How handedness spans the scientific world, from the smallest particles in the Universe to the drugs that cure disease and even the way you hold a pen, goes under the microscope this week as we explore the realms of asymmetry. Plus, in the news, the world's first synthetic chromosome, the goo that stops bones breaking, is there a giant planet lurking beyond Pluto, aircraft black boxes and anti-aphrodisiacs...
In this episode
01:13 - Creating a chromosome
Creating a chromosome
with Jef Boeke from New York University Langone Medical Centre
 This week, scientists have achieved what has been hailed as the biological equivalents of climbing Mt. Everest. They've created a synthetic, fully functional copy of a yeast chromosome. Jef Boeke from New York University Langone Medical Centre is one of the scientists who's leading the project. He explained the project to Chris Smith...
This week, scientists have achieved what has been hailed as the biological equivalents of climbing Mt. Everest. They've created a synthetic, fully functional copy of a yeast chromosome. Jef Boeke from New York University Langone Medical Centre is one of the scientists who's leading the project. He explained the project to Chris Smith...
Jef - We've made a brand new chromosome, starting from a design that we put together on a computer. The original chromosome has about 316,000 base pairs, which you could think of as letters. Starting with that sequence of letters, we carved out sections that we thought would probably the yeast could do without. We inserted some of these and we changed some of the letters in ways that were visible to us. Then we engaged a class of students in the Build-a-Genome Class at Johns Hopkins University who strung together these letters into words, the words into paragraphs, and eventually, we built pages of letters so to speak. And then chapter by chapter, we introduced one chapter of approximately 25,000 letters at a time and replaced the chromosome that was present in the normal yeast with the synthetic counterpart.
Chris - So, you did this in an incremental fashion. You didn't replace the whole chromosome at once. You did little bits of it at a time.
Jef - Precisely.
Chris - Why was it necessary to do little bits at a time? Why couldn't you just make a new one and put it in?
Jef - Well, we are able to do that. But it was risky the first time to do that because by making so many changes to the sequence that we had, we did not know ahead of time what the outcome would be. This was a way to make sure that there were no flaws in our overall plan.
Chris - So, the yeast that you've made, it works, it's indistinguishable when you just look at it in the way in which it grows, running this artificial computer design chromosome compared with wild-type yeast - yeast you would find brewing beer which is the yeast you started with, isn't it?
Jef - That's correct. That's exactly right.
Chris - You picked on yeast chromosome 3, or at least that what you said you've done in your Science paper; so why did you choose that one?
Jef - Well, you might've thought we would start with the smallest chromosome, but chromosome 3 is actually the third smallest chromosome. The reason is because it's actually a sentimental favourite of people who work on yeast and that's because like people, yeast comes in 2 sexes. They're not called male and female, they're called A and alpha. And just like people, yeast can mate and give rise to progenies and the master regulator gene for this process lives on chromosome 3. That's why it was also the very first chromosome whose DNA sequence was determined.
Chris - So, this gives you an insight into whether yeast effectively make male and female forms and if you've made an artificial form of yeast and it still makes male and female forms, you know your chromosome is working, right?
Jef - It's one of the many ways we know, yes. There are many genes on this chromosome. Over a hundred and basically, they all have to be working to some extent in order for the yeast to grow normally as far as we can tell.
Chris - What are the implications of what you've done, of being able to produce and prove that you can successfully produce a chromosome and replace the entire chromosome in a living organism in this way?
Jef - Well, two things. Number one, we can obviously scale up. We can make larger  chromosomes and potentially, we could build chromosomes that could be introduced for example into plants and animals down the road, much smaller versions than the native chromosomes because the yeast chromosomes are quite a bit smaller. But secondly, we've installed into this chromosome a kind of programme that will allow us to - by slipping a switch, a genetic switch - allow us to trigger the formation of thousands of derivative chromosomes, all different from each other. I likened it to a system that automatically shuffles a deck of cards. Only in this case, the deck contains 100 genes. So, we can shuffle the chromosome and ultimately, we'll be able to shuffle the entire genome of this yeast. By doing that, we think we can make greatly improved versions that can be very useful for practical purposes, not just making alcohol which we know that yeast does very well, but many other products like medicines and vaccines, and specialty chemicals.
chromosomes and potentially, we could build chromosomes that could be introduced for example into plants and animals down the road, much smaller versions than the native chromosomes because the yeast chromosomes are quite a bit smaller. But secondly, we've installed into this chromosome a kind of programme that will allow us to - by slipping a switch, a genetic switch - allow us to trigger the formation of thousands of derivative chromosomes, all different from each other. I likened it to a system that automatically shuffles a deck of cards. Only in this case, the deck contains 100 genes. So, we can shuffle the chromosome and ultimately, we'll be able to shuffle the entire genome of this yeast. By doing that, we think we can make greatly improved versions that can be very useful for practical purposes, not just making alcohol which we know that yeast does very well, but many other products like medicines and vaccines, and specialty chemicals.
Chris - Jef Boeke from the NYU Langone Medical Centre. He published that work this week in the journal Science.

06:11 - Black Box Recorders
Black Box Recorders
Now that Malaysian Airways Flight MH370 has officially been acknowledged to have ended in the southern Indian Ocean with all lives lost, attentions have turned to recovering wreckage and piecing together what events might have lead to its crash. It's the aircrafts black box recorder which might hold the most clues. Here's your Quick Fire Science with Kate Lamble and Hannah Critchlow
- An aeroplane's black box is in most cases really two boxes painted a very visible bright orange
- One box contains the flight data recorder which stores 25 hours of information on the how the plane's operating systems are functioning - everything from altitude and airspeed to the movement of individual flaps on the wings.
- The other box contains the cockpit voice recorder which holds the last two hours of cockpit conversation. This continually records over itself while the flight is in the air.
- While investigators are interested in what the crew were doing just before the crash the voice recorder can also be used to hear background noise like emergency warnings in the cockpit and the engine noise.
- Black Boxes are required by international standards to withstand fires over 1000 degrees celcius, being immersed in salt water, deep sea pressure, and an impact velocity of 310 miles per hour.
- To help achieve this memory boards are covered with thick insulation and a steel or titanium shell and are stored near the tail section of the plane where they are less likely to be crushed. Some are designed to self-eject from a plane at the moment of impact.
- But all this insulation makes the box too heavy to float. Each recorder is therefore equipped with an underwater locator beacon which automatically begins to send out loud sonic pings when it is submerged.
- The beacon signal can be picked up a mile away but as the current search are covers over 35,000 square miles investigators are concerned that the black box will not be found before the recorders 30 day battery life expires.
- Another problem is that although sound travels extremely well underwater, in the deep ocean, layers of water at different temperatures and pressures known as thermoclines refract and bend the sound of the pings so that it's more difficult to locate.
- To help the search the US navy have brought in a 'towed pinger locator'. This highly sensitive listening device is lowered underwater to overcome this refraction and is capable of hearing black box pings at depth of up to 20,000 feet
- However, even if the black box from MH370 is not found within this time it does not mean it will never be recovered. The black box from Air France Flight 447 was found two years after the crash after autonomous unmanned submarines were used to scan the ocean floor with sonar.

09:27 - New Dwarf Planet discovered?
New Dwarf Planet discovered?
Scott - Hello.
Chris - So, what is this all about?
Scott - Planetary scientists are getting very excited this week because a paper in the  journal Nature has announced the discovery of a dwarf planet candidate called- well, it is a very unexciting name - it's 2012 VP113. I'm told that the team who discover it refer to it as Biden after Joe Biden, the vice president or VP.
journal Nature has announced the discovery of a dwarf planet candidate called- well, it is a very unexciting name - it's 2012 VP113. I'm told that the team who discover it refer to it as Biden after Joe Biden, the vice president or VP.
Chris - What actually is it and how did they find it?
Scott - Well, they found it using a classic technique for looking for planets which is, you point a telescope at the sky and you watch for awhile and look for anything that moves. So, in fact, they use an instrument called the dark energy camera on a telescope in Chile and they took some observations over, I think one night in 2012. Hence, the 2012 in the name and spotted this thing moving over a period of a few hours. In fact, when they went back and looked at some old images of the sky, it had been seen in some images from 2011. But because they haven't seen it moving, they didn't know it was a potential ball of planet at that point.
Chris - Are they seeing a dark patch moving or are they seeing a light patch moving?
Scott - You see a light patch because it reflects the light from the sun.
Chris - How big is it?
Scott - I'm not entirely sure on this one. It's definitely smaller than the planet Pluto. As you may know in 2006, the planet Pluto was declassified as an actual planet. It's now called a dwarf planet. The reason for this is that Pluto doesn't have enough mass to have pulled in all the objects from its orbit. It hasn't done what's called clearing its orbit. It's simply too small for that. So, there are other objects called plutoids I believe which are found in the same orbit as Pluto. Now, the subject is I think about a 10th of the diameter of Pluto possibly, a 10th of the size. And so, it's also a lot further out than the planet Pluto.
Chris - Given that it is that far out, what are the implications then for the definition of our Solar System and also, for the model of how we think our Solar System formed and the way the planets that we didn't think we had before we've discovered this one and came to be?
Scott - Well, the definition of a Solar System is a bit of a contentious one which is great if you're an astronomer who likes to argue perhaps. There's a few different ways you could think about it. You could think that the edge of our Solar System is just defined by the furthest planet or the furthest dwarf planet we can see. But as we've seen this week, that's a definition that's constantly changing. You could also think of the edge of the Solar System as being defined by something called the Oort cloud which is a hypothetical cloud of objects - icy, rocky bodies lift over from when the Solar System formed. But we don't actually know if the Oort cloud is out there. This object plus another one which was discovered a few years ago called Sedna are possible signs that we've started to spot the edge of this Oort cloud around the edge of our Solar System.
Chris - So, why are scientists saying there could also be a monster, 10 times Earth-sized beasts lurking out there too.
Scott - This is because the eccentricity, so the shape of the orbits of these planets, they are very long and very oval. We expect planets to be roughly circular in their orbits around the sun. and so, the models we have for how the Solar System form aren't really explaining what these bodies are doing at here and why that it's got such an eccentric orbit, such an oval-shaped orbit. So, one of the theories for why that could happen is that they could've been scattered out of an orbit further inside the Solar System by a monster planet. That's one of the theories. I don't want you to think it's the only one. The more likely one is possibly that when the sun was forming, it was part of what's called a stellar nursery, many young stars all close to each other and that one of these stars somehow managed to scatter these objects out of the orbits around the sun.
Chris - Scott, thank you very much. Scott Thomas who's a Space Scientist from the University of Cambridge.
13:28 - Bones contain a goo-based shock-absorber
Bones contain a goo-based shock-absorber
with Melinda Duer, Cambridge University
Earlier this week scientists in Cambridge revealed that the minerals that make up our 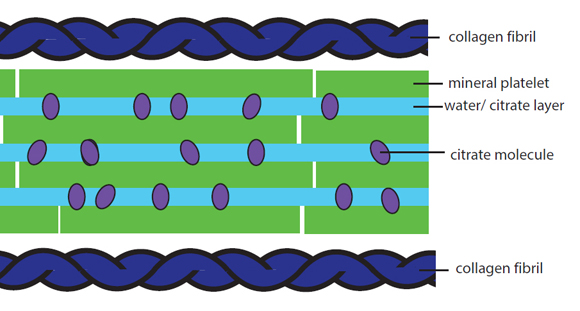 bones actually largely consist of a so-called 'goo', which works as a shock absorber.
bones actually largely consist of a so-called 'goo', which works as a shock absorber.
Melinda Duer, from Cambridge University and led the study, explained the findings to Harriet Johnson...
Harriet - Hi, Melinda. So, what is this goo and why is it so important?
Melinda - Okay, so what we knew about bone mineral before we started this study is that the mineral consists of these really tiny, tiny nanoscopic plates of calcium phosphate. They stick together in stacks between these ropes of collagen protein in our bones. So, that's what we knew. What we didn't know was what held those stacks together. Now, we had an inkling it had to be something plastic or flexible, because it wasn't frankly a stack of little plates like that. It's just brittle. So, that was what we knew. It took us 4 years to work out what was between those plates. But when we finally worked it out, it made complete sense because what's there is "goo" and it's a goo of citrate and water, and some of the calcium phosphate mineral ions as well in there.
Harriet - So, why did it take so long to find out this structure?
Melinda - Good question and it wasn't just because we were being lazy. So, if you're looking at what we call an ordered structure, crystallised structure are a regular ray of atoms and molecules. There's quite a lot of techniques that can help you find out about that. They're well understood. When you're dealing with something where the atoms and molecules are disordered, they're just in some kind of almost random arrangement. It's really tricky to find anything that can help you find any clues about that. So, what we had to do is we use lots and lots of different techniques and we pieced together the information. A little bit of evidence from here and there. Our team at the Advanced Imaging Centre in Cambridge came up with some things, our nuclear magnetic resonance spectroscopy came up with others, and then our fantastic theoretical physicists at UCL Chris Pickard was able to help us put those ideas together into a structural model. We can then do some calculations on, see what kind of experimental event should be coming out of that model and we go back and check, and look for that. Now, we had to go through an awful lot of models before we started to say something that might actually be reality.
Harriet - So, a big project. So, this is believed to be a root cause of osteoporosis and other brittle bone problems. How is that and how can we use this information to improve these conditions?
Melinda - On the macroscopic level, there's going to be a lot of reasons why bones are brittle, but if you drill down to the molecular level, there's a more restricted set of reasons why those molecules either have the wrong structure or the wrong interactions with each other. And so, that's what we're looking at here. Essentially, the citrate and water goo allows the mineral plates to slide over each other. So, as you jump up and down your bones, your bones flex; those mineral plates can actually move and they're not going to be damaged by that impact. But if that goo is not there, then what you got left with are these crystals, these flat plates stuck together and more of this big brittle lump and they're going to be fracturing. They're just a very brittle mass. So, the question is, why does this citrate get in there and why does it sometimes not? Our hypothesis - and this is just what it is at the moment - is it comes down to how this mineral is formed. That's formed - as I've said before - in between these ropes of collagen matrix and those spaces where it forms are really, really small. The citrate delivers calcium to that space to make the calcium phosphate and then it gets trapped. It's a very small space. The minerals are forming. The citrate can't get out. What we think happens when you have osteoporosis or brittle bone disease is that the space the mineral is forming is just much larger. So now, you know forming the mineral inside a calf and of course, the citrate can then get away. It doesn't have to be incorporated. So you end up with large blocks of mineral without citrate and hence, brittle.
Harriet - So, this essential goo is leaking out of the bones and then causing these problems. Thanks to Melinda Duer from Cambridge University. She published that work in PNAS this week.

17:10 - Anti-aphrodisiacs, for flies
Anti-aphrodisiacs, for flies
with Joanne Yew, National University of Singapore
Pheromones are chemicals that can alter behaviours like mating. Now researchers have  discovered the pheromone equivalent of an anti-aphrodisiac.
discovered the pheromone equivalent of an anti-aphrodisiac.
National University of Singapore (NUS) scientist Joanne Yew has found that male fruit flies impregnate a female fly with a cocktail of fats, which puts other males off wanting to mate with her. She spoke to Chris Smith...
Joanne - Pheromones are chemicals that animals use to communicate with each other and we wanted to see what kinds of different chemicals are used by different species of fruit flies. So, the reason we thought we might be able to see something that people haven't seen before is because of a method we worked out a couple of years ago where we use laser to scan the surface of the insects. By doing so, we're able to detect different kinds of molecules then the kind of classical methods that are used to identify these molecules.
Chris - So that laser technique would enable you to study not just what the insect itself is producing, but, potentially, what another insect it's been knocking around with may have left on it?
Joanne - Yes. So, by using a really small laser, compared to the size of the insect, we get really good spatial specificity as to where these different molecules are expressed on a surface and also where they get transferred following some kind of behavioural interactions, like mating.
Chris - And that particular laser technique doesn't just tell you where on the insect. It will actually give you the chemical identity of the substance, will it?
Joanne - Well, it gives you a good guess. So, based on that guess, you know what is the elemental composition, what kinds of different atoms are there and, probably, number of double bonds there.
Chris - When you started doing this with these fruit flies, what did you see that stood out as clearly different or exceptional compared to what previously people had managed to spot using traditional techniques to study pheromones?
Joanne - So, we had an expectation of the kinds of molecules we would see based on their mass. So, we had a laundry list of things to look for; but what we saw that was novel was that, in males only and not females, there were a series of molecules that looked a lot heavier than what had been reported before. And then, when we looked at things like the elemental composition and number of double bonds, it seem to fit the characteristics of what we would expect from a particular kind of fat.
Chris - So, these male flies are depositing into the females' fats which appear to be linked to mating, or influencing mating behaviour?
Joanne - That's right, yes.
Chris - So, what are these fats and how do they change the behaviour of the female and when does the male put them on the female?
Joanne - They are transferring a particular kind of fat called a triglyceride. This is a common kind of fat molecule and the males express it in a region close to their genitals. And so, when they mate with a female, they're transferring sperm as well as these particular fats.
Chris - The triglycerides go onto the female, but do they affect behaviour of the female or do they affect behaviour of other males?
Joanne - We looked at both aspects of that. Females, after they mate, they exhibit a series of rejection behaviours where they really are not interested in mating anymore for some reason. And so, the male who tries to mate with them, she will kick them in the head and run away. And so, we tried to see, if we perfumed the female purely with these triglycerides and not have her mate with the male, would it cause her to do any of these behaviours? But it doesn't seem to be the case. So, it seems that it's more to tell other males to avoid this female.
Chris - Let's just run over that. The female that you coat in this material, in these triglycerides, does not appear to be less receptive to mating, but she is actively avoided by males that could potentially mate with her?
Joanne - Yes, that's right.
Chris - Do you know how the males are picking it up or how they're responding to it? And if they do respond like this, why don't they get confused with their own smells and therefore just go into a non-mating state?
Joanne - Yes, we speculate because its context-dependent and so they're smelling this smell on top of the female's natural scent. That whole bouquet of the female scent, plus this added male fat, is probably the signal that tells other males to avoid this female.
Chris - Now, you've looked just in these insects in this first instance, but do you think that this whole concept - for want of a better phrase, an anti-aphrodisiac, do you think this applies in other animals too and dare I say, even in humans?
Joanne - I think so. I mean, I know so because there is actually large body of literature showing this phenomenon in other insects. In humans, this is hard to say, especially in terms of chemical signals, but it benefits both the male and female to have some kind of signal indicating a period where the female is not interested and when males won't be successful. So, it seems reasonable that it would be a conserved mechanism...
23:10 - Why are some people left handed?
Why are some people left handed?
with Chris McManus, University College London.
We find out about asymmetry in nature: by firstly examining our hands. Do other species have handedness? Is it genetic? And is it true that left handers are bad at language?
Chris Smith spoke to UCL's Chris McManus, author of the award-winning book Right Hand, Left Hand....
Chris - why are some people left handed and some people right handed? Why do humans, and other species, have this asymmetry? And does it matter?
Chris M. - It matters because we're better at using one than the other, but why is it? Why are we better at using one? Well, there are several reasons there. One is it actually always benefits just to use one or two things more than the other.
So, there are monkeys in the wild, chimpanzees who are searching for termites or things like that. They stick a stick into a tree, pull out the termites and they eat them. Half of them is their right paw and half of them is their left paw. The ones who try and use both collect less termites than the ones who specialise in using just one. So, it pays to practice and it's always better to use one rather than the other.
Chris - But am I right in saying you don't see the majority of chimpanzees in the wild who are right-handed in the same way if I went to anywhere in the world and look at any human. I'm going to find that same right-handed dominance.
Chris M. - That's right. Almost all animal species are a mixture or sort of racemic mixture of half right and half left whereas humans, it's 90% right-handed,10% left-handed. That's something that needs explaining. So, something else must be explaining that in addition to being specialised. It's a sense in which we're all right-handed, it would be much easier because we'd be right-handed because we're the same as everybody else. It's easier if the whole society does things the same way, just the same ways it pays to all drive on the right same side of the road, but we're not.
Chris - Ben...
Ben - Is it true that we're all born with a handedness or does it develop?
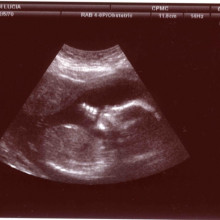
Chris M. - Yes, we're born and in fact, there are studies where you look at ultrasound studies of pregnant women and you can watch the foetus moving around inside the uterus. Lots of foetuses as early as 4 or 5 months of pregnancy such one thumb and about 90% suck the right thumb and 10% suck the left thumb and you follow them through, and they become right and left-handed. So, it's there before birth, yes.
Chris - I was going to say because when my daughter was born, she is left-handed. She is 7 now, but she was very little and she'd sort of be sitting in a high chair feeding herself with one hand or the other, or when she used - she has this thing about pens, loved pens. And I used to sort of play devil's advocate and try taking the pen out of one hand and put it in the other hand, in the right hand, to see what would happen and it immediately went back to the left. So, very early, very, very early, there is this bias, but why?
Chris M. - There is a bias, but it's actually not as easy to see as you might think. I get a lot of parents saying, "Are they going to be right-handed? Are they going to be left-handed?" And children neonates go through a phase up to about a year, 18 months which is called the chaotic phase where they seem to be trying out both hands. I think part of this is just trying to understand how hands work. It was easy in womb. They were floating around in this sort of space-like fluid environment and they could easily move one thumb to the mouth and so on. Put them in air with gravity and life gets a bit more complicated. Plus, they're trying to sit up and do other things. They're exploring a lot of things, but then it's usually by about a year or two years of age that it becomes clear if they're right or left-handed. They can move around.
Chris - What happens if you look in the brain though at someone using their right and left hand who has a right-handed bias or a left-handed bias? Is there clear differences in the way the brain is working?
Chris M. - Well, there's clearly very enormous differences. I'm right-handed and as I move my right hand, the left half of my brain is controlling that. So, I'm doing things from the left half of my brain. Now, part of the story there is that not only am I moving my hand with the left half of my brain and also, talking to you with the left half of my brain because language is in the left half of the brain in most people as well.
Chris - But why should that matter, Chris? Why should it be important that language is in the same part of my brain as my handedness is?
Chris M. - We don't know, but we do seem to find that when you get people who get these modules in the two hemispheres of the brain and there are lots of them in different combinations, some of them start to be disadvantaged. So, presumably there's some sort of optimal biological combination. I suspect it's to do with how bits of brain communicate with other bits of brain because it's a lot of different areas doing different things at the same time. Sometimes it has to move across between the hemispheres which is not a very fast process. Sometimes it has to move backwards and forwards. So, presumably there are optimal ways of doing that and one is to have the hand controlling your speech, language, so the brain hemisphere controlling your language, the brain hemisphere controlling your hand next to one another. And that's probably why I'm waving my right hand around in the air as I'm talking to you.
Chris - You're speculating wildly, but what about the genetics of this? I said my daughter is left-handed. My wife and I are right-handed, but my dad was left-handed. So, is there a gene which causes left-handedness? Does it run in families?
Chris M. - Well, it certainly runs in families and if you take two left-handed parents, they're much more likely to have left-handed children than if you have two right-handed parents. Having said that, they're about three times more likely but still, three quarters of the children of two left-handed parents are right-handed. So, that makes the genetic slightly strange.
Chris - Not easy, yeah.
Chris M. - It's not easy. It does look as it runs in families. There's a lot of chance involved and part of the trouble is, we can't see that very easily in small modern families. Charles Darwin had 10 children. His wife was left-handed and two of those children were left-handed. But with 10 children, it becomes a bit more obvious what's going on.
Chris - What about the power of modern molecular biology and the so-called genome wide association where you can go through enormous numbers of people in the population looking for genetic signposts that sort of co-associate. They crop up at the same time in certain traits. What happens if you do that to handedness?
Chris M. - Well, we tried that and we looked very much. We published the paper last year on it and we hope very much to find the gene. We found nothing and most of the point, we had the statistical power to provide it if it were there. So, it wasn't there.
Chris - So, there's no one gene that causes that?
Chris M. - There's no one gene, but what there does seem to be is, it's more than possible that there's a large number of genes. All of which have small effects. In that sense, it's no different to height, intelligence, weight, and a thousand other complex traits that we're looking at. I think probably what's happening is that you have a left-handed child and a left-handed parent and they will have the same gene producing that. But I have a left-handed mother and a left-handed daughter and it will be a different gene that's made our children left-handed.

29:53 - Can chemicals tell right from left?
Can chemicals tell right from left?
with Stephen Driver, University of Cambridge
So humans have handedness, but is the same true for chemistry? Harriet Johnson was 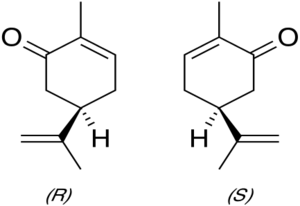 joined by Stephen Driver from the University of Cambridge.
joined by Stephen Driver from the University of Cambridge.
Steven - there are certainly left-handed and right-handed versions of certain types of molecule.
Harriet - And so, this affects the way that they work?
Steven - Chemically, they're the same, but in biological systems, it can be very important to keep track of the differences between the left-handed versions and the right-handed versions.
Harriet - I went to Hills Road Sixth Form College in Cambridge, armed with a smell challenge, using a chemical called carvone, kindly provided by (Dr. Stephan Hugh) from the University of Nottingham. First, I wanted to tell how well the students and staff could identify the smell of carvone. So, I gave a group of them a tube to sniff.
Female - Peppermint.
Female - Peppermint.
Female - Peppermint.
Male - Spearmint.
Male - Mint.
Harriet - So, that was a pretty clean sweep with mint, but I gave another tube of  carvone to a different group and they had a little more trouble.
carvone to a different group and they had a little more trouble.
Male - I think it smells a bit lemony. It also reminds me of baby wipes.
Female - Maybe something you'd have in like shower gels or something.
Female - I thought it smell like aniseed or liquorish.
Female - I wouldn't cook with it. It sounds like musty clothing to me. I don't like it. It's something that makes me could've think of it (yuck!)
Chris - And that was the same stuff?
Harriet - Well, both groups were given a tube of carvone except one was this tube labelled s-carvone and the other one was labelled r-carvone. So Steven, what's the difference?
Steven - So, it's all about carbon atoms. So, the carbon atom wants to form 4 chemical bonds and the four chemical bonds want to get as far away from each other in space as they possibly can. So, they've taken a particular geometric arrangement called the tetrahedron. So imagine if you like, three bonds pointing downwards and outwards forming a little tripod and fourth bond pointing straight up. So, if you put different atoms or different chemical groups onto those four bonds, you form a molecule. It turns out that you can do that as it were in a car with a left-handed way or an anti-clockwise way and the clockwise way. What you have then are molecules that are the mirror images of each other. In chemistry, we call those enantiomers and we say that molecules are chiral. So, they're chemically the same but biochemically, they can be very different.
Harriet - So, they have a different shape and this shape works in different ways.
Steven - And one is the mirror image of the other.
Harriet - Yeah. So, there's different shapes in each of these tubes. So, the S-form is what we'd call left-handed form. And so, that form of carvone is found naturally in caraway which people seem to have a hard time identifying whereas the right-handed is found in mint which we saw people could recognise quite easily. So, these are bonding with the receptors in their noses. So, to test if they could see it the other way, we swapped the tubes around in the groups and this is what they thought...
Female - It smells like a mixture of lemon and mint.
Female - I think I viewed it like in Chinese cooking.
Female - Caraway.
Male - Pepper.
Male - Vaguely, eucalyptusy.
Harriet - And investigating the smelling abilities of the second group, here they are with the right-handed carvone...
Male - I think it smells minty.
Female - Yeah, just like a peppermint.
Female - I think it's spearmint.
Female - Toothpaste.
Female - My aunt.
Harriet - Apparently her aunt did always carry mints on her. So, that was triggering her aunt receptor in her nose. So Steven, is it just smells that this is working with or are there other effects everywhere else?
Steven - There are other effects everywhere else as well. So, in terms of applications, one very important area is pharmaceuticals for example. Artificial senses is another one. We've talked about the nose as a biological sensor, but if we want to make artificial senses, little gadgets that can detect molecules, it may be that they need to be sensitive to the left-handed and right-handed forms as well.
Harriet - And so, there could be serious consequences if the chirality of the shape of the molecule is the wrong way around as we see in some drug examples.
Steven - That's right. So, there are examples we're one enantiomer, so one chirality of the drug molecule is effective and the chirality either is not effective where it can even be harmful. So, it can be very important to get that right.
Joanne - Yeah, the classic example was the thalidomide drug that was prescribed that was completely safe in one form and then they didn't know that the other form would cause these birth defects and that's how that accident happened, right?
Steven - I believe it's actually slightly more complicated than that in that particular example.
Chris M. - I think the problem is that if you have the pure one, then they spontaneously turn into the other one. So, they haven't got a half-life which is very long, so you can't give the pure version.
Harriet - So, it's really difficult to control the shape of these molecules.
Chris M. - Of that molecule.
Harriet - So Steven, this is what you research at the minute, trying to create molecules that only go one way around?
Steven - That's right. So, there are ways of doing that and there are ways of doing that that currently I used in the chemical industry. But they involve a technique known as homogeneous catalysis. So, I need to explain what a catalyst is. If you're trying to do a chemical reaction, you're taking reactants and converting them into products. Often, you need to control how that's done. So either you need to speed the reaction up or you need to push the reaction towards a particular product. In this case, we want to push it towards a particular chirality of product. So, homogeneous catalysis, imagine that your reactants are liquid, your products are liquid, and your catalysts is also a liquid. So, everything is in a liquid phase and you do your reaction, you get your product. Then you have to somehow separate out any leftover reactants and the catalysts so that you can re-use it. That's doable and that's done in industry, but it's difficult. So, we're interested in a different way of doing that. it's called heterogeneous catalysis. That's widely used in the chemical industry and in car exhaust catalytic converters for example. What's happening there is that the reactants of for example, gas phase, the products of also gas phase, but the catalyst is a solid. Typically, it's a metal solid, so it's a surface of a metal. So, we're looking at whether it's possible to use metal surfaces to do enantioselective heterogeneous catalysis.
Harriet - And how is that working out for you?
Steven - Well, we're getting promising results. So, we're looking at how these kind of molecules interact with metal surface whether they're either symmetric surfaces that is or asymmetric. I'm looking at the different ways in which chirality can manifest itself.
Harriet - So very quickly, we saw in the carvone experiment that natural systems like the spearmint leaf are controlling chirality in the oils that they're making. So, why is it so hard to replicate this in situ, in the lab.
Steven - I don't quite understand the question.
Harriet - Sorry. So, these things are happening like in our bodies. We're making chemicals that all go the same way around. So, why is it so hard to do it artificially when you're synthesizing chemicals like drugs?
Steven - I think it's not so difficult to do. It's difficult to separate the products. So, our interest is simply in moving from homogeneous to heterogeneous catalysis to make it an easier industrial process.
Harriet - Okay, fantastic! Good luck with that. Thank you to Steven Driver from Cambridge University.

Are left handed females a rarity?
Chris M. - I'm not going to go down that road, but I think it is rarer to be left-handed in a woman. We reckon there's about 5 left-handed men for every 4 left-handed women.
Chris - Why?
Chris M. - We don't know. There we are, that's easy. Gosh! No idea at all.
Chris - That's unusual, isn't it?
Chris M. - Yeah, but sex keeps rearing its head in the story somewhere.

Which hand is best for sport?
Chris M. - It depends what the sport is. If it's golf there seems to be very little advantage where it's just you and the ball and a hole. If it's tennis or something like that where you're playing with other people then if you're right-handed, you don't play so many left handers. With left handers, mostly play right-handed so they've got the advantage over you. If it's baseball then the pitch itself is asymmetric. The left hander is actually one step closer to first base in the right hander after he'd hit the ball. So then the left handers are advantage from that reason as well.
38:20 - Spin and antimatter
Spin and antimatter
with Ben Allanach, Cambridge University
From a distance the laws of physics and the Universe at large look pretty symmetrical. But, drill down into the fabric of the matter we're made of, and some glaring asymmetries emerge. Chris Smith spoke to particle physicist Ben Allanach from 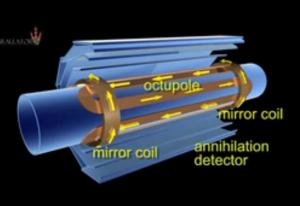 Cambridge University...
Cambridge University...
Ben - It's a weird thing when you look at your hands and they look as if they've got mirror symmetry. They don't look particularly different. And macroscopic large objects don't seem that different through a mirror. But the weird thing is, when you look at the smallest bits of matter; particles like electrons, the outer bits of atoms, they look asymmetric in a very special way. And also, there's an asymmetry between matter and anti-matter which is very important for our existence.
Chris - When you say an asymmetry, I mean, let's just back up a bit here because when we're talking about matter and antimatter we're going right the way back to the Big Bang and the fact that energy gets converted into material in the Universe. And there's not really a fundamental reason why we shouldn't have equivalent amounts of matter and its mirror-image equivalent anti-matter. But, what we see the Universe is made of is matter...?
Ben - That's right. We're all made of matter. There's not much anti-matter around at all. You can make it at places like CERN when you convert energy the other way into matter and anti-matter. The question is, why did we end up all being matter? If you do the sums in the early Universe, the early Universe was a hot cloud of gas and there's matter and anti-matter bouncing around. If you work out the equilibrium of that, you're going to get pretty much equal amounts to a very good statistical average.
Chris - Based on our present models...
Ben - Yeah, based on our present understanding of physics and experiments like CERN give us that understanding.
Chris - But, obviously, when we make the observation, we see we're all made of matter and, as you say, anti-matter, is vanishingly rare. So where's it all gone?
Ben - In fact, when you bring matter and anti-matter together, of course, they annihilate into radiation. So particles of light for example will come out of that. And so, the question is more, why aren't we all made of light, right? Because if you have equal amounts...
Chris - Well some of us are stars aren't we Ben, like you?
Ben - That's right. I think I'm a bit heavier than that! So, there's got to have been some slight difference between matter and anti-matter. In fact, in the particle physics experiments at the end of last century, those differences were starting to come out. So, they feel forces very slightly differently, not to a large extent but if you do the theory of what's going on, you need a slight difference between how matter interacts and how anti-matter interacts, along with some other conditions in the early Universe. And you get a slight tilting that favours one over the other and, in this case, favours matter over anti-matter. And you have to follow all this complicated maths through, but as long as you've got a slight tilting - there's 15 billion years in between where lots of boring things happen - but we end up, luckily, being made of matter and not light!
Chris - But I mean, I was watching Frank Close talking the other day. He's got a video on the internet from when he was giving his talk at the Royal Society - because he got the Faraday Medal this year for public understanding of science - and he was making some of these points. He was sort of saying, "Do you think it's a mistake, or just by chance, that we have arrived at this situation where physics means that we can exist and we're not just energy or light? Or do you think it's extraordinary that, when we compare the nucleus of an atom and the charge on a proton its exactly counterbalanced by the charge on an electron? But then an electron is just one thing whereas if we zoom in on a proton, it's actually got three other little particles inside it. Those are the quarks aren't they? And actually, there are three of those but two of them are sort of 2/3'rds, the other is 1/3, and they balance out to be about the right amounts so that it completely opposes an electron and the two counterbalance. That's extraordinary and it's too much of a good outcome to be chance, isn't it?
Ben - Yeah, I mean the accuracy on those charge measurements is fantastic. I mean, it's one in ten to the ten or something, but I think there's an underlying reason for that. The underlying reason could be the Grand Unified Theory which says that the particles which make up a proton and the electron are two different aspects of some underlying particle where an asymmetry has been introduced in that which splits the two off but it explains exactly why you have this quantization of charge. And the charge of a quark comes in multiples of a third of the charge of the electron. So, there are other examples of theories where you get a precise mathematical relation between the charges of things. I suspect, it's probably due to that, rather than I don't know, there being a billion universes and you just pick at random one where we can exist.
Chris - Are you not in favour of the idea that there might be multiple universes? This is the sort of Michio Kaku parallel universes where we just happen to inhabit the one where the rule of physics work for us.
Ben - The problem I have with it is that you can't test that theory. So, it's really interesting to speculate about it, but I don't think we can ever know even in principle and so, it's outside of science.
Chris - I thought the whole idea was, we're trying very hard to detect gravity waves propagating because the theory says, if we've got these parallel universes then gravity ought to be able to transit between them. And therefore, if we can see the waves of that gravity coming from one to the next, it sort of says they exist. Do you think that because we haven't found it therefore that disproves that these parallel universes can exist then?
Ben - No. The question is, is there any point in space and time where our universe is connected to one of these so-called parallel universes? If there is, I'd count that as being part of our universe because where space is....
Chris - Isn't that mathematical trick, Ben. Are you being crafty?
Ben - That's what we do. That's what we get paid for. No, I mean, if it's connected in space and time it is part of our universe. But you're right. If you've got some foaminess, you should be able to detect that. But I don't count that as this multiverse theory, but it would certainly be very interesting if most of the time, it's not connected, but you know, back on Tuesday last week, it was and you could detect the gravitational radiation from that. That would be one way of me eating my words. But if it goes the other way and you don't see the gravitational radiation, you can't say one way or the other and I suspect that that's going to be the case. And so, that's my problem with it.
Chris - Steven Driver...
Steven - That's just wiped out a whole genre of science fiction!
Chris - Oops! Chris, in your book, Right Hand, Left Hand, you actually make the point early on in there - and this is something that Pasteur said in the early days - that, asymmetry begets asymmetry. So, starting with the asymmetries in the tiny particles that Ben studies, that leads actually up to a whole organism - or even a whole world - with gross asymmetries in it...
Chris M. - That's right. If you keep backtracking whenever you find an asymmetry, something else must have caused it and you can keep going back and back, and back and you end up exactly where Ben is, yeah...

Do athletes use both hands?
Chris M. - There are probably what we call mixed handers and about 10% of the population writes with their left hand but about 20% throws with their left hand. And that means there's about 10% who are left-handed throwers and right-handed writers and probably, they're over represented in sport.

What does D, L, R and S stand for?
Steven - There are simply different systems that have come about for naming them. So, for example, the D and the L, they refer to the way in which certain substances rotate the plane of polarisation of polarised light. So, D is dextrorotatory, L is levorotatory, so right and left. That's one of them. Another of them is another set of arbitrary rules for the order in which bonds are substituted.
Harriet - Thanks and the R is recto and S is sinister, I think.

Is the Universe symmetrical?
Ben - Yeah. Actually, physicists are looking for that. you can look at the afterglow of the Big Bang, the cosmic microwave background and trying to look for that. I think the fair statement is so far, no evidence, no clear evidence for that has been found, but you know, it's a subject of active inquiry. The universe could be spinning which would've caused to give some kind of handedness and should leave some imprint in the afterglow.
Harriet - Chris?
Chris M. - A variant on that one is a while ago, there were claims that there may be more left-handed than right-handed galaxies and they had people going through images of...
Chris - The galaxy zoo.
Chris M. - That's right and going through classifying them. To start with, it looks about 52 to 48% I think and when they look more closely, it was mostly biases on the part of people looking at these things rather than genuine differences. We're not very good at distinguishing right and left in everyday life in fact.
47:45 - Is technology steering human evolution?
Is technology steering human evolution?
Hannah - So, are our modern lifestyles directing the way us humans evolve? Over to Robert Foley, Professor of Human Evolution at Cambridge University...
Robert - Evolutionary changes usually occurs through small genetic shifts which are then selected for and spread through a population. For humans to evolve significantly in a new direction will take immense selective pressures. selection like this, fingers coping with touchscreens is unlikely to be really very strong and certainly, unlikely to be strong enough to spread through such a large population of the human.
Hannah - Still concerned about the speed of natural selection and my digits, I dial up John Armour, Professor of Human Genetics at Nottingham University.
John - Spreading genes by natural selection is incredibly slow. It's a generation by generation process, but it's effective. Humans, because we have ideas and communication, we can spread ideas and technologies and tricks if you like much, much faster than we can spread genes. Of course, we can spread ideas to people we're not related to, where genes by natural selection can only be pass on to descendants. So, I think the answer is that engineering at the market will fix a new touchscreen tablet is easier for human beings as opposed to human beings evolving to adapt to the touchscreen pad.
Hannah - And on Facebook, Gerald McMullen comments that technology changes too fast and natural selection too slowly to make any advantage apparent. Back to Robert Foley for this words of caution.
Robert - Having said that, there are possible ways in which things will change as we use computers more and more. It may not affect our muscular physiology or skeletal biology of humans, but it could well affect the way our brains are wired, particularly as these are often developed interactively with the environment in which we grow and develop.










Comments
Add a comment