The role of lasers in biomedicine goes under the spotlight this week as we explore the workings of photodynamic cancer therapy, find out how laser tweezers can be used to force-feed bugs to white blood cells and hear how a new technique uses laser-powered DNA nanoswitches to spot specific genes. Also, why the proton just got smaller, prompting a reevaluation of some trusted laws of physics, how antidepressants in seawater can make shrimps swim towards danger and a novel mechanism for natural selection - beneficial bacteria! Plus, in Kitchen Science, what the patterns produced by laser light shining through a substance can reveal about its structure.
In this episode
01:30 - The Shrinking Proton
The Shrinking Proton
New research published in the journal Nature suggests that the proton might be as much as 4% smaller than we previously thought, and this discovery might prompt a revaluation of some trusted laws of physics...
Protons are one of the basic subatomic particles - atoms are made of protons, neutrons and electrons (except hydrogen, which lacks a neutron).
The size of the proton is a value used in Quantum Electrodynamics (QED) and in spectroscopy, but so far has only been known to an accuracy of about 1%. Physicists would obviously like a more accurate figure, but only recently has this been experimentally possible.
To get a more accurate measurement, Randolf Pohl at the Max Planck institute for Quantenoptik in Germany used a specialised particle accelerator to alter hydrogen atoms and replace the electron with a particle called a muon - creating muonic hydrogen. Muons have the same charge as electrons, but roughly 200 times more mass. This means it will orbit much closer to the proton and as such interacts more closely with it, allowing us to more accurately probe the proton's properties.
Muonic hydrogen only survives for around one microsecond, but this is long enough to blast the atoms with a pulse of laser light, which causes the muon to jump up to a higher energy level. When it falls back down, it releases some energy in the form of x-rays. Detecting and analysing the energy of the x-rays released tells us the energy difference between the 2 states. This energy gap, known as the Lamb shift, is determined by the size of the proton.
This gives results that are more accurate than other methods of proton measurement, but they suggest the proton is actually 4% smaller than we thought. This could have implications for the theories of quantum electrodynamics, or could imply that the Rydberg constant, a value used in spectroscopy - for identifying what elements we can see in interstellar dust, for example - may not be correct.
Physicists are likely to be queuing up to check their finding, putting every element of the experiment and calculations under scrutiny, so we'll have to wait and see what this measurement really means for modern physics - but this could be a very significant shakeup.

04:19 - Anti-depressants could mess up marine food webs
Anti-depressants could mess up marine food webs
Anti-depressants that end up in sewage effluent could have a major impact on marine wildlife, causing shrimp to swim towards instead of away from light. That may not sound important, but it is, because hanging around in well-lit waters make these animals far more likely to be eaten by fish or birds, potentially disrupting entire food webs.
 Publishing in the journal Aquatic Toxicology, Yasmin Guler and Alex Ford from the UK's University of Plymouth, got the idea for this study from a type of parasite that infects shrimps, making them more likely to swim towards light where they are eaten by other animals, that are the next step in the life-cycle of the parasite. The parasites change their host's swimming behaviour by manipulating levels of serotonin in their brain.
Publishing in the journal Aquatic Toxicology, Yasmin Guler and Alex Ford from the UK's University of Plymouth, got the idea for this study from a type of parasite that infects shrimps, making them more likely to swim towards light where they are eaten by other animals, that are the next step in the life-cycle of the parasite. The parasites change their host's swimming behaviour by manipulating levels of serotonin in their brain.
The researchers wondered if the anti-depressant drugs people take to target serotonin levels and control their mood might have a similar affect on other animals.They studied a common species of crustacean called Echinogammarus marinus, that lives between the tides - lift up a frond of seaweed on the beach and you might well see them hopping and wriggling around - on coasts from the North Pole down to southern Portugal.
The shrimps were kept in tanks and exposed to seawater containing different levels of various drugs including the anti-depressant fluoxetine - better known as Prozac. Over a course of three weeks, the shrimp exposed to 100 ng/L of fluoxetine were five times more likely to swim towards light, instead of away from it, compared to shrimp in clean seawater.
A 2009 study, showed a similar affect in a fish called the fathead minnow. When they were exposed to fluoxetine for 5 days, they become more likely to be caught by predators.
Not all the drugs we take are absorbed in the body, but some pass right through and end up being flushed down the toilet. These drugs aren't removed in normal sewage processing plants, and become concentrated in rivers and estuaries at levels up to and higher than those tested in this study.
Pollution from drugs is currently largely overlooked, but studies like this are starting to draw attention to the potentially devastating ecological problems they cause. It's possible that many other crustaceans, and other marine wildlife, could be similarly affected by hormone disrupters like this and many other manmade chemicals that are finding their way in increasing concentrations into the wild.
07:45 - Growing new brain cells
Growing new brain cells
Researchers in America have identified a chemical that encourages the growth of new neurons - and protects against neurodegeneration.
Steven McKnight at UT Southwestern and colleagues screened 1000 different chemicals - and found 8 candidates that seemed to support formation of neurons in a region of the mouse brain called the dentate gyrus. This region, in both mice and humans, is thought to contribute to the formation of new memories, and is one of the few regions known to have high levels of neurogenesis even in adults.
 One of the eight candidates, P7C3 had favourable pharmacological properties, so the researchers focussed their attentions on this. It works by preventing apoptosis - or programmed cell death - in newborn neurons. The process of neurogenesis is long and perilous - neurons form and migrate over the course of two to four weeks to the desired site, and many do not survive this process.
One of the eight candidates, P7C3 had favourable pharmacological properties, so the researchers focussed their attentions on this. It works by preventing apoptosis - or programmed cell death - in newborn neurons. The process of neurogenesis is long and perilous - neurons form and migrate over the course of two to four weeks to the desired site, and many do not survive this process.
Further research in mutant mice that lack a gene essential to normal brain development showed a marked increase in neurogenesis when on a prolonged course of P7C3. As well as new neurons forming, a measurement of electrophysiological activity showed that the dentate gyrus was functioning as it should be - so the new neurons are properly incorporated and functional.
There was also tantalizing evidence that P7C3 also enhanced the birth of new neurons in aged rats. As rats age, they tend to show a reduction in neurogenesis alongside a reduction in the ability to form new memories. Rats on a daily dose of P7C3 showed an increased ability in standard learning and memory tests, and also a higher rate of neurogenesis.
The next step is to identify the molecular target for P7C3, but this finding could point the way to new neuroprotective drugs that prevent or treat diseases like Alzheimer's.

09:39 - Nature’s firework displays help females spot males
Nature’s firework displays help females spot males
Why do fireflies flash in time? Their rhythmic, bioluminescent displays are extraordinary phenomena, sometimes lighting up entire forests with bright pulses of light. But why it happens is one of nature's great mysteries - there are lots of ideas, but until now no one has experimentally tested any of them out.
Now Andrew Moiseff from the University of Connecticut and Jonathan Copeland from Georgia Southern University in the US have done just that. Their study, in the journal Science, suggests that swarms of male fireflies flash simultaneously so that females can recognise a potential mate from their own species.
Male fireflies fly around giving off a characteristic pattern of flashes, a different one for each species, like a system of Morse code. If a female spots a male of the same species, she will flash back during one of his pauses.
For around 1% of the 2000 firefly species - which are in fact a type of beetle - males will synchronise their flashes over large areas.
To test out their ideas of why this happens, Moiseff and Copeland created a virtual firefly world in the lab. They collected female Photinus carolinus fireflies, a synchronous species from the Smokey Mountains National Park in Tennessee, and flashed an array of LED lights at them, to mimic males of the same species.
When all the LEDs flashed together, females were duped into thinking they were real males, and they responded by flashing back around 80% of the time.
When the LEDs flashed out of sync, the females hardly responded at all - they only flashed back 10% of the time or even less.
It seems that when fireflies are crowded together, females can only make out individual males when they all flash together - otherwise it just looks like a jumble of flashing lights to them.
Flashing males are constantly on the move, which means that if a female focuses too narrowly she could easily miss parts of his characteristic flash pattern as he flies in and out of view. Instead, she probably needs to look over a larger area of space so she can pick out these moving patterns. But if there are lots of males, all flying around and flashing out of time, the patterns quickly become muddled and confused.
The next step of the research will be to work out whether female fireflies' tiny brains are wired up in a way that means they can't detect asynchronous flashes, and ultimately, whether they are driving the evolution of males that flash in time and put on these extraordinary night time firework displays.

13:57 - Naturally Selected Infections - Symbiotic Bacteria and Evolution
Naturally Selected Infections - Symbiotic Bacteria and Evolution
with Professor John Jaenike, Rochester University
Ben - Also in the news this week, researchers in America have identified a novel mechanism for evolution. In the wild, a gene that produces an organism's chances of surviving increases their odds of reproducing. It's usually passed down to the next generation and so, that gene will become more common in the population. But what if instead of a gene giving an advantage, it's actually an infection with a symbiotic bacteria? Professor John Jaenike is a biologist at Rochester University in New York and he joins us now. Thank you for joining us John, what was it that made you look into this in the first place?
John - Well actually, I'd been studying Drosophila and their interactions with nematode parasites in the 1980s and 90s, and back then there's one particular species, Drosophila neotestacea that was really getting clobbered by nematodes which I've studied quite a bit back then. For the past 10 years or so, I've been studying a different sort of infection which are endosymbiotic bacteria that are passed on from mothers to offspring.  We had recently discovered a Spiroplasma bacteria in Drosophila neotestacea, and it didn't seem to be doing anything to enhance its own transmission. So, I wanted to see whether or not maybe they provided some sort of benefit for flies that are parasitized by nematodes and lo and behold, we found in the lab that they had a whopping effect. When we look at this out in the field, it was equally strong or even stronger. So there was more than a tenfold increase in the fertility of female flies if they carried the Spiroplasma.
We had recently discovered a Spiroplasma bacteria in Drosophila neotestacea, and it didn't seem to be doing anything to enhance its own transmission. So, I wanted to see whether or not maybe they provided some sort of benefit for flies that are parasitized by nematodes and lo and behold, we found in the lab that they had a whopping effect. When we look at this out in the field, it was equally strong or even stronger. So there was more than a tenfold increase in the fertility of female flies if they carried the Spiroplasma.
Ben - So they obviously give an enormous advantage in fertility, but is there a trade off? Do the bacteria affect the flies? Do they need to eat more? Is it harder to fly? There must be something - they must be paying something for that advantage.
John - There probably is some cost. We haven't seen anything though. We're just wrapping up right now a population case experiment to look at the dynamics of the Spiroplasma infection in the absence of nematode parasites. We don't see any obvious fitness cost. Fertility is unaffected, the dynamics of the infection, it just seems to act like a neutral trait in the absence of the nematodes. So there may be a cost but it's not big enough for us to detect. The main problem is that the transmission rate is less than perfect. An infected female passes the Spiroplasma onto about 97% of her offspring. So in the absence of any selective benefit, that actually would be lost from the population very quickly.
Ben - And how have we seen the rates actually changing in the population and where have you got your flies from in order to look back historically and see the relationship?
John - Well, in the 1980s, virtually every single nematode parasitized fly that I collected was completely sterile. Recently, the situation has changed dramatically. Now, the vast majority of parasitized flies have some level of fertility - which is actually astounding. So based on that sort of evidence, I've been able to infer that the infection rate by Spiroplasma increased from about 10% in the 1980s to about 70 or 80% today - this is around Rochester. Also, I was able to get some museum specimens that actually, a former student of mine, Dave Grimaldi and I had collected in the 1980s, and we developed PCR primers to look for Spiroplasma in these museum specimens and it turns out that none of the flies, none of the 20, were infected with Spiroplasma from the 1980s. So, the confidence limits on that are around 0 to 15%, so our best guess is that the infection frequency was around 10 to 15% in the 1980s, and that's increased dramatically in the last 20 years.
Ben - That's definitely a significant change. Is this the only example that we know of, of an infection like this offering a selective advantage?
John - There's actually a handful of cases now that have been published. So there's a very nice example of a bacterium called Hamiltonella which provides aphids resistance against parasitoid wasps. There have been more recent studies of Wolbachia which is another endosymbiotic bacteria, conferring resistance to RNA viruses in Drosophila and in mosquitoes. So there are few examples. The previous studies have all been done in the laboratory so we've actually been able to show that this works out in the wild, and also, we've been able to estimate the relevant parameters that govern the dynamics, and those parameters are consistent with this very rapid increase in the infection in the last 20 years.
Ben - So, how can this tell us a bit about evolution? As I said in the introduction, the mechanism is very similar, but instead of a gene, we have this symbiotic infection. What can we learn about the way that the flies have evolved from this mechanism?
John - Well I think this may be the tip of the iceberg, these few cases that have been found so far. People have been surveying insects now for the last 10 years or so for infection by endosymbionts. There are a number of species of endosymbionts that infect insects and it turns out that the majority of insect species are infected by one or more species of endosymbionts. In the vast majority of cases, we have no idea what they're doing and I wouldn't be surprised if in many cases, the endosymbionts actually provide some kind of protection against some kind of natural enemy that they encounter in the wild. It's exactly analogous to adaptation by spread of a beneficial mutation. So all the standard criteria for evolution by natural selection are met in this case, but it just doesn't involve a gene.
Ben - This brings me on to my final question that you said most insects we think of probably got a relationship like this. Can this tell us anything about tackling bugs that are a problem, those that carry disease?
John - There are a couple of human diseases - river blindness and lymphatic filariasis, also known as elephantiasis. These diseases are caused by nematodes that are carried from person to another by insect vectors, black flies and mosquitoes. So it's occurred to us that if Spiroplasma adversely affects nematodes in these insect vectors, the same way it does in Drosophila, one might be able to use Spiroplasma as a means to actually control the spread of these particular diseases which infect tens of millions of people in tropical regions especially Africa. So it's a potentially novel means of controlling these particular diseases.
Ben - Well this is a wonderful finding. A very elegant paper and it's nice that there's so many different angles to take from it, but thank you ever so much for joining us. That was Professor John Jaenike from Rochester University.

21:08 - Therapies with Lasers
Therapies with Lasers
with Dr Martin Austwick, University College London
Helen - This week, we're exploring the role that lasers play in medicine and biomedical research. Later on we'll find out how lasers and special tags can make DNA sequencing much faster, and how laser tweezers allow us to look at how cells interact on a one to one basis. But first, we're joined by Dr Martin Austwick, from the Medical Laser Centre at University College London. Martin, thanks for joining us.
Martin - Thanks very much for having me.
Helen - First of all, what sort of things are lasers being used for in medical treatments?
 Martin - Well the sort of treatments we do at the National Medical Laser Centre are generally arranged arorund treating tumours or conditions which relate to cancer. That's quite a common specialty for laser therapy.
Martin - Well the sort of treatments we do at the National Medical Laser Centre are generally arranged arorund treating tumours or conditions which relate to cancer. That's quite a common specialty for laser therapy.
Helen - And how exactly are lasers used when it comes to treating cancers and tumours? We've got something called photodynamic therapy (PDT), haven't we? What's that all about?
Martin - Well that right. So the traditional, old fashioned approach to using lasers in medicine is use a very powerful laser used as a cutting tool which is a very clean way of excising tissue, but photodynamic therapy uses something a bit more subtle. It doesn't use a thermal laser. It is a very weak laser and what that laser does is it activates a chemical, a photochemical which under the normal circumstances is inert and don't do anything. But when you shine - mostly any kind of light, but we use lasers because they're easy to control. When you shine the laser on them, this drug activates and becomes active and it combines with oxygen in the tissue, and creates these things called reactive oxygen species which can damage the tissue around them. So it's like a very sort of microscopic level caustic burn that we're creating with this interaction of light and chemical.
Helen - And what sort cancers can this be particularly effective in treating?
Martin - It's being used around world on a whole variety of different cancers. The sort of work I've been involved with in London have been one for focal treatment of prostate cancer. It has been used in the past on the oesophagus. There's a new trial being planned on the breast, one on the pancreas. So really, there's a whole range of different options because if you've got a hollow organ like the oesophagus, it's a fairly simple matter to shine the light, put some sort of fibre into the organ and shine the light around it. If you've got a solid organ, you can actually insert a fibre via a clear needle and illuminate the solid organ from within.
Helen - Right. So it's not just a case of shining light from the outside. You can actually introduce it into the body.
Martin - Yeah.
Helen - But presumably, do you have to keep patients in the dark otherwise? Would normal light actually affect them as well?
Martin - Well that's absolutely right. Typically these drugs are administered systemically, the patient will be injected with them or they'll take them orally, and then their whole bodies will be photosensitised. So at that point, you have to keep them in [the dark]. You don't have to be excessive, but it's normally a dimly lit hospital room. And then when it comes to the time that you want to do treatment, you take them into theatre or into clinic, and you just shine the laser on the particular portion you want to treat. So on the surface, you can just shine on a bit of their skin and if it's an internal organ, you can focus the light there.
Helen - And in terms of, from the patient's point of view and in terms of how they're treated and how long it takes, and maybe the after effects as well, how does this compare with perhaps more conventional ways of treating cancers - chemotherapy and so on?
Martin - In terms of focal therapy, in terms of if you're treating an individual tumour, the big advantage of photo dynamic therapy over something like surgery or thermal laser treatment is that photodynamic therapy tends to target the living cells and not the collagen scaffolding that surrounds them. So what you tend to see is that you kill the cancer and when the tissue recovers, when it grows back, you still retain a really good cosmetic effect because the underlying scaffolding of the tissue hasn't been affected. So if you're doing something on the surface, maybe doing something for example on the head and neck, you know, you end up with a sort of treatment that doesn't leave the patient with a lot of disfigurement which can really be important.
Helen - Excellent. And lasers can also be used in another treatment which goes by the name of Photochemical Internalisation (PCI). We've got lots of good words in this story. So what's that one about? How does that differ from what we were talking about in terms of the photo dynamic therapy?
Martin - Well this is a very new treatment that we're really excited about. What this does is it works in a similar way to photodynamic therapy in that it's activating a photoactive drug. But also, you've got the additional factor of a chemotherapeutic agent and there are certain kinds of cancers that are very resistant to chemotherapeutic agents. And what this does is effectively - the chemotherapeutic agent, the chemotherapy drug will be taken up by the cells and the photo dynamic effect when you shine the laser will actually break apart the structures that are trapping and isolating the chemotherapy agents. This means that these chemotherapy agents get to the cell and can actually do what they're supposed to do which is kill the cell.
Helen - It's a way of injecting it very specifically - the chemical agent into particular cells?
Martin - Well, it's more a way of preventing resistant cells from being resistant. So it's breaking down the defences of a cell that doesn't want to be treated by chemotherapy. But it is very specific as you say.
Helen - Are you using this already or this is something that's in development?
Martin - There's a very new trial on patients which has been going for a few months now in head and neck cancers mainly, and I think that's the only one which is being carried out at the moment.
Helen - Okay, so this is something that we can look towards in the future. Well thank you Martin very much for giving us an introduction to how lasers are used in medical treatments. That's Dr. Martin Austwick from University College London.

26:36 - Using lasers to pinpoint DNA molecules
Using lasers to pinpoint DNA molecules
with Alex Knight, National Physical Laboratory
Alex - We're using lasers to look at single molecules, which could be DNA or proteins for example, and you can get a lot more information about molecules when you look at them as a collection of individual molecules. You can get a lot more information about how the molecules differ from each other and how they behave.
So what we need to see single molecules is we need very bright light sources and lasers are very nice in that they're bright, they're stable, and they're a single colour. So it's a very pure form of light that is very easy to work with and because they're parallel, and they form very nice parallel straight beams, they're very easy to manipulate and focus them exactly where you want them. And so, we use a property of molecules called fluorescence where we make the molecules emit light by putting special chemical groups - a tag - on the parts of the molecules that we want to detect then you shine a laser of one colour at them and they emit light of another colour. And that's a very sensitive way of detecting molecules and it's used for all kinds of applications in biology.
So, by having these tags on the molecules, you can do things like: you can count how many molecules are there, or if you use a special type of tag, you can use them to follow what the molecules are doing.
All you see of the molecules is a point of light and it's just like when you look up into the night sky and look at the stars, and it's the same with these molecules. You just see this point of light. But even that is enough to tell you quite a lot. So depending on what kind of experiment you're trying to do and what kind of tag you've put on the molecule, you can learn a lot just by looking at these points of light.
 Meera - So what have been potential applications then or techniques that have benefited from the use of lasers in this way?
Meera - So what have been potential applications then or techniques that have benefited from the use of lasers in this way?
Alex - Well, one example is much faster sequencing of genomes. So we talk about detecting single molecules but of course the nice thing is, you can actually look at many molecules at the same time. You use these tags I was talking about and you put a different tag on each of the four letters that you find in a DNA sequence, A, T, C, and G. And so, by using different lasers, you would see different letters and so, you could read out the sequence in that way.
Meera - So you would scan say, for all of the As and that a certain colour would be emitted, and then all of the Ts in a certain colour would be emitted and you'd combine that all together to therefore then get the sequence.
Alex - That's right. By doing that for many, many millions of molecules all at the same time, you can collect a huge amount of information.
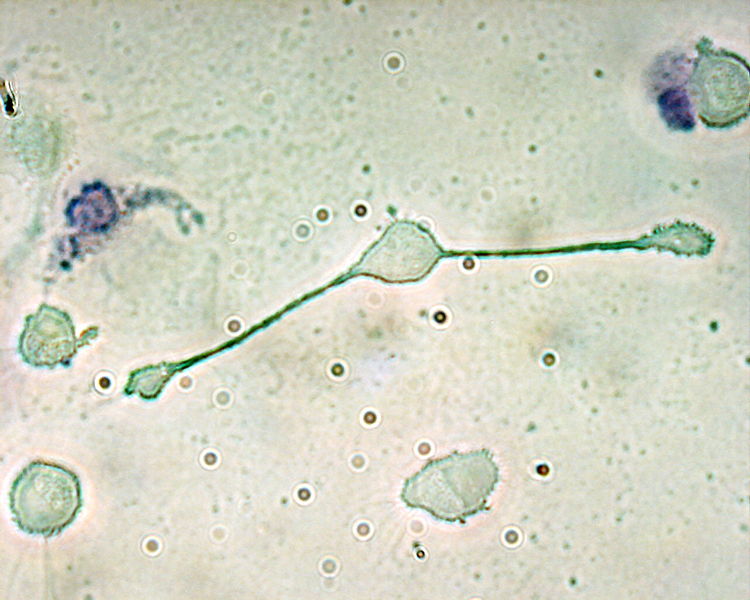
Meera - You've got something here on the screen which does actually resemble a night sky with occasional twinkles here and there, and this is showing us a new area that you're working on which is the field of nano switches. What are these?
Alex - So, one type of molecule we're looking at is actually an artificial molecule which is a switch made out of DNA and this is a collaboration we have with some guys at the University of Edinburgh and these DNA nano switches are very useful for detecting specific DNA sequences. When they bind to this sequence, they switch and you see that as a fluorescence signal that we can monitor with our microscope here.
The reason they're interesting is, you can use them for genetic testing, and the idea is, if you're looking for a particular sequence of DNA, say in a patient, these molecules only switch if they find the exact right sequence of DNA. When you look at the individual molecules, you can actually see this switching happen. And so, when you look at the screen, you can see these points of light and you'll see that they blink on and off, and that's the actual switching process happening.
Meera - And having developed these switches, you can then identify if particular sequences, say the sequence for a particular disease is present then by the fact these switches are switched on.
Alex - That's right, yes.
 Meera - But this principle, using fluorescent tags or probes to attach to specific sequences of DNA has been used before, so what makes these switches better? Are they more accurate?
Meera - But this principle, using fluorescent tags or probes to attach to specific sequences of DNA has been used before, so what makes these switches better? Are they more accurate?
Alex - That's the essence of it. The idea is that they're more accurate because they only switch when they bind to the exact right sequence of DNA because many of the genetic changes that people need to test for are changes of only a single letter in this genetic sequence. A lot of the methods aren't specific enough to detect just a single change very accurately and the idea of this is, if there's a change of a single letter in this genetic sequence, they won't switch.
Meera - So, other methods, say such as fluorescent probes will attach to a sequence even if one base is different because the majority of it is the same, so it will still attach. Whereas these switches weren't fluoresce unless the sequences match exactly.
Alex - It may attach, but unless the sequence is right, it won't switch.
Meera - What's involved with the switch in order to fluoresce and cause the twinkle that we can see here?
Alex - To measure the switching, we actually use two tags, and depending on whether those tags in the molecule are close together or far apart, we'll get a different signal.
Meera - So, attaching onto a desired sequence causes the switch to come together essentially and then emit the light.
Alex - Exactly.
Meera - What would be the potential applications then of this?
Alex - It's brought the era where you can contemplate the idea that everybody would have their DNA sequenced. A lot of people think that the future of medicine is what they called personalised medicine and that's where before you're given a drug treatment, you would have a genetic test to see what is the right drug for you and what is the right dose of that drug. And the idea is that you don't waste time or money giving people a drug that won't work for them, and of course, you don't give people a drug that will make them ill, that will have a lot of side effects. So the idea here is that in future, you would have this genetic test and the doctors would then know what is the best drug to give you.
32:41 - Laser tweezers picking up bacteria
Laser tweezers picking up bacteria
with Dr Clare Bryant, Cambridge University
Ben - A few weeks ago on the Naked Scientists, we heard how a highly focused laser beam can be used as if it were a pair of tweezers - it forms an "optical trap" that allows us to manipulate very, very tiny objects. Now this technology has allowed researchers to try some very novel techniques, such as finding out how cells respond to pathogens on a one to one basis. And we are joined by Dr Clare Bryant who's a Cambridge University researcher and she has been doing just that. Thank you for joining us Clare.
Clare - Thanks, Ben for having me.
Ben - Why is it important that we try and find out how cells respond to each other or how cells respond to pathogens on this one-to-one basis?
Clare - Okay, so a lot of work looking at host pathogen interaction has focused on a sort of population view of how cells respond to pathogens, and some of the work we've been looking at has suggested that in fact, cells don't respond in a universal homogenous kind of way. And actually, to explore exactly how a pathogen interacts with a cell, we need to do it as an individual cell-pathogen interaction, and this has revealed a number of very interesting things, so we're particularly interested in how macrophages (which are phagocytic cells which gobble up bugs) respond to pathogens such as Salmonella which causes food poisoning. And we've found that in fact, despite the fact we assumed that all macrophages will become infected by Salmonella, in fact, they don't. Most of them in fact aren't infected at all which is very bizarre, considering that a macrophage should be the cell that gobbles up all the bugs that are out there.
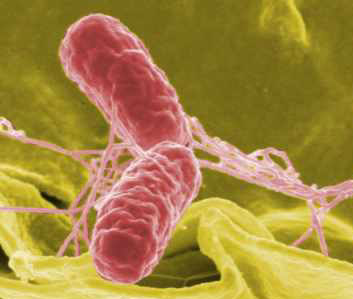
Ben - So, so far we've really just been looking at the average. We've been looking what happens with this whole population. If I can just get into the laser because I had a go with one of these, it's called a holographic assembler a few weeks ago in Bristol and I was amazed of the fact that I was moving these tiny, tiny beads of glass that are a third the size of a red blood cell, but I also had to wear eye protection in order to be in the room. So how do you adapt to laser so that it doesn't damage cells?
Clare - Okay, so what we've done is we use a laser which is set to a wavelength of 1, 064 nanometres and that's actually within the infrared range of lasers, and this has been shown to be able to be used to manipulate cells and the cells remain viable. They remain able to proliferate. And therefore, we're reasonably confident that that's not actually going to damage the cell. The other thing we do of course, is we enclose the cells and the bacterium within an environmental chamber with a gas , the humidity level and the temperature is controlled so that everything is set up for maximum cell viability and bacterial viability, but it's really used in the laser in the infrared spectrum which should reduce any damage to the cell and the bacterium.
Ben - So you've already mentioned that we've looked at how macrophages, big eaters, cope with Salmonella and the fact that some of them don't do quite what we expect. What sort of things can we actually hope to learn from understanding these one-to-one interactions?
Clare - So, we're very interested in how the macrophages are actually able to take up the Salmonella and how the Salmonella is actually able to get into the cell. So, we're going to be able to do things at two sort of levels. So first of all, actually just picking up a bacterium which is actually quite a challenge in its own right because they move very, very fast. It's like a massive computer game, trying to catch these things. They have tails which makes them spin and roll, and run.
Ben - They're some of the fastest things in the world for their body length, are they?
Clare - They are indeed. Yes, they are indeed. You have to be very, very fast with your tweezers. Once you've caught them, you then are able to take them up to the macrophage. You're able then to look not just at how the bug and the macrophage sort of interact with each other. We can measure the physical processes that are involved so we can measure the time it takes for the bacterium to be taken up by the macrophage. We can also try to explore the kind of forces that are involved which is an element of bacterial infection we haven't really considered in any way at all. And then the other thing we're able to do is we're able to use cells that lack specific receptors that we believe are important for taking up the macrophage, taking up the bacterium rather, so then we'll be able to see, okay, how do these receptors contribute to the process of the Salmonella uptake, but also to the physical processes that may be involved. And the counter to that is we can take bacteria that lack the specific proteins that are important for uptake into the cell and see how that really affects not only the physical interaction with the cell, but the uptake process itself. So we're going to be able to understand the whole process that's involved. Further to that macrophages are existence sort of a number of different phenotypes, so we'll be able to take different types of macrophage and see which ones are actually important for taking up the cells, which ones are important for taking up the bacteria and allowing them to grow, and which ones don't take them up at all and try and explore what the physical difference is in their cells actually are and why that affects the ability of the Salmonella to get into the macrophage.
Ben - Can we use this trick to be even more specific as well? I was mentioning manipulating these tiny glass beads. Can we just coat them with say, one particular protein we're looking at and then find out exactly how a macrophage responds to just this protein without all of the other factors that complement it?
Clare - Yes. So some of the preliminary work we did was to look at a toxin which is present on Salmonella called endotoxin and we're able to coat beads with endotoxin, and endotoxin is believed to be anti-phagocytic and what we were able to do was to compare beads specifically coated with endotoxin, (the beads were the same size as the bacteria) and then compare those to beads that had been coated with endotoxin, and an antibody which is important it's called an opsonisation process which enhances phagocytosis. And what we could see if we compared uncoated beads with endotoxin coated beads and opsonised beads was that the endotoxin beads were much, much, much slower to be taken up into the macrophages whereas the opsonised beads were taken up really quickly, as were the beads that didn't have any coating at all. So there's a whole plethora of experiments we should be able to do to allow us to precisely investigate those kind of processes.
Ben - And what is the next step for you? What's the goal you really want to chase at the moment?
Clare - So the goal for us at the moment is to really explore why some macrophages take up Salmonella and some macrophages don't take up Salmonella, and then the key question for me as a biologist that I'm really interested in is to explore which of the receptors are important. I'm particularly interested in receptors that recognise various pattern associated molecules that sit on the outside of the bugs, and they drive the innate immune response of a macrophage to the bugs. So I'm really interested on how those receptors specifically affect the uptake of Salmonella into the cell, and whether or not there's a link then between the uptake of the Salmonella into the cell and the immune process then occurs downstream of that.
Ben - Well thank you ever so much. We could have to leave it there. That's Dr. Clare Bryant from Cambridge University. She is using laser tweezers to really look at something we've never looked at before, and that's the way that cells respond to pathogens on a one-to-one basis.
How does a biologist end up working on lasers?
We asked this question of Clare Bryant, a biologist who now works in a multidisciplinary team working with lasers...
Clare - It was quite interesting, actually. I started doing some work with a colleague of mine called Julia Gog in the Maths Department and we had a few beers one evening and we were chatting about the sort of basic principles that were involved in how Salmonella infect cells. She asked me a lot of really awkward questions and I was sitting and thinking, "Well, do I know the answers? - Well I think I do". I went away and looked in the literature and in fact, not many of the answers were actually known.
So we started to talk about this and then we started to collaborate with a physicist called Pietro Cicuta and when we had our experimental questions, Julia would say, "What about this?" And I say, "Well I think it does that, but I can't quite build the kit to do it so I don't quite know how to do this." Pietro said, "It's all right. I can build a piece of gear!"
So we started the three-way collaboration with awkward questions. My ideas for biological experiments and Pietro then saying, "Yeah, we can do this. We can do this this way, that way or the other." It's then proven to be just really, really exciting to do experiments. I never dreamed it possible, to be honest.
Ben - So, this is how the real novel stuff comes about, isn't it? It's when researchers from different fields get together, maybe over a beer, maybe for coffee perhaps, and this fantastic fabulous research comes out.
Clare - Yeah. It's just amazing to me. I hadn't been sure how multidisciplinary work would really happen and having had this kind of interaction with these guys, I'm now trying to learn their language. Obviously, the mathematical language is very, very different from the biological language, and they (Julia and Pietro) are embryo biologists, but the kind of interaction between the three of us is just proving to me to be a real eye-opener and an obvious way forward for us.
What will happen to a marble dropped down a core through the Earth?
We've answered this question before - here and here...
Ben:: What the theory suggests is that if you were to drop a marble or jump down this hole, you would accelerate as you get towards the centre of the Earth, and then when you pass the centre of the Earth, you'd start to decelerate [accellerate in the opposite direction - for physics pedants]. But because of the energy you'd built up, you would in fact go all the way through to the other side. Now, if you're lucky and had something to grab hold off then you could get out on the other side, but if not, then the same thing would happen again. You would turn back and you'd oscillate one way then the other and just bounce around between sides of the Earth. The theory is it should take around the same amount of time that it takes to orbit the Earth, so somewhere around 45 minutes from one side to the other.
You might notice from our previous answers that this assumes the tunnel to be evacuated of air - as the air pressure at the core would be incredible, and you would experience very high drag - a marble would be fine, but could you hold your breath for 45 mins?
Could you grow bacteria on an iphone?
Helen - I don't have a touch screen. I've got an old fashioned one with buttons, but I imagine the same thing would count with buttons as well as touch screen.
Ben - I think it probably would.
Helen - I suppose it all comes down to how well you wash your hands after you've been to the bathroom - what kinds of bacteria that pass through our systems end up on our hands and on our phone. Will they survive and live on our phones? That's the question I suppose. Would they be able to actually form colonies?
Ben - Well bacteria can survive quite a long time actually on various surfaces and we know of around about 1,000 different bacteria species that will live on our skin. I think you probably could tell the key areas that are touched most because you're going to put down more grease on there, you're going to put more skin cells which will act as food, and you're going to put more bacteria on there, so yes, possibly. It could be a very good way to find out what buttons people press most often which is really quite disturbing when you think about it that way...

Why does an ice cube melt in a glass of water?
Ben - I regret that Dave Ansell isn't here because he would give us a wonderful and distinctive answer in his very individual style, but I'm going to give it a go.
I'm not a physicist, but what I think happens is you get an equilibrium between molecules of water that are frozen in the ice and molecules in the water, and they will move back and forwards. So constantly, you get these molecules that are becoming water or forming back into the ice structure. This equilibrium is temperature dependent. So, when it's above a certain temperature, more of these molecules are going to come out of the ice structure and into the water structure, and this means in total, you'll get less ice, more water, and eventually, the ice cube will melt.
Now of course, the interesting thing is looking at the fact that because of the density of ice, if you put an ice cube in a very full glass of water, so much of it is floating above the surface that when it melts, your glass of water still won't overspill.
Helen - It's actually a really important question as well, not just through ice in our glasses, but ice in the sea and why that doesn't melt or when it does melt, and how that will cause or won't cause changes in sea level as well. So I think that's a really, really big question from what sounds like something that's just about having a nice cool drink on a hot day. Actually, it's got really big implications for all sorts of things going on in the natural world.

52:38 - Why was the New World slower at developing technology?
Why was the New World slower at developing technology?
We put this question to Jeff Oliver, lecturer in Archaeology at the University of Aberdeen...
Jeff - First, I think it's important to say that the popular perception would have it that the Americas were a backwater to the technological prowess of Europe. But I think this is actually very simplistic and perhaps an unfair stereotype. Speaking very broadly, part of this has to do with the development of very complex cultural, economic, and political formations which partly hinge on the development of agriculture. In fact, in parts of the Americas and Europe we see very similar kinds of development. Now in terms of things like guns and armour, things like that which perhaps allowed the Europeans an advantage early on, the difference here comes down to a number of factors that separate Europe from America. Europeans were exposed to gunpowder - invented in China and effectively improved upon through the development of science, but I think once again, it's very simplistic to say that the Americas are not as technologically sophisticated in other terms.
Diana - So sometimes, you just have the sort of events that lead to you discovering how to make iron or how to make guns and this can change your entire technological development, but if agriculture is indirectly an important step in the development of technologies, why might the old world have developed agricultural before the new world?
Jeff - It is interesting for instance, yes, we see earlier forms of agriculture in the Middle East. We see it much later in North America. This has got nothing to do with a unilineal notion of progress whereby all societies should follow on this sort of similar ladder of progress. It comes down to very specific historical situations. Probably in the Americas, it has something to do with fewer people. We see less of a need for the development of agriculture earlier on in the Americas because we have less of a population than in places like Europe. So it's almost certainly coming down to a situation like that. Diana - It may have been a lack of population pressure that meant agriculture came to the Americas much later. The founding population of humans there is thought to have been very small indeed, at about 15000 years ago, but there could have been a whole load of other factors like geography and climate.
But we can't really say that the Old world was more advanced than the New when some societies there could still forge some rather impressive things out of gold or weave very fine textiles. We had a few emails this week mentioning the guns, germs and steel advantage of European conquistadors. And Deogratias said that trying to get the trajectory of new world societies to fit that of old world ones doesn't really work, but if you ask the question the other way around - why were the European invaders not so advanced? You'd be asking about the differences in morals and values.
- Previous The Proton is Shrinking!
- Next Why do Moths Fly Towards Light?











Comments
Add a comment