of cards. And if you could just peel one of those cards off, that's effectively what you're doing when you produce a single layer of graphene.
Chris - And when one stretches or applies a force to graphene. We said it's about 300 times stronger than some forms of steel. Is that because you're literally pulling along the sheet? So, your trying to pull carbon atoms apart, and so you're spreading, effectively the force of trying to pull on it, across the entire sheet amongst all the atoms - and that's why it's so powerful?
James - So, 2004, when the first isolation happened, the so-called superlatives. Now, all these fantastic properties that have now being discovered since that first isolation - like stronger than steel, more conductive than copper, transparent, flexible, stretchable. Effectively you're stretching that carbon chain, and it's got these properties that give it such a wonderful range of possibility for future applications.
Chris - If one looks at chicken wire, the striking thing is that it's mostly all holes. So, if one looks at graphene, that presumably is also, mostly all holes - is that why light goes through it so easily and so well?
James - It's not fully transparent, It's about 97% transparent as a single layer. Graphite, as you probably know is dark. It's black. It's not transparent at all. But one of the properties you can now have is you can now start using those layers in different ways. So, for example, you can make it transparent. You can make it strong. You can make it flexible. You can also make it permeable or impermeable to gases and water which is another reason why there's a whole range of properties being looked at for graphene.
Chris - Why is it then impermeable to gases? Because you've got these fairly big holes, if I return to my chicken wire analogy, I've got lots of space. Why wouldn't, say, water molecules or small gas molecules or gas atoms fall through the holes?
James - So, the scientists now are doing a whole range of experiments. Having isolated the graphene, we can now effectively... I'll call it, tune the different layers of graphene. So, through experimentation it's been shown that you can effectively tune the layers to only allow certain size of molecules to pass through or to be repelled, if you like, by the graphene barrier. So, ultimately, for example, water desalinization is an area of huge interest in research, both for research applications but also for commercial applications. Imagine having a membrane made out of graphene that could separate, for example, dirty, salty water and you could produce pure drinking water through that membrane - what a fantastic commercial application, if you could actually achieve that.
Chris - Could you do the same thing with gases? Could we clean up waste gas or scrub out, say, carbon dioxide from a power station flue? Can you filter gasses in this way, in a molecular way with graphene?
James - I've mentioned it's tuning properties. So, again, there are different applications for gases, for liquids. A simple experiment we've done in the University of Manchester is one where we've separated water from fuel, just to give an example of a liquid. But also, as you say, we're looking at separating gases. So, cleaning up the air might be a possibility for graphene in the future.
Chris - Is it easy to do that, James? Does it take a lot of tweaking to get the graphene to do that and, therefore, is it scalable? Could we make an industrial filter based on graphene?
James - Lots of the experiments today are still being done in a laboratory. But increasingly now, we're starting to see applications being developed using graphene as an additive, probably in the first instance. But yes, it is still quite a challenge to scale up and to get the purity, and the volume of the graphene for these commercial applications today. But graphene is still relatively young - 2004. For a new material, it's still relatively young. So, we're starting to see quite rapid advances but, not yet to industrial scale for some of these exciting applications but we're getting there.
Chris - Graphite is a very good conductor of electricity, isn't it? So, presumably therefore, graphene which makes up the graphite is too. Why is that? And why might that be useful?
James - So, again, these fantastic properties of mixing different layers, different forms of graphene... again, graphene is a very broad term and there are many different ways of producing graphene from different processes - either form graphite or from chemical mixing of materials. But by certain ways of producing that, you can also try and enhance the electrical conductivity. So, again, some fantastic opportunities, either as a coating or as an additive into a polymer or into a composite, we can exploit some of those thermal management properties. So, the transfer of heat away from a light bulb, for example, is a good example. Again, a partner we're working with in Manchester has used a graphene layer within an LED light bulb to dissipate the heat more effectively than a conventional metal filament. So, again, you're starting to see graphene appear in products for the future.
Chris - If it's made of carbon and therefore likes oily things, does this mean that you could potentially also add it to things like oils and have a better lubricant for your engine that will also take heat away from the components it's trying to lubricate?
James - Sure. Yeah. To date, people are looking at the graphene properties for lubricants. Today, there are companies who are actually adding graphene to lubricants for drilling to take the heat away from the drill head. But again, companies are potentially looking at exploiting those properties in a whole range of different applications, which again, is why it's so exciting from all these different markets and applications for the future.

34:06 - Making graphene industrially
Making graphene industrially
with Catharina Paukner, Cambridge Nanosystems
Graphene certainly has some pretty impressive properties, but to harness these features we need to actually make it. Sticky tape is probably not practical for this! So how do we do it? To find out, Amy Goodfellow went to see Catharina Paukner at Cambridge Nanosystems - one company that produces graphene in industrial quantities by breaking apart methane molecules and using the carbon that's released...
features we need to actually make it. Sticky tape is probably not practical for this! So how do we do it? To find out, Amy Goodfellow went to see Catharina Paukner at Cambridge Nanosystems - one company that produces graphene in industrial quantities by breaking apart methane molecules and using the carbon that's released...
Catharina - Graphene is really hard to make on a large scale because this is a single atom layer of carbon. And generally, single atom layers are not very stable and they don't like to exist. We start out with something that's called hydrocarbons or a gas, like methane, the classic greenhouse gas. And we want an all-carbon product so we have to crack it. And the way we do it is we employ a microwave.
Amy - A microwave like I would have at home?
Catharina - The same you would have at home, just a little bit higher powered. You have a power generator, a wave guide and you heat the gas with that wave.
Amy - So, basically, the high energy of this microwave cause these atoms to break apart, and that's how you get your carbon?
Catharina - Yes. Exactly. That's how it is. That's actually the beauty of our process. The only two things we need is electricity and methane. You just think of it as different steps along one way. You have the gas flowing, at one point it breaks, in the next it forms graphene, in the third it gets filtered and then compacted and processed.
Amy - Shall we go and have a look at what's going on? Oh! We get to wear some nice lab coats!
Catharina - So, what we have in here is five of our small-scale reactors.
Amy - These are a lot smaller than I thought. They look like small greenhouses, and in each one there's this sort of about meter long by half a meter in diameter cylinder?
Catharina - These reactors allow us to produce approximately 1 to 2 kilograms of product per day.
Amy - What's actually going on in here?
Catharina - So what we see here is the microwave I was describing earlier. With a slightly more industrial layout than...
Amy - Yeah. This doesn't look anything like a microwave I have at home. It's literally just a silver box. That's all. No window to see your food going round.
Catharina - Not exactly. Yeah. The wave is generated in here and transported through this wave guide. You can see here where the gas is injected. Here is the microwave that cracks it. Then here we have the part that is, the reactor part where graphene is formed.
Amy - So, the cylinder I was describing is just a bit where the graphene actually forms after this cracking has happened?
Catharina - The internal structure is what drives formation of graphene.
Amy - So, it's all going left to right, basically, as we're looking at it?
Catharina - Yes. So, the graphene is formed and transported in the gas stream and sort of filtered out and drops into this hopper, and then hydrogen is emitted through this pipe and goes into an incinerator. So, this is a tiny sample of graphene in a glass pot. You can see it's a very, very low density fly powder.
Amy - Yeah. Because as you shake it around, it sort of almost floats, in a way. It's like one of those toys you have at Christmas where you shake it and it has snowflakes inside.
Catharina - Yeah. It is. It has very low density obviously as you would expect from a single sheet of graphene.
Amy - Yeah. I suppose it doesn't weight very much, does it?
Catharina - It doesn't . So, the amount that you see here would be a approximately 2 grams - maybe one and a half?
Amy - Oh wow! And that's quite a big pot. So, two hundred milliliters weight about 2 grams.
Catharina - Yeah.
Amy - How much would this 200 milliliters be worth?
Catharina - This is sort of our top of the range product, and we sell it mainly for research purposes and for very high-end products. So, we set it at approximately 10 pounds per gram.
Amy - What about the less fancy graphene?
Catharina - We sell different grades of graphene tailored towards the application. Where large-scale applications are required, our carbon we sell it for approximately 45p per gram.
Amy - What's the difference between these two?
Catharina - The different qualities of our material vary in terms of layers, on top of each other. So, while our top range product is sort 70% single layer, the grades as they go down will more have layers - between 10 and 15 in our sort of lowest grade.
Amy - So, if we were looking at some powder of you lowest grade. Would it be a bit less floaty? Compared to this fancy one?
Catharina - It would be slightly denser. Yes.

39:44 - Applications of graphene
Applications of graphene
with Andrea Ferrari, University of Cambridge
What is graphene actually doing for us right now? Andrea Ferrari leads the Cambridge Graphene Centre, which has been set up to investigate the science and technological applications of graphene. Andrea is working on a range of projects that are already delivering promising results. He started by explaining to Chris Smith, some "concrete" findings...
Graphene Centre, which has been set up to investigate the science and technological applications of graphene. Andrea is working on a range of projects that are already delivering promising results. He started by explaining to Chris Smith, some "concrete" findings...
Andrea - Cambridge Graphene Centre works with a variety of companies, almost 40 at the moment. And one of these companies is called Italcementi. It's one of the biggest concrete producers in the world and within the so called graphene flagship project which is a 1 billion Euros project that will try to bring graphene to the factory floor within the next 10 years. We are trying to help them, together with the University of Bologna to develop a new kind of concrete that will be self-cleaning but especially, will reduce pollution. And to do so, graphene is added to the surface of concrete. The concrete has also other nanoparticles inside and helps trap gasses that could be potentially toxic - such as NOx's.
Chris - We can already get surfaces to clean themselves up though, can't we? Famous structures such as king's cross station in London has self-cleaning glass where light hits the glass, it drives a chemical reaction that burns off the dirt effectively. Are you sort of doing the same thing here then?
Andrea - Yes, indeed. In terms of self-cleaning concrete, this company has already a brand out that you can purchase. What happens is that if you put graphene in it, you can potentially double the efficiency of the process.
Chris - That's a massive difference!
Andrea - Yeah. A huge difference, and graphene is enhancing the properties of the nanoparticles that are already present there.
Chris - Right. So, on the surface of the concrete are some nanoparticles. But in glass, they use titanium dioxide. Is that the same in concrete?
Andrea - Indeed. It's titanium dioxide and what the key finding is if you put together titanium dioxide and graphene, you enhance the process by a factor of two or even more.
Chris - Do we know how that works?
Andrea - That is the sticking point. We don't know exactly how it works and that's why we are trying to help. We are trying to conduct some basic physics investigation in order to figure out exactly the process that is underneath this fantastic property.
Chris - Would you have to pour concreate which has got graphene right the way through? So, that's going to be an enormous amount of graphene when you think how much concrete get made every year, or is this like a veneer. You smear this on the surface of a concrete and you then get the cleaning just on the surface.
Andrea - It is a surface effect, so we are speaking about if a relatively thick layer at the surface of the concrete. So, not the entire concrete will be full of this material. But even this will require significant production of graphene.
Chris - And will it work indefinitely?
Andrea - At the moment, there are no long-term studies. But we do know that graphene is really resilient, so there's no reason why they should stop after many years.
Chris - Goodness! And what sorts of applications might there be - just buildings in cities or could it be used in other places as well?
Andrea - Another interesting application is in tunnels where you have to have cars going through. As you know at the moment you have some ventilation systems, but having a concrete that is able to reduce pollution will be much safer and in the case of a fault of the ventilation system, the concrete will still be able to reduce the pollution in the gallery. So, that's a very interesting application as well.
Chris - Is it not dark in a tunnel though?
Andrea - It is dark but the light that you use is not visible light so we can still work under these conditions.
Chris - Could you retrofit this to existing buildings? I'm thinking that there are lots and lots of buildings built in concrete in big cities all around the world. They're all getting dirty. There's lots of pollution around. Could we veneer buildings with this after they've been built or does it need to be part of the fabrication?
Andrea - I think in principle, you can do that, but of course I'm not a builder. So, you probably have to ask one of them.
Chris - What about other aspects of graphene interacting with lights? Because one of the other claims that's being made is that it's extremely good, potentially, in the world of lasers. Why is that?
Andrea - Yes, indeed. We heard before from James that graphene is very transparent but actually it's also the most absorbing material for the thickness it has. Because the thickness of graphene is one-billionth of a metre, so when we speak about 2% of a one-billionth of a metre, it's a huge number. So, graphene can help create extremely fast Lasers.
Chris - Define an extremely fast Laser. What does that mean?
Andrea - We are speaking of the order of one-millionth of a billionth of a second of a pulse, which is really record property.
Chris - Why is that useful? Why do I want a laser that just sends me the briefest of all brief flashes?
Andrea - For example, to send data to. In the future we want to exchange information, streaming high-definition movies in a few seconds and that's optical telecommunications, are one of the options. So, it's certain you want to have extremely fast pulses. Another application is for surgery, because in this way you can ablate material. For example, if you think about laser surgery without cooking the surface of the skin.
Chris - Useful.
Andrea - That would be quite useful as well.
Chris - How does the graphene do that then? How would one incorporate graphene into a laser to achieve that effect?
Andrea - Graphene works like a sponge for water. So, if you think about water flowing and you put a sponge under the water. The sponge will fill with water, and momentarily the water will not flow underneath the sponge. Then you squeeze the sponge, the water will keep going until it gets filled up again. So, graphene does exactly the same with light. Momentarily it can absorb all the light by hanging on to it.
Chris - Any colour? Any Wavelength?
Andrea - Any wavelength. It's the only material that will work at absolutely every wavelength of light.
Chris - And that's why it looks so black when you put lots of it together to make graphene, isn't it?
Andrea - Indeed. Because it's 2% per layer, so if you have 20 or 40 layers, then it's completely black. But if do so, you don't have a laser. So, you still have to use them...
Chris - And how do you give it the metaphorical squeeze of the sponge then, to get the light out. How do you do that?
Andrea - The good thing is that the graphene does it by itself. So, it's one property that is intrinsic to the graphene. You don't need to squeeze it. The excited states in graphene go to the ground state, and that's the squeezing, extremely fast of order of a millionth of a second. Millionth of billionths of a seconds by themselves. So, you don't need to squeeze it. It does it for you.
Chris - Therefore, the application would be you make some kind of graphene filter that you put in front of or within your laser. And you then fire the laser light into it, and it then does or takes care of the creation of the pulses.
Andrea - Yeah. If you think, for example, of a simple laser pointer, if you put graphene on the front of it, then you will get laser pulses out of it.
Chris - What about the electrical properties of graphene? Because one of the other things that people are very excited about is it is, as James was saying, very conductive and, therefore, might be able to make a big difference to data transmission and the way computers work. I know my computer, my mobile phone gets very, very hot when I'm doing even the most mundane thing with it.
Andrea - Yes. This is linked to what we said before. Graphene has an exceptional interaction with light. And so, in order to transmit data, even the laser we already discussed, you need then what is called a modulator in order to give, like what we are doing now, we go on air so we need to put the message in. And then we need the detector, which is like an antenna, the receiving end. Graphene can do the three parts that we discussed but the consumption of energy that is ten or even one hundred times smaller what silicon is doing at the moment.
Chris - So, that should save the cellphone battery for a little bit longer, shouldn't it?
Andrea - Yes, indeed. It should also not cook your pants when you have the cellphone on.

47:44 - The future of graphene
The future of graphene
with Karl Coleman, University of Durham
Graphene has only been around as a material for 10 years, which is a pretty short time in terms of new materials and getting them from the lab into commercial applications. What does the future hold? Karl Coleman is a Professor of Chemistry at Durham University, and chief scientific director at Applied Graphene Materials, a company which manufactures graphene and advises its customers on how to incorporate it into their products. Karl explained to Kat Arney just what all these amazing properties are actually going to do for us...
in terms of new materials and getting them from the lab into commercial applications. What does the future hold? Karl Coleman is a Professor of Chemistry at Durham University, and chief scientific director at Applied Graphene Materials, a company which manufactures graphene and advises its customers on how to incorporate it into their products. Karl explained to Kat Arney just what all these amazing properties are actually going to do for us...
Karl - Yeah. So, we heard some great applications and potential applications from both James and Andrea. In the future, I guess what we're looking at, we're looking from a commercial perspective. We're really after what we call early wins, some people like to call them low hanging fruit. But I prefer the early wins, a more positive approach.
Kat - So, is this just sticking graphene into current products to make them better?
Karl - It's slightly more sophisticated, but yes. So, what we would call sort of drop-in technology where you'd actually take a current process or a product where you'd just really improve those properties, enhance the properties of that material. So, we're talking there really of things like paints or coatings, oils and lubricants that we heard mentioned earlier, composite materials, or even in energy storage - so, super capacitors or batteries. So, really actually using graphene in a more drop-in technology sense but making use of more than one of those properties that graphene has. We heard a lot about the exceptional properties that graphene has, but really what we want to do in the commercial sense is to make use of perhaps more than one of them and then you can get that cost-performance benefit.
Kat - I love the idea of using graphene, maybe I can have my graphene tracksuit and my hoverboard. Are there going to be more fun applications of graphene that people are starting to explore?
Karl - Yes. There are. I mean, just last week there was an article in Nature. So, that was a graphene kirigami. So, kirigami's just a form of origami but with cutting. So, they were able to fabricate springs, hinges, and all sorts of weird and wonderful things. And if you put an iron pad on the graphene spring, you can actually move it in a magnetic field. So, you can stretch it in a magnetic field. So, you can see how these could be used in stretchable electrodes or wearable electronics, wearable devices. There are lots of fun things around the corner.
Kat - And are there any things that you think that graphene actually won't be the right thing for? We do hear so much about it. It seems there's an incredible amount of hype around this material as well. Is there anything that it probably won't turn out to be good for?
Karl - Oh! That's a tricky question. We like to focus on the positives, so the things they will be good for. It's hard to say. There are lots of potential applications for graphene out there but there's also lots of competition. Some of the technologies that we're talking about, mainly in electronics, there's competitive materials. So, it's going to be tougher for graphene to really make a step change perhaps in some of those applications. So, we are really hung up on silicon. We like silicon, the advancement in silicon is phenomenal. So, will you really see it in chips? Maybe on its own - no, I don't think so. But perhaps as a hybrid. So you could see it incorporated with silicon to deliver something that you can't do with silicon. So, there's the combination. So, replacing silicon - say no. But used in conjunction with silicon, then yes.
Kat - Another area that people are very interested in with sort of the nanomaterials is in potentially in medicine in drug delivery. We heard about the possible use in surgical lasers. But what do we know about its safety in the human body, if people do start making nano devices out of graphene that could go inside us?
Karl - Yes. If you're talking implants, and of course the toxicology and toxicity of graphene does come into play, or potential toxicity. So, there are lots of efforts out there in the scientific arena where people are looking at the potential toxic, or to see if graphene actually has any toxic effects. So, that's still an area that's very open, and an area that people are researching very intensively. There's no evidence at the moment to perhaps suggest that it's going to be any different to any other carbons that are perhaps out there that people are using. So, it's really watch this space I guess for what people come up with in that area.
Kat - So, I'm not going to ask you to throw your crystal ball too far into the future, you know, the graphene spaceship heading to Pluto, but maybe in the next five years what do you see as being the key applications that will really start to come to fruition?
Karl - So, the applications we're likely to see. So, we've seen the earlier adopters - so sporting goods. Graphene's already out there in tennis rackets, bicycle wheels and bicycle components, so we're in the next phase. And so I think we're likely to see it in coatings, oils, and lubricants. Composites, perhaps a little way down the track. But the big one to watch out for, perhaps, is super capacitors. That has an enormous amount of potential.
Kat - What's a super capacitor? I'm a biologist.
Karl - It's another way of storing energy. So, it's a bit like a battery but you store it through ions rather than electrochemical work. So, you separate the ions in a device, you can charge them up in seconds. A battery takes hours to charge up, where as a capacitor you can charge up in seconds. But that's the simple differentiate between the two.
Kat - Over the past half an hour we've heard of some really incredible and exciting applications but what do you think are the hurdles that need to be overcome? We've heard a bit about how we can make it on a more industrial scale. What are the really big challenges to get graphene into the prime time?
Karl - We're making headway in graphene synthesis, that's going well. There's still more things to solve in terms of the synthesis and isolation purity of the material. But really, if you're looking at some of these applications in that you have to get it into something. So, graphene dispersing in a variety matrices is going to be very, very important. And that can be tricky. So, it's very important to work with people, companies or technologies that really know their products or process, and then help and work with them to get graphene to disperse into their products or media. And then really to deliver the benefits of graphene that we're all talking about.

How many people are needed to avoid inbreeding in a population?
Connie - Well, Louise. That question all sounds a little Adam and Eve. And it's also something the naked scientists have been wondering. You see, we recently bought a desert island in the Pacific, and we want to avoid any sticky situations in the future. So, how many people do we need to start with to keep our island healthy? When I asked you on Facebook and Twitter, Glenn Fisher thought that only one was a safe bet, whilst Jay Michael Antovics II thought that it might depend on what definition we use. Maybe Professor Mike Weale, a statistical geneticist from King's College London can help us.
Mike - So, inbreeding means different things to different people. So, there no one single answer to this question. Everyone is related to their partner somehow, it's just a question of how far one needs to go back in time before a common ancestor is found.
Connie - Oh wow! So wait a minute! Does that mean I'm technically related to my boyfriend?
Mike - Technically, yes! I mean, to stop all relatedness between all mating partners, you would need, in fact, an infinite number of people.
Connie - Okay. I see. But our island isn't going to be infinitely big, and more importantly, I'm not sure I can stomach the idea of being related to my boyfriend. And that must also mean that absolutely everybody is inbred, which just doesn't feel quite right. Surely, there's another way?
Mike - Well. Yes. There is. To a population geneticist, the definition of inbreeding is simply a situation where mating partners are more closely related than what's expected by chance. So, using this definition all one needs to do to avoid inbreeding is to select mating partners purely by chance, as though you were in the lottery. And then, the population can be as small as you want. Well, you need to have at least two. But in a small population, even one that was enjoying some hedonistic version of the national lottery, mating partners will unavoidably tend to be more closely related to each other.
Connie - And I suppose that can't be good for the future?
Mike - Yep. In the short term, this increases the chances of people suffering from certain types of genetic diseases, diseases such as cystic fibrosis or Tay-Sachs disease, for example. This is because these diseases are caused by inheriting a bad genetic variant both from one's mother and from one's father. And the chances of them both having the same bad genetic variant are increased if they are closely related to each other.
Connie - Okay. So, where does that leave us then?
Mike - Ultimately, there's no magic population threshold that will make this problem go away. But a study in 2002 suggests that a population of 160 onboard a so called generation spaceship travelling to the stars should be able to keep itself genetically healthy. So, this would be a reasonable guideline for your desert island. In fact, real human populations on islands in the Pacific have survived population crashes down to as few as 20 people, but I wouldn't recommend this as a way to keep your desert island either healthy or happy.
Connie - Well. There we have it. It all depends on your definition. I think to be on the safe side, I'm going with at least a few hundred. Who wants to come?










Comments
Add a comment