What happens when a blacksmith meets a metallurgist? This week we explore what's happening at the molecular scale when the smithy works a piece of iron, we meet the superalloys that survive temperatures way above their melting points inside jet engines, and at the Rolls Royce precision casting facility we discover how precision plane engine parts can be cast from a single metal crystal. Also, in the news this week, how the blind brain has a built-in sonar, an attractive new magnetic material turned on by a current, and a new technique to detect troublespots brewing inside arteries. Plus, Kitchen Metallurgy - an experiment to show how you can manipulate metal molecules for yourself!
In this episode
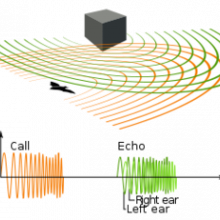
01:27 - How human brains can echolocate
How human brains can echolocate
Researchers from Ontario this week have identified which parts of the brain are involved in human echolocation. Traditionally associated with bats, whales and sonar, echolocation is a technique used to find one's position by bouncing sound off surfaces and waiting for the echo.
 The study, performed by Lore Thaler and colleagues at The University of Ontario and the Rotman Research Institute, compared the brain activity between two study subjects. One had been blind since thirteen months; the other had developed blindness in adolescence.
The study, performed by Lore Thaler and colleagues at The University of Ontario and the Rotman Research Institute, compared the brain activity between two study subjects. One had been blind since thirteen months; the other had developed blindness in adolescence.
Both these individuals used clicking sounds, made with their own mouths, to glean information about their surroundings. Both blind subjects were able to tell when a panel placed before them was flat or concave and whether it was 20 degrees to the right or left. Outdoors, they could tell if they were standing in front of a car, tree or lamppost. But, the researchers had to overcome the problem of echo-locating inside the brain scanning fMRI machine, where it's noisy and there's nowhere to go. So what they did was to pre-record echolocating sounds from microphones at the subject's ears and then played the recording back to them inside the machine.
Publishing in PloS One, they found that in both blind subjects the calcarine cortex, an area of the brain normally dedicated to processing visual information in sighted people, displayed greater activity when the subjects listened to the echo sounds. This implies that they could see something from the sound of the echoes. This was compared to fairly constant levels of activity in the part of the brain typically used for processing auditory information when they listened to the echoless clicks - so it does look like they're seeing with sound rather than hearing.
03:52 - Attractive new magnetic material
Attractive new magnetic material
A material that temporarily transforms itself into a "permanent" magnet when a small voltage is applied has been discovered by scientists in Japan.
Although substances with similar behaviours have been described  before, these have worked in this way only at super-low temperatures close to absolute zero. But what sets this new material apart is that it exhibits this magnetic phenomenon at room temperature.
before, these have worked in this way only at super-low temperatures close to absolute zero. But what sets this new material apart is that it exhibits this magnetic phenomenon at room temperature.
Tokyo University's Tomoteru Fukumura and his colleagues, who have announced the discovery in this week's edition of the journal Science, made the new magnetic material by replacing 10% of the titanium atoms in titanium dioxide (TiO2) with cobalt atoms. The resulting titanium cobalt oxide compound, they found, is naturally "paramagnetic", meaning that it is weakly attracted by an applied external magnetic field. But, if a small voltage is applied, then the material becomes magnetic in its own right.
By making fine measurements of an entity called the anomalous Hall Effect, the team discovered that, when electrons are pushed into the substance by a voltage, the electrons of the cobalt atoms are triggered to all adopt the same direction of spin, which is what underpins the magnetic field created by a ferromagnetic material. This effectively turns the material into a "permanent" magnet, at least until the voltage is shut off.
Dubbed "chameleon magnets" owing to their ability to change their appearance in this way, the discovery is very exciting because it has the potential to open up a range of applications in an emerging field known as spintronics.
For instance, electrons can tunnel from a magnetic source material through a barrier to a second magnetic target much more easily if the two have the same magnetisation. And so a magnet that can be turned on and off could provide a miniature microswitch or transistor.
Magnetic materials also alter the polarisation of light reflecting off their surfaces. So if a magnet can be turned on and off very quickly and with high resolution, coupled with a polarising filter such a system could be used to control the brightness of a light source very rapidly in order to send a greater density of data down the fibre-optics that underpin modern communications networks, including the Internet.

06:47 - The Shape of the Electron
The Shape of the Electron
with Jony Hudson, Imperial College London
Diana - What is round and measures 1 billionth of a millimetre across? Yes, you guessed it, it's the electron. Theories have predicted that these particles should be spheres, but proving this has been tricky. Now after 10 years of trying, a team at Imperial College London have succeeded, as Jony Hudson explained to Chris.
Jony - So what we've been working on is we've been trying to measure the shape of the electron. The conventional theory suggests that the electron is round like a point particle, but we've been wanting to check that and to see whether it really is round.
Chris - Should that really make a difference?
Jony - Yeah. It's important actually and the reason it's important is because although our current theories predict that the electron should be round, some of the advanced theories that go beyond that predict that it won't be round, that it'll have a distortion. And so by doing this measurement, we look at it as a way to search for physics beyond the physics that we know about at the moment.
 Chris - And just to give people some grasp as to the scale of the problem that you're grappling with, how big is an electron?
Chris - And just to give people some grasp as to the scale of the problem that you're grappling with, how big is an electron?
Jony - The size of the electron, it depends how you measure it. The classical, what they call the Compton wavelength is one measure of the size of the electron, and that's 10-12 metres, so 1 billionth of a millimetre.
Chris - Pretty small in other words.
Jony - Yes.
Chris - How on Earth do you go about trying to size up something like that?
Jony - If the electron were perfectly round, when we put it in an electric field, it would just spin around its axis in a perfectly regular way, whereas if it's not round when we put the electron in the electric field, it will develop a kind of wobble, a very distinctive motion where it's wobbling around its axis. A lot like if you were to put a gyroscope on a stand at an angle, that kind of wobbling motion. And so we look for that wobbling motion.
Chris - Is that because the electron itself is an electrically charged particle? So if you have something which is electrically charged moving, and it's moving in an electric field therefore, the two fields are going to interact and that's going to impart a movement on the particle that you can then pick up?
Jony - Yeah, that's basically it. If the electron were round, then it wouldn't matter which way it was oriented with respect to the field because it would just look the same. If you turn a perfect sphere around a bit, it still looks the same. Whereas if the electron say if it were egg-shaped, it would have a bit of distortion to it, it matters which direction it's pointing in relative to the field and so, there's a force on it, trying to align it with the field, and it's that force which creates the wobbling motion.
Chris - So how did you actually do it?
Jony - So what we do, we don't just use a bare electron because there are some real technical difficulties with working with electrons, namely that they're electrically charged. So what we did instead is we used the electrons in a particular molecule, a molecule called ytterbium fluoride and this molecule has an unpaired electron orbiting around the outside of it, and that's the electron that we studied. What we look at is if the electron isn't round, it starts wobbling in its orbit around the molecule. So actually, what we really look at is we look to see how the molecule spins.
Chris - And what do you find?
Jony - What we find is, as best we can tell and we've looked really very carefully, there's no wobble. The electron shows all signs of being round at our current sensitivity.
Chris - So that means or that must have quite big implications for other things in the quantum realm then?
Jony - Yeah. Like I say, our current theory of physics predicts that the electron should be really almost exactly round. And so, the first thing to say is our work doesn't contradict our current theory of physics which was - I guess a disappointment for us, it'd be much more fun if it did contradict the theory, but you can't have everything! What it does do is if you look at some of these theories. You know, I said people propose these theories that go beyond our current theory of physics. Some of them predict that the electrons should be really quite distorted and actually, we've shown that it's rounder than some of those theories would predict. So what's it allowed us to do is it's allowed us to constrain and guide the development of theories that go beyond our current theoryof physics. It places limits on what theories could possibly be right.
Chris - And do you think all electrons are made equal? What I mean by that is, in the context in which you studied the electron, it behaved like that, but what would happen if you took a different molecule as the donor and studied that? Do you think you might be able to get an electron that was distorted?
Jony - No, no, no. There are no forces in the molecule that can distort the shape of the electron. What we're measuring here is how the electron comes from mother nature. The molecule itself is not placing any forces on the electron that are distorting it.
Chris - So you've nailed that one, myth busted, but are there any actual physical applications now that you are armed with this knowledge that you can use to take this forward?
Jony - Well one thing that physicists are very interested in is there's a big mystery in cosmology, a big mystery in describing the origin of the universe, and the mystery is what they callthe matter-antimatter imbalance. So our current theory of physics, the best one we have, says that in the Big Bang, matter and antimatter were created in equal measure. Further, our theories of physics says that the laws that govern antimatter are basically the same as the laws that govern matter. And so, the logical conclusion would be that we should have an equal amount of antimatter and matter today. But astronomers have looked and you can search the skies. You can look wherever you like. You find only incredibly tiny traces of antimatter and this is a big mystery for people.
Now one potential solution is that there's a slight difference in the behaviour of matter and antimatter, and this would mean over the billions of years that the universe has existed, it could tip the balance and the matter could start to dominate over the antimatter. If the electron is not round, then you can show that it's not possible for it to behave the same as its antiparticle, the positron. So if the electron is not round, this would indicate that there has to be a difference between the behaviour of matter and the behaviour of antimatter. And so, that's one of the motivations for studying this. People are looking for a difference between the behaviour of matter and antimatter. They think there must be one that we haven't discovered yet.
Chris - So could you take your technique and apply it to the antihydrogen that has been successfully made at CERN and ask that very question?
Jony - That would be a fantastically interesting experiment, but I would say our experiment was so difficult that it took us over a decade of extremely hard work to do. Their experiment is so difficult that it took them over a decade. If you sort of multiply them together, I can't even begin to imagine how difficult that would be!
Chris - Jony Hudson from Imperial College and amazingly the precision with which they say they've made those measurements is equivalent to, were you to scale the electron up to an object the size of our solar system, their measurements are precise to within the thickness of a human hair. Amazing piece of work. It's published this week in the journal Nature.

13:39 - Being a Culture Vulture makes for a Happy Vulture
Being a Culture Vulture makes for a Happy Vulture
People who take part in or attend 'cultural' activities tend to have better physical and mental health, according to a new study out of Norway...
Publishing in the Journal of Epidemiology and Community Health, Koenraad Cuypers and his team sent surveys to tens of thousands of people in a rural region of Norway asking questions about their activities, happiness and perceived health. The researchers then took the results, ran some statistical tests and found this association between cultural activities and health.
Cuypers, who's from the Norwegian University of Science and Technology, found that it was especially men in whom this relationship was most apparent. More than females, males reported higher levels of satisfaction, happiness and better perceived health if they took part in the receptive activities such as watching sport or going to a museum.
They also found that people in lower socioeconomic groups were less likely to attend either type of cultural activity, and that women were especially happy if they took part in more creative forms of culture. In addition, people tended to take part in more cultural activities the older they became up until they reached their forties, when it started to decline again.
The researchers stress they can't identify a causative relationship; they don't know if a bit of culture makes you happier or if happier/healthier people tend to involve more culture in their lives.
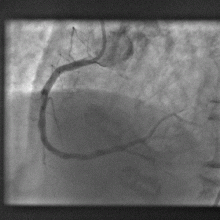
16:33 - Bottleneck cleared in clogged artery research
Bottleneck cleared in clogged artery research
A dye used medically for over fifty years could be a shot in the arm for investigating coronary artery disease, scientists have found.
Writing in Science Translational Medicine, a team from Boston, US, led 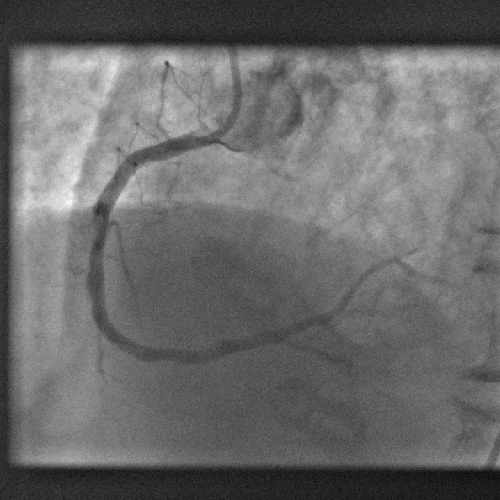 by Harvard Scientist Farouc Jaffer have discovered that indocyanine green (ICG), chemical formula C43H47N2O6S2Na, which has been used safely for over five decades to measure blood flow through the heart and liver circulations, can be used to highlight inflammed atheromatous hotspots threatening trouble in narrowed arteries.
by Harvard Scientist Farouc Jaffer have discovered that indocyanine green (ICG), chemical formula C43H47N2O6S2Na, which has been used safely for over five decades to measure blood flow through the heart and liver circulations, can be used to highlight inflammed atheromatous hotspots threatening trouble in narrowed arteries.
The team make the point in their paper that, although there are techniques for studying the progression of arterial disease in large calibre vessels like the aorta and iliac arteries, there is currently little on the table to aid in the identification of potentially troublesome areas for smaller arteries like the coronaries.
Seeking to address this challenge, the researchers surveyed a range of chemical tracers that might be helpful in identifying patches of rapidly progressive arterial disease. Such regions, which show more intense inflammation, are more likely to trigger thrombosis (clotting) and blockage of the vessel and are therefore a higher clinical priority for treatment.
Being highly lipophilic (fat-loving) the team wondered whether ICG might meet their needs and flag up fatty deposits in the vessel walls. It also absorbs and emits near-infrared light, making it a useful tracer molecule.
The team injected the substance into rabbits with damaged aortas, which are similar in size to human coronary arteries. Under a mciroscope, the ICG was found to bind with high selectivity to the diseased segments of the vessel. Moreover, it bound best in regions that were showing signs of rapid disease progression.
To determine how such an agent could be used for human imaging, the team inserted a probe into the aortas of a further group of animals and used near infrared imaging to plot the pattern of ICG signal coming from the inside of the vessel wall. This, they found, was a very close match with two other imaging techniques, one using ultrasound and another x-rays.
This shows that ICG could be used as a highly-effective and safe tracer to sniff out the smoking gun underlying arterial disease and therefore help cardiologists to direct their attention to those areas that need their input the most.

20:32 - Planet Earth Online - Science from the Skies
Planet Earth Online - Science from the Skies
with David Davis, James Johnson and Carl Joseph from the NERC Airborne Research and Survey Facility
Chris - Earth observation from satellites is an extremely important way of examining everything from glaciers to tree cover in the Amazon. But sometimes, satellite measurements aren't quite enough. Planet Earth podcast presenter Sue Nelson met members of the Natural Environment Research Council's Airborne Research and Survey Facility and spoke to pilot Carl Joseph.
Carl - What makes this aeroplane special is that it was actually manufactured as a scientific platform. So instead of really actually fitting the cabin area with seats, we've got a 2.1m hole in the floor that allows us to put a load of scientific instruments in.
Sue - One of the crew members is James Johnson who's an instrument operator. What instruments do you actually operate onboard this 'plane?
James - The instruments that we're going to operate which is the main core suite we have onboard are the LIDAR - the LIght Detecting And Ranging System - and the Eagle and Hawk sensors, which are hyperspectral systems looking at shortwave infrared and very near infrared.
Sue - Let's go on board the plan briefly before we take off... It's quite narrow isn't it, less than a couple of metres across, and we're both having to stoop.
James - The instruments down the end there, the sensor that you can see there, that is the LIDAR, which basically...
 Sue - The silver box.
Sue - The silver box.
James - The silver box at the end there, yeah. That will produce a digital elevation model of the ground. So it's a laser that fires down to the ground and we pick up the return and can actually map the ground and see the contours of the ground.
Sue - What would that information be used for?
James - I suppose that they would use the information for looking at various different things such as erosion. You can see the difference that something has moved, for example a glacier; you can actually see from six months ago if we've surveyed a glacier in Iceland or Greenland you can then go back and do another LIDAR survey and you can see how it's changed, its features have changed, over a period of time.
Sue - In front of the silver box, the LIDAR, we've got a bigger, sort of an old-fashioned 1990s stereo really isn't it, like a stack of black boxes.
James - Yes, that is the hyperspectral system. So what that does is looks at the ground again and it looks at it in the shortwave infrared and the very near infrared bands. So it's not looking at what you can see with your eyes specifically, it's looking at, for example, yes that's a green leaf but what kind of green is it, is it an oak tree or a beech or something like that?
Sue - That can tell the difference?
James - Yes, you can look at stuff like that.
Sue - Good grief! Now, the pilot Carl said there's a hole in the middle of the aircraft, but I can't actually see that hole, is that because this instrument is placed over the hole?
James - Yes, all of these instruments are placed over the trench that is in the middle of the aircraft. There's no danger of anything or anybody falling out.
Sue - Well I'm very glad to hear that because we're about to take off to do a calibration flight, is that right?
James - Yes, that's correct. What we're actually doing here is we are checking the instrumentation, checking it is working.
...
Sue - Well we're several thousand feet above Southwest England now. Rather unusually, we are sat with our backs to the pilot instead of facing the direction of travel which I must admit, even for somebody like me who loves flying, it's not that great on the stomach.
James - That's right. I know what you mean.
Sue - And you've got 6 computer screens in front of you as well as a more mobile flexible one in front of me where we can see the ground beneath us.
James - Basically, what we have here is the webcam. So this is a continuous reel of all of the ground that we're flying over and then the LIDAR, we have the LIDAR controls which is a laser, that's firing to the ground and taking an elevation model of everything that we are flying over. On this screens here, one of them is the eagle which is one of the hypospectral systems onboard, and the hawk. And as you can see, as we're going across the ground, it is looking on what we are flying over.
...
Sue - Remote sensing missions can last up to 5 hours. But all the information collected helps fill the gap between what scientists can obtain in both satellites and on the ground. Pilot and electronics officer David Davis.
David - There's a lot of work completed post-flight by our data node in Plymouth Marine Laboratory.
Sue - So it's not just land use, glaciers, climate change - it can be oceans as well?
David - Yes, we can cover everything from marine, archaeology, geology, ecology and anything in between. So if the scientists can think of a good project for it and put their application in, providing their science graded to a high enough standard, we'll fly it.
Chris - David Davis, James Johnson, and Carl Joseph from the Airborne Research and Survey Facility talking with Planet Earth Podcast presenter Sue Nelson.

32:56 - The Art of Alloying
The Art of Alloying
with Howard Stone, Cambridge University
Diana - We humans have been moulding metal to our will for at least 3,000 years. In fact, our use of metals mark the beginning of the aptly named Bronze Age. These days we fine tune the properties of metals to perform in extreme circumstances - inside nuclear reactors, jet engines, rockets, et cetera. To find the right combination of properties, we rely on metallurgists like Howard Stone at Cambridge University's Department of Materials Science and Metallurgy.
Howard - Well one of the first things I don't think people often get much an appreciation for is the fact that metals are actually crystalline materials. In other words, if you look at them on a very microscopic scale, at the atomic scale, you will see that the individual atoms are arranged in a very precisely defined periodic array or pattern. What you have to imagine if you were looking at your piece of cutlery or the tip of your ballpoint or even that legs of your chair is that the metal itself is built up of all these little individual crystallites, exactly or analogous to grains of sand on a beach or sugar in a bag of sugar. Only that there was no space between the grains. It's been, if you like, pressed together so that there's no distance between these little individual crystallites.
Diana - But not all of the crystals in any given metal will be the same.
 |
| Body-centred cubic structure |
 |
| Face-centred cubic structure |
Howard - The individual crystal structure, the different arrangements that can be adopted by these metals, have a profound influence upon the properties of the materials. And the presence of these phases as we refer to them - in other words regions of material which have these different crystal structures - does have a very important influence on the properties of metals themselves. It's our role as metallurgists to work out what the best possible combination of phases we can get out of a material is and therefore, the best possible set of properties that we can possibly achieve.
Diana - One way to alter the crystal structure of a pure metal is to add other elements. Steel for example is a mixture of iron and a small amount of carbon, but its properties are surprisingly different from both original materials.
Howard - If we add any other element to any given element, metallic element, so if we for example add carbon to iron, then we might expect to form new phases. In steels, we can expect a whole series of carbides as a possibility, and the morphology and crystal structure of those individual carbides can be very different, depending upon the conditions in which it's been processed and also the process of other alloying elements, and that will have a very profound effect upon the properties of the resulting material.
Diana - These mixed metals or alloys may be able to perform a given task far better than the pure metals and as such, are found all around us.
Howard - Well if you look at your average piece of tableware cutlery, you're probably looking at a steel, so an iron based alloy, which contains significant quantities of principally chromium and nickel. If you look at an aeroplane for example, most of the air frame will be made of aluminium alloys. The engines themselves may very well contain far more exotic things like titanium and nickel alloys as well.
Diana - For extreme environments like the high temperature high torque conditions inside a jet engine, we turn to a subset of alloys called the superalloys.
Howard - Nickel based superalloys justifiably deserve their name because they're the only class of materials that are able to operate at a combination of very high temperatures and very high stresses, whilst maintaining their surface stability. i.e. not being degraded too badly by the environment in which they operate.
Diana - But what is it that makes these particular alloys so good?
Howard - They're based principally on a combination of nickel and aluminium. So nickel is an element which adopts a face centred cubic structure. In other words, if you looked at the atomic scale, you'll see the atoms arranged in little cubes with the atoms on the corners of those cubes and then one in the middle of each face. Now with the addition of aluminium, the alloy will form precipitates, in other words, an additional phase which comes out of the material, embedded within the material, which are Ni3Al. In other words, for every 3 nickel atoms, you'll find another aluminium atom and they adopt a very precise crystallographic arrangement relative to each other. If you imagine my little cube again, I will have aluminium atoms in the corner of the cube and now nickel atoms in the face of each cube. Because of the similarity of that particular crystal structure with the nickel itself, these precipitates are able to form in very good registry with the matrix in which they form, and this creates a real barrier to motion of atoms past each other through the crystal structure and really resists deformation at very high temperatures.
Diana - Making the most of superalloys can involve adding small amounts of very exotic metals which can improve the properties but as is the case with exotic things, this comes as a cost.
Howard - Well over the last 50 years, we've done everything we can as metallurgists to improve the properties of nickel based super alloys by considering all the possible alloying elements we can reasonably include. Commercial nickel based super alloys for turbine blade applications for example, now can contain 9, 10 or even more elements in very precisely controlled quantities to get optimal properties. Some of the latest elements we've considered including include things like ruthenium which is a very rare platinum group metal. And that has extended the temperature capability that super alloys are now able to operate beyond those of previous generations. But these are very rare, very expensive elements. The real challenge for us is to identify whether there are any other alternatives to nickel based super alloys and that is a challenge which many people have been investigating over the last 50 years.
Diana - That's Howard Stone, assistant director of research at the Department of Materials Science and Metallurgy at Cambridge University.

39:08 - Metal Casting with Precision
Metal Casting with Precision
with Paul Withey, Casting Specialist for Rolls Royce
Chris - This is the Naked Scientists. I'm Chris Smith. If you've been on a flight recently, then there's a very high likelihood that you were carried aloft by a jet engine. These work by compressing air and squirting in fuel which then burns, expands, and generates thrust. But this also creates one of the harshest imaginable environments inside the engine where the gas stream routinely exceeds 1500 degrees Celsius - that's well beyond the melting point of standard metals. To withstand this takes specialist superalloy components which are produced with a very high precision and to find out how they're being made, Ben Valsler has been to the casting facility of one of the world's top jet engine manufactures, Rolls Royce in Derby, where he met Paul Withey.
 Paul - I'm the casting specialist for Rolls Royce and this facility is the precision casting facility in Derby. It's Rolls Royce's largest foundry and it makes turbine components for large civil engines and military engines. This is one of the single crystal facilities in the world which is one particular branch of precision casting that makes only single crystal components and the benefit from an aerospace perspective is that if you take out all the boundaries between each of the crystals, you get the strongest possible metal that you can, and that's really needed in the hot part of the engine. So like a sugar cube when you crush it, you actually don't break each of the individual grains, but you break the boundaries between the grains. It's the same in aerospace metal, so we make single crystal components to get rid of those grain boundaries and have the strongest part we can.
Paul - I'm the casting specialist for Rolls Royce and this facility is the precision casting facility in Derby. It's Rolls Royce's largest foundry and it makes turbine components for large civil engines and military engines. This is one of the single crystal facilities in the world which is one particular branch of precision casting that makes only single crystal components and the benefit from an aerospace perspective is that if you take out all the boundaries between each of the crystals, you get the strongest possible metal that you can, and that's really needed in the hot part of the engine. So like a sugar cube when you crush it, you actually don't break each of the individual grains, but you break the boundaries between the grains. It's the same in aerospace metal, so we make single crystal components to get rid of those grain boundaries and have the strongest part we can.
Ben - So what's the actual process? How do you go from raw metal to jet engine parts?
Paul - There's a number of different stages. If you imagine the component as not being a solid component but being a hollow component, we have to make a ceramic model of the hole we want. So we make that component called a core, inject wax around it, so we end up with the wax model of the component with the hole in the component filled with ceramic. Then we take that model, we'd go into what we call the shell room where we actually cloak it or clothe it in ceramic, and then we'll replace the wax with the metal and then there'll be the solidification process that's the heart of this process that means we can grow a single crystal. We have to pour in a vacuum so everything is hidden away behind steel walls.
Ben - Could we have a look anyway?
 Paul - Of course. Let's go.
Paul - Of course. Let's go.
...This is what we called a wax room. It's a room in which we take the initial ceramic cores that we talked about earlier and we're going to inject the wax around them. The room is a mixture of wax injection machines where we're actually putting the steel dyes in there and injecting the wax into the shape we want, and assembly benches where we're taking those wax shapes and putting them into the orientation on the runner system that we need for the further processing of the parts.
Ben - So now, can we follow some of these wax structures through into the next stage?
Paul - Of course we can.
Ben - We've come through a large door and into the second stage. The last room was very warm. It smelled of melted wax. In here, it feels a lot colder, but it's also a lot noisier. What happens in here?
Paul - Now this is a shell room. It's actually no colder. It just feels colder because of the controlled humidity, but actually, it's held at exactly the same temperature so the wax is exactly the same size in one room as it is in the next, and what we're going to do is take those wax assemblies, we're going to clean them, wash them, and then we're going to dip them in a ceramic slurry, and then take them out, drain them, and then sprinkle grit on them, and then allow them to dry for a couple of hours and then bring them back and do the whole thing over and over again up to 10 times.
Ben - So, the slurry is sort of the sandy stuff that will turn into your ceramic layer.
Paul - It's actually a liquid but actually has a bit of a ceramic glue in there called a silica sol that will allow that all to stick together and the bulk of it is delivered as a gritty dry sand sprinkled on top of the wet surface.
Ben - So how thick a layer of ceramic do you end up with?
Paul - It's normally somewhere in the region of about 5 millimetres.
Ben - And where do we go to next?
Paul - The next stage of the process is to take the wax out and then move on to casting.
 Ben - So the wax moulds are now invested with their layer of ceramic and we've come through into a very, very warm room. What happens in here?
Ben - So the wax moulds are now invested with their layer of ceramic and we've come through into a very, very warm room. What happens in here?
Paul - This is the room where we take the wax out. We wheel the moulds in, we close the door, we press the button, and in 4 to 5 seconds, we inject superheated steam at 180 degrees which allows the wax to melt out really quickly. The wax doesn't melt as a whole. It only melts on the surface and doesn't conduct heat well. If we did it slowly, the wax will expand more than the shell could take and we'd blow the shell off the outside. So this way, we get the wax out without breaking the shell.
Ben - And now, we're actually left with a ceramic mould that we can then pour metal into?
Paul - We now have a mould with a core in the middle of it held in space that's now ready to cast.
 Ben - And now, with the ceramic layer fired, we actually get through to the bit that involves molten metal, the bit that most people think of when you talk about casting. It doesn't look like I would imagine it would - I can't see these rivers of molten metal.
Ben - And now, with the ceramic layer fired, we actually get through to the bit that involves molten metal, the bit that most people think of when you talk about casting. It doesn't look like I would imagine it would - I can't see these rivers of molten metal.
Paul - The whole point of this process is that we control it as much as we can, because we have to take that mould, we lock it onto a copper cooled chill at the bottom and that allows this to have a very cold end of the mould. We then push the whole mould up into a chamber that's at around 1500 degrees C, allow it to bake there for about 20 minutes and then we melt a charge in a crucible above that, pour that charge into the mould itself. The mould at 1500 degrees C is above the melting point of the metal, so most of it stays molten except for the last little bit that hit the copper chill and then froze as lots of different grains, all orientations, hundreds and thousands of grains. We then take the mould and we draw it into a cold chamber so there's a temperature gradient from hot to cold and those grains all start to grow. It just so happens that the grains that grow the fastest are also the grains in the orientation we want for mechanical properties purposes. So by a natural selection process, they grow really quickly upwards and sideways, and kill off all the other grains.
Ben - So you're getting a single crystal because that one crystal structure happens to just be a bit more stable and perhaps outcompete the other crystal structures that could form?
Paul - That's correct.
Ben - So how long does it take for this single crystal structure to form?
Paul - It takes just over an hour to make a single crystal component of the size that we tend to manufacture and that's for the large civil engines of a component that's about 10 centimetres to 15 centimetres tall.
 Ben - So once they've been poured, once they've had a chance to set, where do we take them now?
Ben - So once they've been poured, once they've had a chance to set, where do we take them now?
Paul - They then move to the back of the facility where we'll cut-off each of the individual components and then use a strong alkali solution to remove the core which then leaves us with these castings with the right hollow inside them in the right place.
Ben - So once the products have come through, they've cooled, they've had all the bits of excess cut off, how do you check that they are what they need to be?
Paul - We have to check that they are a single crystal, and we'll do that by dipping them in acid and then looking at whether there's a grain structure there, and if there isn't, we've got a single crystal. We'll also check using x-rays to see whether they're the right orientation of crystal, we'll use gauges to check the external profile is correct, the length is correct, and that they're all at the right tolerances. We'll also use a dye penetrant to look for surface defects and we'll use x-rays to check for internal defects.
Ben - And how perfect do they need to be? What error margins do you have?
Paul - In terms of the crystal orientation, we've got a few degrees of orientation that we need to hit. In terms of dimensional requirements, the drawing usually requires us to be within 0.1 or 0.2 of a millimetre of the required position, and so, we'll check every blade to those kind of dimensions. And unlike certain industries where you can sample inspect, once we've got a metal part, every part goes through every inspectional operation.
Paul - This is one of the finished blades and this is actually gone through the next stage of the operation which is machining and the drilling in of the film cooling holes into the internal core passage. It's about 12 centimetres tall and it survives the really high temperatures and pressures and stresses inside the engine.
Ben - What conditions could that piece of metal now put up with?
Paul - Well this turbine blade itself will face conditions of the gas stream around it when the engine is operating being 250 degrees above the melting point of the metal and the stresses when it's spinning around at about 10,000 revs per minute is the equivalent of hanging a lorry off the end of it in a static case. This blade then has to take 750 horsepower or more out of the hot gas stream to power the upfront parts of the engine. So, each one is about the same horsepower as a formula one car. That gives this a component that will last more than 5 million flying miles and it's quite an arduous environment to live in.
Chris - Paul Withey, taking Ben Valsler around Rolls Royce's precision casting facility in Derby. Rolls Royce reckon that at least 200,000 people around the world are being held safely aloft by their engines at any moment in time. In other words, right now.

Stiffening Paper Clips - Metallurgy in the kitchen

53:23 - At what speed does windchill give way to frictional heating?
At what speed does windchill give way to frictional heating?
We put this question to Kevin Knowles from Cranfield University and Holger Babinski from Cambridge University...
Kevin - I'm Kevin Knowles. I'm professor of aeromechanical systems for Cranfield University and I work at the Defence Academy of UK in Shrivenham, Oxfordshire. In essence, the answer to the question is that aircraft wings are cold because they fly high in the atmosphere, the region known as the troposphere, and atmospheric temperatures fall with altitude in the lower atmosphere. So an aircraft flying at about 10 kilometres which is typical above the ground, a day where it's only 15 degrees Celsius at sea level, will be experiencing temperatures of about minus 50 Celsius outside.
If, however, you move fast enough in excess of the speed of sound then there's a significant kinetic heating effect and that's what spacecrafts experience. For example, if an aircraft were again flying at 10 kilometres altitude, but now at nearly at 7 times the speed of sound, then the temperature that it would feel due to kinetic heating, the highest temperature would be about 10 times the ambient temperature, but measured in degrees Kelvin.
Now, 10 kilometres altitude, the temperature in Kelvin is 223, so Mach 7 spacecrafts would experience 10 times that, so its heat temperature would be 2,200 Kelvin or about 2,000 degrees centigrade. At 10 kilometres, those two effects balance out if you're flying at about Mach 1.2, so at that speed, the kinetic heating is just enough to bring the peak skin temperature up to sea level temperature.
Diana - So it's not so much the wind-chill effect as the fact that planes fly around in the colder part of the atmosphere. Travelling at Mach 2, Concorde used to experience surface temperatures over 100 degrees Celsius causing the fuselage to extend by as much as a foot. But what if you're a cyclist?
Holger - Hello. My name is Holger Babinski and I'm professor of aerodynamics in the engineering department of Cambridge University. How much friction you need to compensate for the fact that the air is cold around you depends very much on the temperature difference between your body temperature and the air temperature.
You asked how fast you would have to cycle, so I shall assume that you're cycling at sea level. There, the surrounding temperature on a cold day might be something like 17 degrees. And in order to generate enough friction to bring the air up to body temperature, you have to cycle at about 63% of the speed of sound. With the speed of sound at sea level, that is pretty much 480 miles per hour. So you have to be pretty fast to achieve that.










Comments
Add a comment