How do you make a new metal? This week, we follow a novel alloy from PC to plane, finding out how computer modelling and design can help us create new metals with exciting new properties. We also discover how these newly-designed metals are forged, treated and tested before they form the basis of a new generation of jet engines. In the news, deep-sea dwelling bacteria that are still digesting a meal dating from the time of the dinosaurs, a shot-in the arm for ageing satellites and a brain-interface device to permit paralysed patients to control robotic arms...
In this episode

Satellite Rescue
Satellites are extremely expensive pieces of kit, even without the cost of launching them. A modern telecommunications satellite can cost billions of dollars. Unsurprisingly, their owners want them to keep functioning for as long as possible, however they have to maintain the right orbit to be useful, and as soon as they run out of fuel, their orbit will slowly start to change until they are useless. Satellites are solar powered and built to last, so that most satellites finish their lives perfectly serviceable, but just in the wrong place.
 A company called Vivisat is hoping to change this: they have designed a relatively small spacecraft called a Mission Extension Vehicle or MEV, which is powered by efficient ion thrusters. The idea is to fly the MEV up to the satellite, dock onto the motor which every geostationary satellite uses to get into circular orbit. The MEV then takes charge of pointing the satellite and keeping it in position, allowing it to keep working for several extra years.
A company called Vivisat is hoping to change this: they have designed a relatively small spacecraft called a Mission Extension Vehicle or MEV, which is powered by efficient ion thrusters. The idea is to fly the MEV up to the satellite, dock onto the motor which every geostationary satellite uses to get into circular orbit. The MEV then takes charge of pointing the satellite and keeping it in position, allowing it to keep working for several extra years.
This is another example of space starting to be used in a more innovative way, which hopefully will lead to exciting things in the future.

01:33 - Making Metals: Designing A New Alloy
Making Metals: Designing A New Alloy
with Professor Roger Reed, Birmingham University
Dave - Metal alloys are mixtures of different chemical elements. Adding certain elements can make a metal harder, others can alter melting point or help the metal to resist corrosion. But there are tens of metals to choose from, so how can we work out which to include and in what proportions? Professor Roger Reed, Director of Research at the School of Metallurgy and Materials at Birmingham University, joins us to explore this. So Roger, what's the traditional way of working out how to make an alloy?
Roger - Well, traditionally of course, a lot of so-called "bucket chemistry" has been used to find out which elements to use for the alloys. So you would typically mix up elements such as nickel and chromium, and aluminium and titanium, and so on for the nickel based superalloys, and then you would test them in mechanical testing apparatus, and work out whether one has adequate properties for a given application. So, for example, in the jet engine, you might be interested in making sure that the alloys have got sufficient strength or toughness, or even something as complicated as creep resistance. So a lot of empirical studies have been done by 'make test' iterations. You make the alloys, you test them and perhaps up to maybe a few hundred alloys, you have one which you think you can use.
Dave - That sounds like a hideously time consuming and expensive process.
Roger - Well, that's right. Many people spend lots of time and of course, lots of money. For a typical turbine disk for a jet engine - it may be up to a million pounds or perhaps even ten times that to have the alloy ready for use in the engine with all of the necessary test data measured carefully.
Dave - So, what are you doing which is different?
Roger - We've been working on computer modelling programmes to try to use theoretical methods to come up with what we think are the optimum compositions for the sorts of components for jet engines. So the idea is that you would do the necessary theory in calculations on the computer and then to choose the alloy composition which you think is best matched to the properties that you need for these applications.
Dave - So, are you starting off at the atomic scale with quantum  mechanics or doing something slightly different?
mechanics or doing something slightly different?
Roger - There are different ways to do it. So yes, certainly we make use of quantum mechanical methods; so-called density functional theory where a number of assumptions solves Schrödinger's equation to work out the bond energies for the different elements and in particular for the energy associated with the different elements swapping positions on the crystal lattice. So that's certainly something we do. We also use high level thermodynamical methods based on solution theory which have got now parameters in there which describe how the bonding behaves.
Dave - So essentially, you're using quantum mechanics to get figures which you then feed into another larger scale model which you then feed into another one?
Roger - We come up with what we call merit indices and those merit indices are figures of merit which describe how any given trial of synthetic composition would behave in creep, or in fatigue, or in strength. And we do that by making estimates for suitable defect energies which describe how easy it is for that alloy to deform. It comes down to a number of composition dependent energies which we can estimate using the theory that you just described.
Dave - I guess you're not just dealing with two metals in an alloy. You're dealing with many. Does this add more difficulty?
Roger - Yes, that's quite right. The kind of alloys that I look at in my research are the nickel-based superalloys. Those are the alloys which are used in the very hot parts of the turbine engine, of the type that you would use for a jet propulsion or for electricity generation in a power generation circuit. Those alloys are quite complicated. They typically have maybe 7, 8, 9 - maybe as many as 10 different alloying elements. We add chromium, for example, and cobalt. The chromium is added for much the same reason that you would add chromium to a stainless steel - to make it oxidation and corrosion resistant. We add also elements such as aluminium and titanium to promote precipitation strengthening. That makes the alloy start to behave a bit like a composite, so you have small particles of a second phase present which adds strength to the materials. Then we add other exotic elements, much rarer and more expensive refractory elements like tungsten, candidum, and even rhenium. And even some precious metals like ruthenium have been shown to confer very best properties particularly in creep, where one is interested in making sure that the alloys don't deform and stretch continuously over time under the stress that they experience in the engine.
So yes, there's quite of range of elements which are added, and this does provide the complexity which makes the computer modelling methods useful because if you think about it, each element needs to have a particular concentration associated with it. The concentration ranges are quite wide and with so many elements there, perhaps 8 or 9, or 10, the number of combinations that you can come up with synthetically is quite large. That's the power of computer modelling methods; we are a ble to do that sorting and ranking, and prediction on the computer without the need to go to the laboratory to make the alloys and do the testing. That does two things. It reduces the cost, but it also allows us to identify the compositions quickly. In a market where you need to get new alloys into the engine quickly, so that new products can be flown within perhaps the period of 1 or 2 years, rather than the traditional 8 or 9 years, that's very, very useful.
ble to do that sorting and ranking, and prediction on the computer without the need to go to the laboratory to make the alloys and do the testing. That does two things. It reduces the cost, but it also allows us to identify the compositions quickly. In a market where you need to get new alloys into the engine quickly, so that new products can be flown within perhaps the period of 1 or 2 years, rather than the traditional 8 or 9 years, that's very, very useful.
Dave - Are you fine tuning principles which people have worked out by the rough and ready system or are you working out new things which can go into the mixture?
Roger - New theories have been needed, yes. Of course, we are building on the shoulders of the giants as they say. Lots of people have worked on these sorts of alloys over the last 20, 30 years and of course, they can traced back to Whittle's very first engine which flew round about the time of the second World War. But theory has been needed to explain and to put in place composition dependent theory for the important properties such as strength, creep and toughness. I think in the last 5 or so years, that theory has matured and that's really what's allowed us to propose these new computer modelling methods to do the alloy design using theoretical methods.
Dave - So once you've got one of these, once you've run your models, what do you pass on to the guys that actually got to make the things?
Roger - Typically, we would suggest alloy compositions which we believe to be useful. That would mean the exact concentration in either weight percentage or maybe atomic percentage - percentage of the different elements there. So for example, for a turbine disk alloy, we may specify perhaps 15 or 16 weight percent of chromium in the latest alloys. You may then have smaller amounts of aluminium and titanium, tungsten and so on. We specify the compositions which we believe are required to lead to the very best properties.
09:32 - Making a New Metal
Making a New Metal
with Dr Nick Jones, Cambridge University
Ben - Once we have a recipe for a new metal, how do we find out if it lives up to its initial promise? Dr. Nick Jones from Cambridge University invited me along to the Department of Materials and Metallurgy to find out more about the process and he also joins us in the studio. Now Nick, thank you ever so much for coming in.
Nick - Absolute pleasure.
Ben - So, we've heard from Roger that you get given this sort of recipe. Where do you then start?
Nick - Well, as Roger said, they can be either given me on a bit of paper as a weight percent or atomic percent. If they're an atomic percent, then I need to convert them into something a bit more useful for me which is weight percent. And then I'm into the lab to go our raw elements cupboard where we have a whole variety of little pots containing pure elemental metals and then I start weighing them out, just like you would for baking in a kitchen, only with a more accurate set of scales.
 Ben - How important is it that it's accurate? If you're getting atomic percentages, then presumably, your measuring has to be incredibly accurate.
Ben - How important is it that it's accurate? If you're getting atomic percentages, then presumably, your measuring has to be incredibly accurate.
Nick - Well obviously, the more accurate we can be with the measurements I make, before I start making anything any further, the more likely we are to actually be on the composition that's been specified. So, if I muck it up at this stage, very much like a cake, it's not going to be what they ask for. At which point, someone like Roger gets probably quite annoyed with me and I have to have another go.
Ben - And you took me to see some of the equipment that you actually use. So, once you've got your measurement, you then need to melt it. And for this, you took me to see an arc melter.
Nick - So what we're looking at here is, this is an arc melter. We use this for actually melting up some of our compositions. So, we have our raw elements which we have weighed out and we place them in one of these little receptacles, so this finger-shaped recess here. Having put them all into that recess, we would put the whole system under vacuum. Oxygen goes into metals very easily particularly when they're hot. We don't really want that, so we'll pump that down a reasonably high vacuum and then before we actually start melting these, we backfill the system with argon which is an inert gas before striking an arc, which is a bit like welding. Then we use that to vary the current, the strength of the arc, to actually melt the elements together. Things mix very well as a liquid, much better than they do when they're solid. So we want to get the thing nice and hot, into a liquid form so everything mixes together and we get one uniform solid.
Ben - So, that was your arc melter which works in the same way as arc welding, you're actually using a very large electric current and that generates lots of heat. So why would you want to do it that way instead of using maybe induction - where you set up an electromagnetic field and that encourages the metal itself to get hot - or even in a more traditional oven?
Nick - I think in many ways, on some of the elements we're trying to melt, we want to get some quite high temperatures. Roger mentioned that in some of these Nickel based superalloys that he's designing, there are some refractory metal elements in there, and they have very high melting temperatures. And one thing the arc gives us the ability to get to these very high temperatures. So, we can make small amounts, and these are quite small, about the size of your little finger, to try out these materials. Now, if we wanted to make bigger quantities, we might well use a different type of furnace like an induction furnace, but we may have to melt a couple of those elements together first to get them to a lower temperature. So it's easier to melt them in these other types of furnace.
Ben - And do you have any problem whereby if you're passing a current, you are obviously also creating magnetic fields around it? Does that affect the structure that you end up with? I mean, would you get a different structure if you had used a different way of heating it?
Nick - I think that these things cool and that gives us the structures they form. I've never actually personally looked at whether we get an influence of magnetic field. I know some people do look at things in steels where they try and use magnetic fields while the material is at certain temperatures, but in a lot of the alloys that certainly I deal with, they don't have magnetic properties so I'd get away from that one, so I guess I get off lightly.
Ben - So what do you do next? You've obviously melted it. You've allowed it to cool. You have your little test sample. What's the next step?
Nick - Well one of the first things I would do is take a little slice of that material and I prepare it, and then actually go and put it into a scanning electron microscope. And from that, I can use a technique of having a very quick check on the composition to see whether I'm actually in the right ballpark. And from there, we would actually go and get it sent off to have a more accurate compositional analysis done. If I'm a long way off the composition, like I've just weighed something out wrong because I'm having a bad day, and those happen! Then I need to have another go. There's no point continuing what could be a quite lengthy process of fully characterising and doing these very long heat treatments as we saw in our introduction. I need to make sure I'm right to start with.
 Ben - So, you've said that these are samples of metal about the size of a finger. If you're then cutting off bits, what size samples are we dealing with?
Ben - So, you've said that these are samples of metal about the size of a finger. If you're then cutting off bits, what size samples are we dealing with?
Nick - Well that can vary, but typically a few millimetres in length and if you just imagine taking off the right tip of your finger, it could be something as small as that. The amounts of material we can look at can be quite small actually and this gives us a representative idea of what might happen with these materials.
Ben - You've just mentioned heat treatment and this was the next thing that you took me along to see. So, let's hear a bit of that.
Nick - So what we're looking at now is a box furnace. This happens to one that goes to 1100 degrees centigrade. They are, as they say, just boxes with heating elements down either side and then ceramic walls all around them. We would use these for putting in our samples to give them heat treatments to allow lots of diffusion to occur when they're at high temperatures, and this makes sure we can assess the crystallographic phase that they will form, the different phases. We do this for a long period of time. We've got two more furnaces next to us that a colleague of mine is currently using. There are at 800 and 1,000 degrees centigrade and they're both on for a thousand hours. So quite long heat treatments to allow us to get these materials to equilibrium and make sure we understand what their equilibrium phases are.
(Door shut)
Ben - Sounds that that could do with oiling I think, but what's the point of the heat treatment? What changes would you expect to happen in that thousand hours at 1,000 degrees C?
Nick - Well, it allows all the elements to move around within the solid. So these things are still solid at these temperatures and as I said, the elements can diffuse in that material, move around to where they'd like to be, their lowest energy positions within that solid piece of metal. As Roger mentioned early on, this could actually lead to the formation of precipitates in some case, which as he said, makes the material behave more like a composite. And we may want to form these because they could strengthen the material. So we need to form these precipitates and allow this diffusion to happen. We need to give them the ability to do that which means high temperature and then the time to actually do it. It doesn't happen very quickly. Diffusion, particularly of some of the refraction metals, is very slow.
Ben - So we're going to end up ultimately with a very different metal compared to when it was first melted. When you heat treat it for this long, the internal crystal structures are very different and so, the properties should be different.
Nick - Well that's very much the case and they can be. Sometimes, what you get out when you've just cast it isn't quite what you want and hopefully, by giving these long heat treatments, we get to the equilibrium phase that they naturally want to form. And then if we change those temperatures, we can induce these precipitates to form and therefore, yes, we can start producing a material with very different properties than would've been there in the as-cast material.
Ben - And what then do you need to do? So you've already taken an initial sample from the immediate as-cast metal and you know the structure of that. Once you've done the heat treatment, do you need to do the same sorts of tests again?
Nick - Absolutely. We need to see what that microstructure now looks like. So this is up from the atomic scale, what it actually looks like on the microscopic level. We can see quite a lot from how these precipitates form, we have to go and have a look at them because the size they adopt and the morphology, so that's the shape, can be quite important. And these things are being designed in. As Roger said, they're actually looking to design these features in, so I need to check whether we've thought about it right and actually try to manufacture it, and if we haven't. It's time to another go again.
Ben - But as well as the chemical structure of it, presumably, you need to work out if the properties of that metal are the properties that you were looking for. There must be mechanical tests that you need to do.
Nick - Well precisely. We use a range of mechanical tests. This can be from something as simple as a little diamond indenter going in to give us some idea of the hardness. Roger mentioned strength so we could assess that with the tensile test and we have machines which can do that at higher temperatures to give us some indication of whether the room temperature properties might be good, but is it also going to perform at the high temperatures that you find in a gas turbine engine? Other things Roger mentioned, he said chromium in there for corrosion protection and so, we might look at oxidation and these things get very hot as I said in my piece about the arc melter. Oxygen absorbs into the metals and then we've got oxides forming at the surface and that's bad because it's consuming your component. So we might want to test how good the oxidation resistance of these alloys are because that's something that's key to their design.
19:02 - Alzheimer's protein damages blood brain barrier
Alzheimer's protein damages blood brain barrier
A protein associated with Alzheimer's disease causes damage to the blood vessels of the brain, ultimately leading to a leaky blood brain barrier and an increased risk of neurodegenerative disease. Identifying this mechanism offers new targets for treating and even preventing disease.
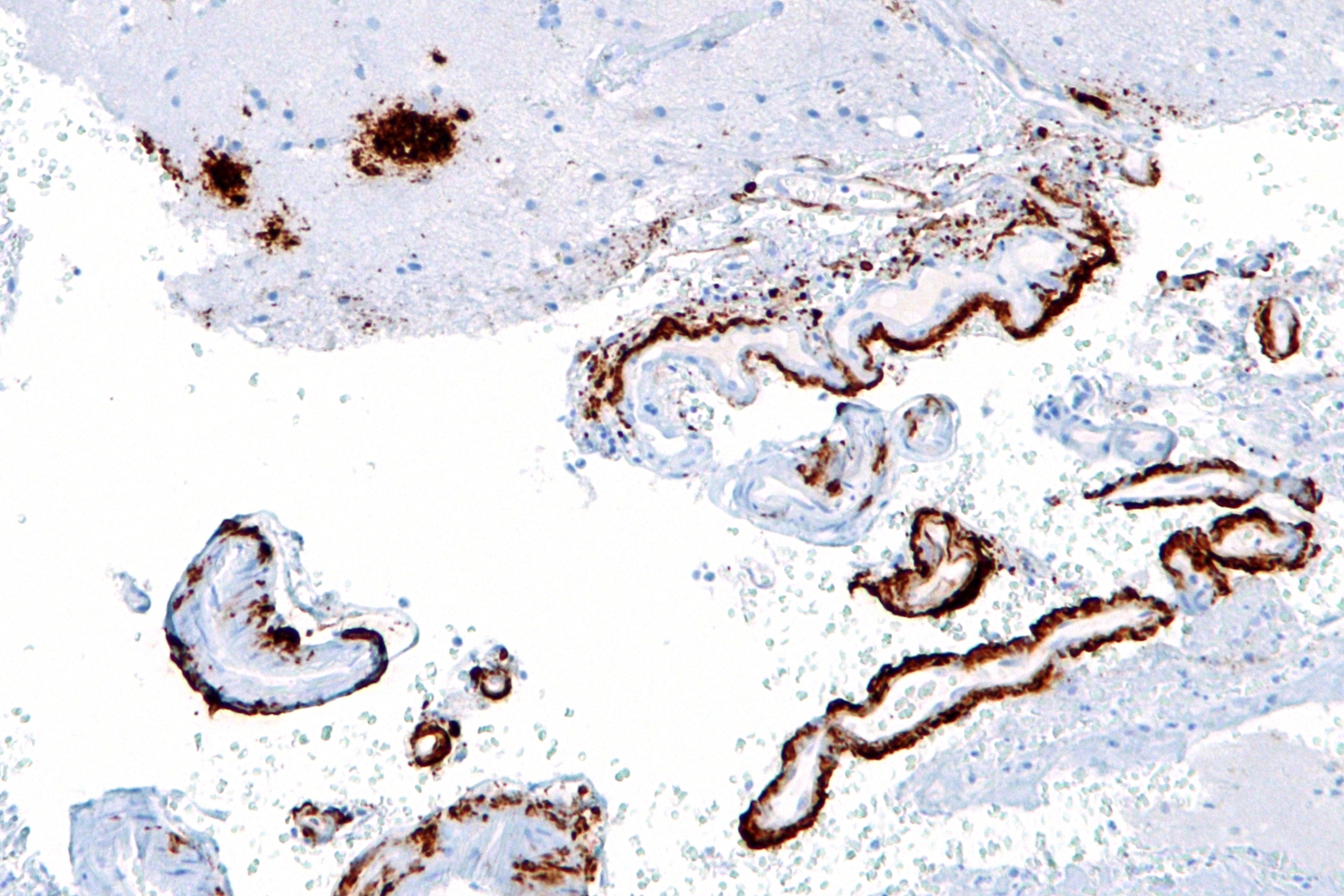 APOE4 is one of three common forms of human apolipoprotein E, and is a major genetic risk factor for Alzheimer's disease, as well as being associated with poor recovery from brain injury. Now, research published in the journal Nature shows that APOE4 activates a pro-inflammatory pathway in pericytes, cells found in the walls of brain capillaries and responsible for regulating blood flow and controlling the blood brain barrier. Activation of the pathway leads to increased uptake of toxins in the brain essentially by introducing a leak to the blood brain barrier.
APOE4 is one of three common forms of human apolipoprotein E, and is a major genetic risk factor for Alzheimer's disease, as well as being associated with poor recovery from brain injury. Now, research published in the journal Nature shows that APOE4 activates a pro-inflammatory pathway in pericytes, cells found in the walls of brain capillaries and responsible for regulating blood flow and controlling the blood brain barrier. Activation of the pathway leads to increased uptake of toxins in the brain essentially by introducing a leak to the blood brain barrier.
The blood brain barrier plays an essential role in allowing nutrients in and keeping toxins out of the brain, and dysfunction of this barrier can lead to damage and ultimately neurodegenerative disease, including the build up of beta amylase plaques that are characteristic of Alzheimer's.
Robert Bell from the University of Rochester in New York, and colleagues, targeted a particular component of this pathway, cyclophilin A. This is a protein which is inhibited by other forms of APOE, and is known to be involved in blood vessel damage elsewhere in the body, being implicated in heart disease as well as Alzheimer's. Mice carrying the ApoE4 gene express 5 times more cyclophilin A than those carrying the other versions of the ApoE gene.
Blocking cyclophilin A in mice, either genetically or using the immune suppressant drug cyclosporine, reversed the damage and dramatically cut the leakiness of brain blood vessels. This offers a good target for treating or preventing the damage that can ultimately lead to Alzheimer's, the most common cause of dementia in older adults.

21:24 - Origami, by light
Origami, by light
There are more and more applications for very small 3D structures, ranging from displays to aerials, but whilst we are very good at making minute 2 dimensional structures the third dimension is much more difficult. The Japanese developed ways of converting the 2D paper to beautiful 3D structures in the art of origami, and more recently mathematicians have formalised the process, but folding structures on the micrometer scale is rather fiddly. There have been some approaches involving liquids and using surface tension, or by adding actuators, but these are constrained or more expensive.
 However Jennie Ryu at the University of Colorado and collegues have come up with a very neat solution. They have developed a process which involves taking a sheet of polymer, stretching it in one direction, and then shining UV light onto areas of the surface which they want to produce a hinge. The polymer is designed to permanently stretch when exposed to the UV, so they end up with the upper layer longer than the under side, meaning that when they stop stretching the sheet, it will want to bend. The sheet is then stretched in other directions, and exposed to form other hinges. When a shape is cut out of the polymer sheet, it is already folded to the correct shape.
However Jennie Ryu at the University of Colorado and collegues have come up with a very neat solution. They have developed a process which involves taking a sheet of polymer, stretching it in one direction, and then shining UV light onto areas of the surface which they want to produce a hinge. The polymer is designed to permanently stretch when exposed to the UV, so they end up with the upper layer longer than the under side, meaning that when they stop stretching the sheet, it will want to bend. The sheet is then stretched in other directions, and exposed to form other hinges. When a shape is cut out of the polymer sheet, it is already folded to the correct shape.
By varying the exposure, they can change the angle the polymer folds to, and by exposing a larger length they can produce a smooth curve rather than a sharp bend.
So far they have folded up a box about 10mm across, but they have developed tools that should allow them to build structures much smaller, which could have interesting structural properties, optical properties, or even make useful scaffolds for growing tissue on.
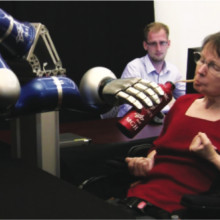
23:58 - Brain Interface Gives Paralysed Patients Freedom to Move
Brain Interface Gives Paralysed Patients Freedom to Move
with Professor John Donoghue, Brown University
Ben - Imagine not being able to just pick up a glass and have a drink, and instead, having to rely on others to help with this most simple of tasks. This week a brain interface device has allowed a paralysed stroke victim to drink for herself for the first time in 14 years by controlling a robotic arm just using her mind. Professor John Donoghue from Brown University reports on this work in the journal Nature this week and he joins us now. John, thank you ever so much for joining us.
John - Well, thank you. I'm pleased to be here.
Ben - What did you set out to show with this piece of work?
John - Previously, in 2006, we had shown in spinal cord injured patients that they were able to control a cursor on a computer and type or play a video game. But we didn't know whether people with paralysis, like these two people with stroke that we studied, would be able to control something that's as complicated as a robot arm. This was very important because in order to be able to do things for yourself, you have to be able to reach out and grab something; your drink or your morning toast. Otherwise, you're completely dependent on other people. So our test was to see whether people were able to control something this complex.
 Ben - So, what is the increase in complexity from controlling a cursor on the screen, through to controlling a robot arm? Is it the extra dimension, the fact that you're working in 3-dimensions instead of just two?
Ben - So, what is the increase in complexity from controlling a cursor on the screen, through to controlling a robot arm? Is it the extra dimension, the fact that you're working in 3-dimensions instead of just two?
John - Well, it's even more than that because if you want to grab onto something, you have to open and close your hand and you have to do it whenever you want. So, the challenge was to ask this tiny population of cells that we were picking up in the brain to not only be able to move in 3-dimensions, that is, put your hand anywhere you want in space but also to be able to open and close your hand whenever you want it.
Ben - How does the implant technology actually work?
John - We put a tiny sensor about the size of a small pill on the surface of the brain and it has 100 hair thin electrodes that go just into the surface and they pick up these very weak impulses that come out of neurons. The impulses are taken out and transformed into a command signal. So it's a pattern of brain activity that becomes a command signal to run the robot arm.
Ben - So, it's a computer science problem as well as a neuroscience problem, and a technology problem because you need to interpret what's going on in the brain in order to turn that into a reliable signal?
John - Actually, yes. This is a wonderful example of how it's necessary for engineers and clinicians, and neuroscientists, and computer scientists, all to work together in a team to solve the problem of picking up brain signals, transforming them into meaningful control signals, running them through a computer and robotocists of course, we work with robotocists to be able to take that command signal and actually make the robot arm do what the person is thinking.
Ben - I understand that one of your patients had actually had this implant for quite a long time and one of the things that people have been worried about with this sort of work is that the implants would either generate some sort of response, perhaps a local inflammatory response, or they would just fur up or they would break and stop being useful over time. This seems to be really good demonstration that actually, these things have longevity.
 John - Well this is very encouraging that an implant in the brain can last a long time, but we have to be very cautious. It's only one person so far. We've had several others that we've studied for nearly a year, but we're concerned that the materials may break down and we began to see certainly evidence of that as one of the problems.
John - Well this is very encouraging that an implant in the brain can last a long time, but we have to be very cautious. It's only one person so far. We've had several others that we've studied for nearly a year, but we're concerned that the materials may break down and we began to see certainly evidence of that as one of the problems.
Ben - In particular, your patients have had a brain stem stroke. So they are essentially isolated from their own body. Do you think that the same techniques could work with people suffering from other forms of paralysis, perhaps spinal injury or stroke in another region of the brain?
John - Basically, what a stroke like this does in the brain stem is it disconnects the wires or the fibre pathway that goes from the brain to the spinal cord and then ultimately out to the body. So, for any person who has an intact and functioning brain, this technology could potentially work. So there are millions of people with various forms of paralysis - spinal cord injury, stroke, stroke in other areas other than the brain stem, and a disease like ALS or Lou Gehrig's disease in which the connections in the spinal cord to the muscles die, and then again, break the pathway from thinking about movement to actually making the movement or disconnecting the brain and the body.
Ben - And could we use other parts of the brain as well? So, could we actually have multiple systems running at once, one to control a robotic arm and then perhaps another one to control a wheelchair for example?
John - Could be, just like we have parts of our brain to control our legs and our arms. Of course, it all is coordinated together and we can't run too many things at once or we might get into trouble. But it's very encouraging as we learn more and more about how the brain turns thoughts into actions. We know there are very large networks and many areas involved and all of those are potential candidates. We could tap into the earlier parts of the stream to get additional arm signals or we could go to the leg area for example, perhaps to control a wheelchair or even exoskeletons on the legs of a paralysed person to make their legs move so they could walk.
Ben - And it's obviously wonderful to be able to control a robotic arm, but we have seen other work where they've been using a special type of electronic stimulation to actually make muscles move. Could ultimately this be used to bridge the stroke site, bridge the gap and reconnect muscles to what the brain is thinking? Could we get people controlling their own limbs again?
John - Yes, that's something that we're working on. We're very interested in reanimating the muscles and this is work of Hunter Peckham and Bob Kirsch at Case Western where they've implanted stimulators in the body and it leads to wires that go to the muscles or to the nerves. And when that stimulator goes off, the arm contracts. There are about 600 people that they've implanted that are standing up or moving their arm to some extent as a consequence of this wonderful technology. So we're working with them to try to say, instead of controlling it with an outside switch - so the current people will use a button that someone can push or a switch on their shoulder so if they can wiggle their shoulder, they can make their hand open and close. But now, imagine we could connect Braingate, this brain sensor, take the brain signals and wire them back into this stimulator which then goes to the muscle. So we should have a physical repair for a broken biological system.

30:47 - Bacteria fed by dinosaurs found 'barely alive'
Bacteria fed by dinosaurs found 'barely alive'
A community of microbes has been found living in 86 million year old deep sea clay, although they're not exactly thriving - they seem to be surviving at the minimum energy requirement needed to qualify as "alive".
 Along with colleagues in Germany and the USA, Hans Røy from Aarhus University in Denmark collected long cylinders of mud known as sediment cores from deep under the Pacific Ocean sea bed during a cruise of the current system known as the North Pacific Gyre. The sea bed beneath the Gyre is particularly interesting, as oxygen can penetrate many metres below the surface, unlike other ocean areas where oxygen penetrates just a few centimetres. This makes them a good place to look for unusual forms of microbial life, which could in turn help our search for life outside our planet.
Along with colleagues in Germany and the USA, Hans Røy from Aarhus University in Denmark collected long cylinders of mud known as sediment cores from deep under the Pacific Ocean sea bed during a cruise of the current system known as the North Pacific Gyre. The sea bed beneath the Gyre is particularly interesting, as oxygen can penetrate many metres below the surface, unlike other ocean areas where oxygen penetrates just a few centimetres. This makes them a good place to look for unusual forms of microbial life, which could in turn help our search for life outside our planet.
Writing in the journal Science, the team measured oxygen and carbon concentrations at different depths and, by making some assumptions about factors like the degradability of organic matter, were able to calculate how oxygen consumption changed with depth.
They then measured the numbers of microbial cells, which falls off rapidly with increasing depth. At the surface they found 108 (100,000,000) cells per cubic centimetre, but by 20m below the surface, in sediments laid down over 66 million years ago, they were finding just 1000 cells per cubic centimetre. Further down the cells were so sparsely distributed as to be uncountable, but evidence of their presence was still seen in oxygen consumption. One interesting finding was that the drop in density of microorganisms lagged behind the drop in oxygen concentration - suggesting a gradual change in the way oxygen is consumed.
The oxygen consumption rate did eventually reach a stable minimum, with the deepest microorganisms consuming 3 orders of magnitude less oxygen than similar cells in culture. This incredibly low respiration rate suggests these cells may be existing in a long-term stationary state, subsisting on the bare minimum to survive and unlikely to be growing or dividing.
Our present understanding of microorganism respiration is built mainly on our experience of culturing fast-growing microbes at the surface, so it's no surprise that what we discover deep under the sea bed doesn't match our expectations - even less so when you consider that this layer of sediment has seen no new food since the dinosaurs!

33:34 - The Spread of Superbugs, Cellular causes of Tinnitus and Precise Plant Pollination
The Spread of Superbugs, Cellular causes of Tinnitus and Precise Plant Pollination
with Ross Fitzgerald, University of Edinburgh; Martine Hamman, University of Leicester; David Kring, Universities Space Research Association; Mark Johnson, Brown University
The Spread of Superbugs
Large city hospitals acts as breeding grounds for the highly resistant bacterium MRSA and facilitate its spread to more regional locations.
A study, led by Ross Fitzgerald from the University of Edinburgh, sequenced  the DNA of 87 samples of Methicillin-resistant staphylococcus aureus, or MRSA, from patients in over 15 hospitals across the UK.
the DNA of 87 samples of Methicillin-resistant staphylococcus aureus, or MRSA, from patients in over 15 hospitals across the UK.
The team deciphered the genetic make-up of the samples to trace the origins of infections found in more local settings and found that hospitals in big cities act as a hub for transmission of MRSA between patients who can then carry and transmit the bacteria to their local setting...
Ross - You don't have to actually have a disease to be infected with MRSA, but they can carry the MRSA with them to those regional hospitals. We might be able to identify patients who become colonised when they're in these city hospitals. If we can screen and identify patients who are MRSA carriers, we could then treat them using topical antibiotics to decolonise or remove those MRSAs so they're not then spreading them back to those regional centres.
---Cellular Causes of Tinnitus
The cellular mechanisms underlying the onset of tinnitus have been discovered by scientists at the University of Leicester, publishing in the journal
Hearing Research.
Working with nerve cells from the brain's dorsal cochlear nucleus, where signals are relayed from the ear to be decoded by the brain, Martine Hamann found that on exposure to loud sounds the erratic signalling in these cells, known to cause the ringing or buzzing associated with the condition, are caused by malfunctions in potassium channels within the nerve that normally regulate the its electrical activity.
Martina - You can have a variety of tinnitus. It can be on one ear or on two ears. It can be different frequencies, or the perception can actually be altered differently it can occur at different times of the day So it's very difficult to pinpoint the mechanism, considering it is quite diverse. So, finding something that is actually so specific can be quite promising in finding a specific target.
The work leads the way for potential drugs to restore the functioning of these channels, enabling the nerves to return to their normal state...
---Bombarding the Earth and Moon with asteroids
 The Earth and moon were bombarded with asteroids nearly 4 billion years ago, re-shaping their surfaces in the process.
The Earth and moon were bombarded with asteroids nearly 4 billion years ago, re-shaping their surfaces in the process.
It's known that our planet and the moon were bombarded with objects thought to be either asteroids, comets or protoplanets approximately 3.9 billion years ago.
Now, writing in
Science, David Kring from the Universities Space Research Association in Texas, analysed samples of moon rock from the Apollo 16 mission, which contained 30 surviving fragments of the colliding objects and identified the culprit to be asteroids - providing greater insight into our early solar system...
David - This is a period of bombardment that immediately precedes the earliest isotopic evidence of life on the Earth and so, we're constantly trying to address, could this bombardment have something to do with the origin and early evolution of life To the extent that we can tease apart more details of the objects hitting, the pace at which they hit the Earth and the moon, and what it is they delivered to the Earth and the moon will better help us evaluate those questions.
---Precise Pollination
The precise way in which plants pollinate has been uncovered by scientists at Brown University in the US.
During pollination, hundreds of pollen-containing sperm stick to the stigma of plants such as Arabidopsis and create tubes through which they deliver sperm to the plants' ovules. Precisely two sperm per ovule, as any extra can result in poor seed development. But the way this is controlled was previously unknown.
Working with models of Arabidopsis in the lab, Mark Johnson and colleagues found that the plants generate a signal when sperm have successfully passed down the tubes to fuse with the egg inside the ovule to prevent any further pollen from landing.
Mark - Plant fertilisation process waits until the moment when fertilisation has been secured to preven t multiple pollen tubes from coming. So, in a case where defective sperm are delivered, that ovule continues to attract more pollen tubes. So, if we can understand the molecular mechanisms that are the basis of these systems, we can then improve the systems or perhaps we can engineer them to be more robust and be able to resist adverse environmental conditions.
And that work was published in the journal
Current Biology.

38:31 - Indoor Avalanches - Planet Earth Online
Indoor Avalanches - Planet Earth Online
with Nathalie Vriend at the University of Cambridge
With summer on its way (apparently) scientists who study the physics of avalanches have had to develop ingenious ways of studying what happens when snow cascades down slopes at up to 100 miles per hour. And they don't have to go on the piste to do it.
Nathalie Vriend at the University of Cambridge's Centre for Mathematical Sciences simulates avalanches by using what looks like a big metal phone box leaning on its side. There are no glass walls - just a rectangular frame and a narrow conveyor belt on a slope.
leaning on its side. There are no glass walls - just a rectangular frame and a narrow conveyor belt on a slope.
Planet Earth podcast presenter Sue Nelson went to see Nathalie's experiment for herself and discovered something familiar sliding down that slope - and it wasn't snow... it was sand...
Nathalie - Both snow and sand consist of small particles and these particles, when they start to move, they interact and collide with each other. Because of that the physical processes that occur on this small scale are actually very similar. So, we're doing our experiments with sand instead of snow.
Sue - Can you demonstrate to me what you actually do? Can we turn this huge contraption, this conveyor belt of sand, on?
Nathalie - Yes of course, I am happy to... I'm opening a valve that opens the centre flow. The sand is being let in at the top of the incline. The distance where the sand can avalanche is about three metres long, so we're bringing the sand to the top and it starts to avalanche down.
Sue - I can see now some of the sand coming out of a little tube at the top of the slope which is just a bit higher than me, so it's a couple of metres high.
Nathalie - At this moment the sand is not flowing fast. What we can do is actually open the valve a little bit higher so that the sand starts to come out in bigger quantities.
[Sound of falling sand]
Sue - Oh gosh! That's really interesting; it looks like your slope of sand - the central bit - now looks like a flowing molten river of sand.
Nathalie - Indeed, indeed. And if you look very carefully you can see that the middle part is flowing very fast, but there are bits on the side, like dykes-
Sue - Oh yes, they're turning round and not moving so much.
Nathalie - Exactly, and there are actually some grains that are static or not moving at all.
Sue - Is that what happens when snow goes down a slope?
Nathalie - Yes indeed. When a mass comes down a mountain it goes the fastest in the middle and the velocity goes further to zero towards the sides. Therefore if you look very carefully at deposits on the mountain you can always see that the snow carved its way through a deposit and left debris on the side.
Sue - How did you choose the angle of your slope?
Nathalie - The angle that avalanches usually occur is between 30 and 45 degrees. That's actually pretty steep, it's much steeper than you would ever drive on. Let me change the flow rate right now because then you can see some other features as well.
Sue - Oh gosh, that's interesting. Instead of a free flowing river it was almost like a drop of treacle.
Nathalie - So in this case, because we reduced the flow rate there is not enough sand coming through to form this continuous river. Because of that the sand accumulates to the top of the incline and just starts to avalanche, the slope fills when there is a certain amount of sand available. So you get these intermittent avalanches and they almost look like tongues going down the incline.
Sue - What do you hope to learn by studying this flow, this avalanche of sand? Because a certain amount is known already?
Nathalie - It is really difficult to understand exactly what's going on because as you can imagine, if you have a flow of water down a incline, researchers know pretty well how to investigate water down an incline. But if you have a solid material the physical laws aren't very well understood. In this case you have this odd mixture of different phases; as you look in the middle of river of sand it looks like a flowing stream and it goes very fast and there is a lot of motion going on. But if you look at the side of the river it is static and that's really hard to model. So the bigger picture is that we want to understand where the avalanches are going, how far they get, how forceful and how big they are and what kind of pressures occur in them. The reason is that we want to understand where we can build buildings and roads and where it is safe for people to live.
In the past people relied on historical records but they may not be very accurate anymore because the climate is changing so, snow fall and temperatures are changing as well. Also, not every point in the world has historical records. We want to be able to model snow avalanches from a physical point of view to actually be able to apply it to every valley and every mountainside in the world.

43:52 - New Metals in Jet Engines
New Metals in Jet Engines
with David Rugg, Rolls Royce
Ben - In pursuit of better efficiency, jet engine manufacturers like Rolls Royce are constantly searching for materials that will enable their engines to run at higher temperatures, more quietly and give out more power. I met Dave Rugg, Materials Specialist for Rolls Royce under an enormous Trent jet engine to find out what happens once a new material is made.
Dave - The first thing that we have to consider is what we're actually going to use the material for. The key things there really are the load regimes that it's going to be subject to, so is it going to be high temperatures, is it going to be high fatigue loading? And also, how we would actually go about manufacturing it in a cost-effective way or indeed if it's manufacturable at all at real length scales.
Ben - So, what sorts of properties are you looking for? Say, if you need to make a turbine blade, what do you need that metal to do?
 Dave - There's a very wide range of loads that the materials are subject to. For turbine blades, obviously, one of the key things is how the material behaves at very high temperatures; so there we would be looking at creep behaviour, which is basically the material stretching under load at high temperatures. But also, there's a lot of other loading that a material is subject to, so that can be things like fatigue loading because of the centrifugal induced stress, but also gas loads because the blade is having to do a lot of work or extract a lot of work from the airflow.
Dave - There's a very wide range of loads that the materials are subject to. For turbine blades, obviously, one of the key things is how the material behaves at very high temperatures; so there we would be looking at creep behaviour, which is basically the material stretching under load at high temperatures. But also, there's a lot of other loading that a material is subject to, so that can be things like fatigue loading because of the centrifugal induced stress, but also gas loads because the blade is having to do a lot of work or extract a lot of work from the airflow.
Ben - Now, we are next to a Rolls Royce Trent engine. How many different materials go in to making something like that?
Dave - We've got hundreds of different materials and product forms that go into an engine. In terms of the main alloys, we're probably in the order of low tens, but lots of different product forms all have different mechanical properties associated with them.
Ben - And once you've got a material, it presumably needs forming into the right shape and testing in a way entirely different to just testing the material on its own. Does that introduce new hurdles, new problems?
Dave - Absolutely and it's a key area, really the most important part of the materials organisation within the company. The material behaviour in component form can actually be impossible to predict from specimen behaviour. So what we have to try to do in the material introduction process is to try and minimise our costs associated with specimen testing but give ourselves a high level of confidence when we do component tests that are going to be successful.
Ben - Presumably also, all of the ways of tooling, all of the actual manufacture techniques also need to fit with the properties of the material?
Dave - Absolutely, and you find with quite a lot of materials, we have to actually use tailored manufacturing routes and there can be as much work associated with coming up with optimised manufacturing methods as there can be with the alloy design itself.
Ben - So, once you actually have something, it's gone through all of your tests, what's the process then to make sure it gets into the next generation of engine?
Dave - The key thing there is the engine development project and the chief engineer there will undoubtedly have a very big task on his hands, or her hands, in terms of meeting the efficiency and performance targets. Quite often, materials are integral to being able to produce that improvement. So, the chief engineer might be looking for design improvements and in order to do that, they might need materials that operate at high temperatures, high stresses. But if there's a window there in the engine, where that material will give specific gain then the chief engineer potentially be interested in picking up a new material. What then happens is that the engine development programme would actually use components made of the new material and through the development programme, they'll be subject to loads and temperatures that are well outside anything the material would normally see in a commercial operation.
Ben - And how much of a gamble is there on the fact that you'll get the materials that you want? These things are years, even decades in development. So when you've set a challenge for the universities to provide a material that meets certain goals, can you then set about designing an engine, trusting that you'll get it?
 Dave - That's an extremely good and rather difficult question and to answer it, I would say it's probably easier to look backwards in time, rather than forwards. If you'd asked me 20 years ago if we would have new titanium alloys that we'd be using in engines I'd have said it was going to be very difficult because they're already very well evolved and pretty much optimised. But of course, history would've proven me wrong. We've got a lot of advantages now in terms of modelling and understanding the physical mechanisms behind material behaviour that are actually giving us new insights into the way that materials work, and therefore, much more scope for producing really tailored alloys by understanding the physics rather than just doing the evolution empirically.
Dave - That's an extremely good and rather difficult question and to answer it, I would say it's probably easier to look backwards in time, rather than forwards. If you'd asked me 20 years ago if we would have new titanium alloys that we'd be using in engines I'd have said it was going to be very difficult because they're already very well evolved and pretty much optimised. But of course, history would've proven me wrong. We've got a lot of advantages now in terms of modelling and understanding the physical mechanisms behind material behaviour that are actually giving us new insights into the way that materials work, and therefore, much more scope for producing really tailored alloys by understanding the physics rather than just doing the evolution empirically.
Ben - Do you ever get a huge breakthrough that you didn't expect and have to go back to the drawing board?
Dave - Absolutely and I think some of the relatively modern materials in terms of intermetallics as an example, where the materials are somewhere between a metallic and a ceramic in terms of their behaviour, that's been quite a big surprise for the industry over about the last 20 years. Although, trying to make those work in a real engineering sense is quite difficult because you pay for the benefit one way or another. So, in the case of intermetallics, that's by way of them being rather brittle which has its own challenges then in both manufacture and in use.
Ben - How do you go about saying to universities, "This is what we want and this is when we want it"?
Dave - Well, there are a few key things that drop out to that and some of them aren't particularly intuitive. One of the prime examples would be that in terms of understanding current product, there's a huge amount of research going on at the moment with well-established alloys that give us a much better insight into the actual physics of how the materials work and then that gives you the opportunity to actually develop new materials from that. But we do also have a variety of different means by which we can feed that information and those targets into the universities so that we set challenges, basically, to the academics and say, "In 10 years time, we would really like an alloy that's got an extra 10°C temperature capability" which may not sound like much but it's an incredible differential in terms of the product or an extra x% of fatigue strength.
Ben - So it's not just the metallurgists and the materials scientists on this, but the physicists as well that you are getting on side?
Dave - Absolutely. There is some key work going on there in terms of both atomistic modelling, molecular dynamics, crystal plasticity modelling, that's actually informing the alloy development process. There are some materials being developed now, and in fact have actually seen service already, that essentially have been developed on the computer rather than in the foundry, which just quite remarkable.
Ben - And we've been talking as if the jet engine is the final product of a long chain, but if you're developing new and interesting physics, you're increasing the understanding of the materials, that must also feedback into the academic community. So not only do we get new, better, more efficient, more powerful engines, but we also get a better understanding of the world around us.
Dave - Absolutely. Materials are a great system to work on. Metallic materials, as an example, actually behave like composite materials and you can learn a great deal about the way that physics actually works by studying real things in real environments.

Are rare earth elements used in alloys?
Nick - Rare earth elements are just a certain part of the periodic table. It's just a group of elements and some of them, such as Yttrium and Lanthanum, do find some small percentage uses in some modern alloys but as you say, there's no large scale use of them, just due to their high cost.
Ben - Roger, when you're designing new alloys or when you're modelling them, do you work around the cost? Are there ways that you can replace things that rare earth elements would otherwise do?
Roger - Yes, it's certainly true that we consider the cost because certain elements, like rhenium for example in turbine blade alloys, are now very expensive, several thousand pounds per kilogram. When one is interested in building a jet engine with perhaps 100 blades per row then the cost can become significant quite quickly. So certainly, there are calculations done to trade off the relative benefits of elements like rhenium and tungsten, and molybdenum and other refractory metals against their disadvantages which are then costs and also their scarcity.

Do atoms diffuse through solid metal?
Nick - We're absolutely talking about it still being a solid and the atoms do move through them and there are processes that occur. That's why we say they mix better in a liquid state because things can move more easily than when they're forcing their way through a solid lattice. But that's why we have to give them time and temperature. The temperature helps in getting them going, makes things move a little easier and the time is just to allow them to move the distances, but yes, they are moving through the solid state.
Ben - And Roger, when you're designing a new metal, again obviously, you have to take into account the sort of heat treatments it's likely to go through. How do you model that into it as well?
Roger - Well again, there are thermodynamic databases available for estimation of the melting temperatures and the heat treatment temperature windows that are available. Those of course are based on thermodynamic potentials which have been estimated, and put into computer based models. Most of the time, those models are accurate and can be used to estimate whether any given alloy has an appropriate heat treatment window to allow that to happen without the melting occurring. Interestingly for the turbine blade alloys, they all have to be heat treated at temperatures really quite close to the melting temperature, but obviously, lower than it so that the turbine blade won't start to melt incipiently during the heat treatment process.

What materials does a car consist of?
Nick - We've talked quite a lot tonight about some quite high performance alloys. We've talked about nickel-based superalloys, and these are quite expensive materials. Cars want to be a bit cheaper, we want to be able to afford them from our own pockets so we quite often use more common alloys such as the steels, so iron based alloys, or aluminium alloys which are a little bit cheaper, due to their cheaper raw materials.
55:12 - Can you create a magnet so powerful it crushes the object it attracts?
Can you create a magnet so powerful it crushes the object it attracts?
We posed this question to Joe Brown who works at Oxford Instruments designing superconducting magnets for use in research labs and the clinic.... Some of the work I do involves calculating the forces of attraction between the magnets and any magnetic materials in the environment. This means that the question, can we make a magnet so strong that it will crush the object it attracts is of particular interest to me. Magnetic materials in the general sense are those known as ferromagnets. The most common example of which is iron found in the steel that makes up many objects in our everyday lives. When positioning our superconducting magnets in laboratories which often contain large quantities of steel in the building framework, laboratory furniture, and experimental equipment, we have to perform calculation of forces between the magnet and this local environment. This calculation shows that for any magnet which we can conceive of today, the simple answer to the question is no. The magnetic forces are insufficient to crush any object it attracts. This is not to say that the forces involved are small and it is quite possible to have situations where a loose metal object such as a steel waste bin can be accidentally attracted and firmly stuck to the outside of a magnet system. In such circumstance, the bin will come off worse in the encounter, ending up severely bent. To help prevent such accidents, large magnets such as those used in MRI scanners are magnetically shielded using special configurations of the windings making up a magnet, reducing the magnetic field around it. Special consideration is also made to the position and surroundings of the magnet and you'll not find objects like steel waste bins within an MRI suite.










Comments
Add a comment