Shark Camouflage in Australia
This week, we have a final show from Perth in Western Australia. Chris Smith and Victoria Gill find out how camouflage wetsuits might help protect surfers from sharks, hear about a new development in muscular dystrophy treatment, how sea sponges can be used to mend fractures and whether the chemicals that a cell produces just before death can help us reverse the damage caused by stroke. In the news, why money makes the world go round, the comet that will be lighting up the skies in November, the eniromentally green military flares that could result in clearer firework displays and the scientists that have produced the world's most accurate clock.
In this episode

01:12 - Why Does Money Make the World Go Round?
Why Does Money Make the World Go Round?
We only have to look at the effect of the financial crisis recently to know that  money makes the world go round. But Gabriele Camera and her colleagues considered in PNAS this week why exactly money is essential for the functioning of modern society.
money makes the world go round. But Gabriele Camera and her colleagues considered in PNAS this week why exactly money is essential for the functioning of modern society.
Modern society involves us interacting and co-operating with hundreds and thousands of strangers, whereas our ancestors very survival depended on working with small groups of people, all of whom you would know and trust intimately. The researchers were interested in whether money played a role in facilitating this co-operation with people we have no knowledge of.
First participants took part in a control task. Paired up with anonymous strangers each was randomly assigned the role of Producer or Consumer. Producers were given the option to share some of their good with Consumers (who would be rewarded for the transaction), on the presumption that when the roles were reversed others would happily do the same in the future. Researchers found that as the group size increased, the co-operation decreased. When only two people were in a group 70% of meetings resulted in co-operation, but in a group of 32 it was only in 28.5% of meetings that people were willing to give away their goods, trusting in a future return. So we can see that in large unfamiliar societies, like we have today, co-operation would be low, but does money help to change that?
Next researchers introduced tokens to the scenario. There were purely symbolic and couldn't be exchanged for either goods or real life money. Now when pairs met they had the option of giving away their goods, not doing anything or asking for a token in exchange for their goods. When both of the pairs decisions matched a transaction took place. The team found that the introduction of tokens, even though they were only symbolic, increased the number of people willing to co-operate in large groups. In the group of 32, co-operation increased from 28.5% in the control test to 51.7%.
These results imply that money, is an evolutionary stable way of allowing large groups of strangers, like the modern day societies we have today, co-operate and therefore plays a key role in how modern societies function. However it does have a down side. When tokens were introduced, although co-operation stabilised, no-one was willing to offer their goods as a gift anymore.

05:14 - How are comets like cats?
How are comets like cats?
with Robert Massey, Royal Astronomical Society
Earlier in the year, astronomers were making predictions that a very bright  comet will be visible in the night sky in November and December. But over the past few months, it hasn't been brightening quite as much as some people were hoping. Many people are now rather doubtful about how spectacular it might be. But why are comets such unpredictable objects? To find out, Dominic Ford caught up with Robert Massey from the Royal Astronomical Society
comet will be visible in the night sky in November and December. But over the past few months, it hasn't been brightening quite as much as some people were hoping. Many people are now rather doubtful about how spectacular it might be. But why are comets such unpredictable objects? To find out, Dominic Ford caught up with Robert Massey from the Royal Astronomical Society
Robert - It's contentious. The object that we were looking forward to at the end of the year I think still are is comet ISON. It was discovered by robotic network of telescopes in Russia and it's been named after that, although to be fair, there are actually a couple of scientists who probably need to be credited for it as well. But this object was thought, because it was coming very close to the sun and also, was very, very active a long way from the sun, the supposition was, that when it got close into the inner solar system, it would become very bright and certainly once it passed by the sun and was heading out in space again.
Now, there is some debate as to whether that's still likely to happen because the activity levels that was on the comet haven't lived up to expectations. Now, this is actually nothing unusual with comets. Quite often, they can be active when they're far out particularly if they haven't come in to the solar system before and that looks like it might be the case for ISON, and then the activity can fall off a bit and that seems to be happening here. So, it may not be quite the object that people were expecting and there's always been an unfortunate tendency for these things to be hyped. But that said, it can still be quite a nice comet as these things go. It could still be visible to the naked eye late in the year, particularly in the northern hemisphere. So, we in the UK might get a really good view of it in the last couple of weeks in the year. But we'll just have to wait and see. It's one of those things where comets are a bit like cats. The description is that they have tails and they do exactly what they want.
Dominic - What does that tell us about the structure of the object?
Robert - The difficulty in understanding their activity is that they can have a variety of materials. I mean, they have ices, mostly frozen water and other gases as well inside. But when they get close to the sun, that stuff heats up and it goes straight from being ice to gas because you need some kind of atmospheric pressure for a material to become liquid, so it doesn't go through that phase. Now, the determining factor is really, how much of it is exposed. If the comet has quite a thick rocky crust or debris crust or dusty crust then it's much harder for the heat of the sun to reach inside and then also, there are fewer vents for the material to come out. If that's thinner then you'd expect more of the material inside to come out. So probably, those factors, the exact proportion of water ice and all those things determine how active the comet is likely to be.
Dominic - From memory, when ISON makes its closest approach to the sun in November, it's going to be actually incredibly close, that will be a very extreme environment that those ices will be exposed to.
Robert - Well, ISON is going to go within about 1.1 million kilometres from the sun's surface. Given that the sun is 1.3 million kilometres across, that's really a very close approach indeed. Now, there's again, a lot of debate about exactly what will happen because there is another object, comet Lovejoy that made a similar approach a couple of years ago and turned out to be quite robust. It actually came though it quite well.
There was a paper by the astronomer Royal of Scotland, John Brown on this around the same time and he pointed out that you have to consider that although it's a hot environment being that close to the sun quite clearly, there isn't much in the way of the solar atmosphere at that point so objects can pass through it. The ones that plunge directly onto the sun's surface clearly, are going to be destroyed, but further out, it's a lot harder to tell. So, I think the jury is out on whether it will survive. If it does survive, it may turn out to be a very nice comet indeed we just don't know.

08:57 - Quick Fire Science: Nuclear Power
Quick Fire Science: Nuclear Power
This week, nuclear expert Mycle Schneider, formerly an adviser to the French and German governments has said that he's deeply worried about contaminated cooling water leaking from tanks at the site of the Fukushima nuclear reactors...
- Huge amounts of energy can be released by joining or fusing small atoms together to make larger atoms, or by splitting apart larger atoms like uranium.
- In nuclear reactors, atoms such as Uranium 235 and Plutonium 239 are bombarded by neutrons which causes them to split in two, a process called fission.
- When atoms undergo fission, they often release more neutrons, which can go on to hit another atom, creating a chain reaction.
- A kilogram of uranium in a nuclear reaction can release more energy by fission than 10 000 tonnes of coal, gas or oil, and all without releasing any greenhouse gases.
- In a nuclear bomb, all of the energy from several kg of uranium or lutonium are emitted within a single microsecond, creating an immensely destructive burst of energy.
- The first controlled release of nuclear energy was in a reactor built in 1942 under the stands in a chicago university american football stadium. But it wasn't producing power, just plutonium to build America's second nuclear bomb.
- The first commercial nuclear power station was Calder Hall in Cumbria. It was built in 1956 and generated 60 megawatts of power.
- Unfortunately many of the atoms left over after fission are unstable and can release some of their remaining energy over days and years in the form of high energy particles and gamma rays, known as radiation.
- This radiation can damage the DNA in cells which can cause cancer, or in very large doses radiation sickness.
- On average less than a third of 1% of your annual radiation exposure is due to nuclear bombs and nuclear power. Between a third and a half of your exposure is due to medical procedures such as X-rays and the rest is due to natural radiation.
- Today in the uk 19% of electricity is produced by nuclear power compared to 4.6% by renewables and the percentage is dropping as older plants are being decommissioned.
- The last nuclear power plant to be built in the UK was Sizewell B, completed in 1995...

11:14 - When Will Greenland's Ice Sheet Melt?
When Will Greenland's Ice Sheet Melt?
with Anthony Payne, University of Bristol
Greenland is home to some of the largest ice sheets in the world, and if those melt, they could cause global sea-levels to rise by as much as several metres. For low-lying parts of the world, including Cambridge, that could be very bad news...
So, how much of a rise in global temperatures would be required for those ice sheets to melt? Anthony Payne, from the University of Bristol, has built computational simulations to find out, which he presented in the journal PNAS this month.
Anthony - Greenland represents one of the major sources of freshwater in the world and is the second greatest potential contributor to future sea levels with something like 7 or 8 meters worth of potential sea level rise. So that's why a lot of research is centred on understanding what's happening in Greenland over the last decade.
Dominic - You've looked at two mechanisms by which this melt can occur - both the melting itself and also, sliding of that ice. How do you separate those two effects?
Anthony - We did two separate sets of experiments. One, where just looked at the effects of melt by itself. We incorporated results from recent climate models on how air temperatures were warmer in Greenland and how that will affect the amounts of ice melt in Greenland. Then in the second set of the experiments, we allow that additional melt water to interact with the flow of ice sheets, with the idea being that if there's more melt water around, then potentially, the flow of the ice sheet could be lubricated and therefore, the ice sheet would flow faster.
Dominic - How do you go about doing those experiments though?
Anthony - Well, the experiments are performed using ice sheet models that run on supercomputers and typically take 2 or 3 days to run. You give the model information on the geometry of the ice sheets, how much ice there is there, or where it is on Greenland, you give information on things like future climates, how much warming is there like to be over Greenland, how much melt will there be over Greenland, and how much snowfall will change over Greenland. The most important part of our work has been to develop a mathematical relationship that depicts how increased amounts of melt water could affect lubrication and therefore, the flow of the ice sheet.
Dominic - I'm imagining that lubrication must be a very difficult thing to model because there must be all sorts of factors affecting how stable that ice is.
Anthony - What we have done is to take a shortcut and using empirical field data to parameterise the effect, which means that we treat the effect as a black box and we use field data to say, "There's this amount of imput i.e. melt water. This is what the observation suggest the output would be." In particular, what we've used is information on the seasonal speedup of the flow of the ice sheet. So, they use GPS receivers scattered over the ice sheets, that record the velocity of ice flow, and then you can look at the records from those recorders and they'll show that the ice flow speeds up in the summer or most likely, the early spring, and then slows down in the winter. So, what we assume is that the winter flow is with no lubrication and the summer flow is lubricated. So, if we look at the ratio of those two, we get a handle on how important the melt water effect is.
Dominic - Are you validating those models in comparison with your observational data from the ground?
Anthony - So, the validation that we have attempted is to compare observations of ice flow against what the models predict. On the whole, they do a fairly good job.
Dominic - And obviously, you're also very dependent on these models of the future climate to know how this melt will proceed in the future. Where are you getting this from?
Anthony - For our particular study, we cooperated with Zavio Satweis who works in the University of the Age and does regional climate modelling of Greenland. But I think the important thing to say is that, when you compare his models to other models that have been used to predict what's going to happen in Greenland, his model falls pretty much in the middle of the group. The results we get are so strong that we think we could use any climate model and get similar set of results.
Dominic - Often, with these questions of climate change, you find yourself in what we call very nonlinear processes where suddenly a process reaches some critical point where it takes off. How do you know that as the climate changes, we're not going to move in to a very different regime which your models aren't covering?
Anthony - The modelling wasn't intended to be a prediction of the future. It was intended to assess whether a particular set of effects, those related to basal lubrication of melt water availability were going to be important. So, while there may be lots of other thresholds in the system that we're currently unaware of or weren't the subjects of the particular paper that we've written, we're after understanding what affect this particular effects and whether this particular effect is going to be important or not.
Dominic - And what's the conclusion? What kind of sea level rises might we expect to result from this?
Anthony - We found that there was very little sea level rise associated with this particular effect. Our ballpark figure is roughly 8 millimetres by 2200 which in comparison to other sources of sea level rise over the next century is fairly minimal.

16:43 - Green Flares for the Military
Green Flares for the Military
A discussion about green flares might bring to mind the colour, but the military  also cares about their pyrotechnics environmental credentials. 'You might not think the military and environmentally friendly go together,' explains Jesse Sabatini from the US Army's Picatinny Arsenal, 'but they really do.'
also cares about their pyrotechnics environmental credentials. 'You might not think the military and environmentally friendly go together,' explains Jesse Sabatini from the US Army's Picatinny Arsenal, 'but they really do.'
As regulation surrounding soil and groundwater contamination increase, the simple act of training on a military range can have wide reaching implications. Sabatini and colleague Jared Moretti are working to make the tools of the military a little more green and in their most recent work have replaced the perchlorate salts in flares with the cheap and widely used 5-amino tetrazole (5-AT). The goal, stresses Sabatini, was to match the performance of in-use formulations, not just meet the minimum required.
Perchlorate salts have been widely used as an oxidiser in explosives and pyrotechnics because they're stable but supply a large amount of oxygen when burnt. And because perchlorate salts are so widely used and easy to make, they are also cheap. However, concerns surrounding the role of perchlorate salts in human health are growing and the US now has stringent limits on the amount of perchlorate allowed in drinking water (15 parts per billion federally, although some states have set their limits even lower). That's led to some military ranges being closed for clean-up and troops being unable to train extensively with the flares they might have to use in combat.
Sabatini has already come up with a solution to the problem, or rather two, by replacing the perchlorate with high nitrogen salts of strontium and barium for red and green flares respectively. This proved that pyrotechnics can work with high nitrogen compounds instead of perchlorate salts, something already established in propellant and explosives research. But, says Sabatini, there are a number of drawbacks to using such specialised compounds. 'You've got a big cost issue because, number one, you've got two different syntheses you have to do and neither of them are commercially available. That obviously adds to the cost,' he explains, adding that qualifying ingredients and new formulations also adds to the cost.
Energetic punch
So Sabatini teamed up with his colleague Moretti to look for a cheaper alternative that was still high in nitrogen. Nitrogen-rich compounds don't release large amounts of oxygen, like perchlorate does, but instead release large amounts of energy as they break down to release nitrogen gas. That energetic punch can kick-start combustion.
After trying various compounds that looked like they might do the job, the pair settled on the small molecule 5-AT, which is used in explosives, airbags and the pharmaceutical industry. That large market means the compound is much cheaper than Sabatini's metal salts. By tweaking the formulation, Sabatini and Moretti made perchlorate free flares using 5-AT that matched the performance of two currently used flares, the red M126 A1 and green M192.
'I'm always impressed with the work that Sabatini does,' says Thomas Klapötke of the University of Munich, Germany, who also works on energetic materials. 'Not only is it scientifically sound but he also has the right feeling to make it applicable. There's no point in replacing perchlorate with something less toxic if the price goes up.'
Of course, as always there's a long way to go from lab success to seeing the new formulation replacing the old. A host of tests still await the new flares before they are ready to go in the field.
But if the new formulations work as well in tests then there are many other pyrotechnics that could also be reformulated to remove perchlorate, including noise generating pyrotechnics and fireworks. For those applications, adds Klapötke, there could be an unexpected benefit - 5-AT generates clear nitrogen gas instead of smoke. 'If you go out on bonfire night and look at the fireworks it's often quite smoky. If you produce large amounts of N2 you can increase the visibility of the firework,' he adds. It's a long way to go, but perhaps the general public as well as the military will benefit from this new approach.

19:30 - The world's most accurate clock
The world's most accurate clock
 Researchers at the National Institute for Standards and Technology have built the most accurate clock ever made, accurate to one second in ten billion years...
Researchers at the National Institute for Standards and Technology have built the most accurate clock ever made, accurate to one second in ten billion years...
The clock works by precisely monitoring the frequency at which atoms of ytterbium naturally vibrate, producing light waves of very characteristic frequencies and colours.
The key to making any clock as accurate as possible is to build a system that ticks or vibrates at the steadiest frequency possible. In a pendulum clock, friction between the clock's parts and with the air around the pendulum bob interferes with its steady swing and make it swing unpredictably.
Atomic clocks, on the other hand, are able to achieve much better accuracy than clocks with larger swinging mechanisms, because it's much easier to put single atoms in very controlled laboratory environments, away from external interfering influences.
The difficulty, however, is that the electromagnetic vibrations produced by atoms typically have frequencies of billions, or even millions of billions, of cycles per second. Counting those cycles to keep track of time requires some of the fastest electronics in the world. Although ytterbium atoms are known to be very stable, it hasn't been possible to use them in an atomic clock until now because of the high frequency of the light they produce.
Using time standards like these allow scientists to make very precise measurements of how long processes take. For example, the Earth rotates on its axis once every 24 hours, and that is what gives us day and night. But movements of landmasses and ice sheets can trigger minute variations in the Earth's rotation, which can only be measured with the world's best clocks, but which can tell us about the structure of our own planet.
Physicists want to test Einstein's theory of relativity, which predicts that clocks should run slow when moved at very high speeds, but the effect is so tiny that it can only be measured with incredibly good clocks.

23:58 - Camouflaging Wetsuits
Camouflaging Wetsuits
with Professor Nathan Hart, University of Western Australia
Last year there were eight shark attacks recorded around near Perth in Western  Australia, almost double the annual average over the last decade. Seven of those eight were attacks from the infamous great white shark.
Australia, almost double the annual average over the last decade. Seven of those eight were attacks from the infamous great white shark.
Now a team of researchers from the University of Western Australia has come to the rescue. Professor Nathan Hart's research on shark vision has inspired the design of a wet suit that should be less visible to sharks or may even be shark-repelling. He spoke to Victoria Gill
Nathan - Well, there are actually two suits which have been designed based on the science we've done. The first is a black and white banded suit which is really designed to mimic a sea snake. So, it's warning coloration. There's anecdotal evidence that certain species of shark don't like eating sea snakes although we know that some do. There's also a camouflage design which works on the same principles as camouflage you see army people running around in, but in this case, specifically designed for the underwater light environment.
Victoria - The warning suit then is essentially you're disguising people as sea snakes.
Nathan - That's the idea, yeah. There's obviously some disruptive element to that pattern as well. What we did was to take our knowledge of the shark's spacial resolution, in other words their ability to resolve detail, to set the banding widths and that information was taken by the company who were designing the suits.
Victoria - So, what we're essentially looking at is quite a stripy suit.
Nathan - That's right. It's a stripy suit. We wanted to make sure that it would be visible to the shark from a certain distance away. Although sharks have very sensitive eyes for brightness, they're not actually acute in their vision. So, we're used to being able to discern very fine print in a book for example. Sharks have nowhere near that kind of ability and have very coarse vision, if you like. Because obviously, if you make the bands too small, the black and white eventually, just merge into grey at a certain distance. So, we needed to make sure it was very bold pattern and the sort of allied technology was really making sure that it resembled the sea snake. So, we needed to make sure that it was biologically proportionate.
Victoria - If we move on to the camouflage suit then, can you describe what that looks like first of all?
Nathan - If you think of a usual camouflage pattern, you might see someone in army gear. You normally have slightly different shades of green. In this case, it's slightly different shades of blue that are woven together in fairly large sort of swirls of colour. This specific pattern of the camouflage has come up with in concert with a wetsuit designer. So, it obviously has to be fashionable. But the different colours used are designed to match the light environment you see underwater.
Victoria - And what do you know about the shark's vision in order to understand how this camouflage would work for their visual system?
Nathan - So, although suits to us look blue. To a shark, they'll probably actually look like shades of grey because it turns out that sharks are probably colour blind. Now, we've looked at a range of shark species from things like bull sharks to carpet sharks and we're currently working on tigers and white sharks. But we expect they're going to be all very similar, basically that they have a single type of cone in their retina which makes them cone monochromats and then probably colour blind. So, the task then becomes matching brightness against the background rather than worrying about colour.
Victoria - So, these cones, these are pigment sensitive sensors in our eyes. So, we have three and if the shark only has one, then it would not be able to discern the different shades of colour that we can see. How did you discover that?
Nathan - So, we used a couple of different techniques. The first is a photometric technique where we actually take a piece of retina out the back of the shark's eye. We lay it flat and shine a very narrow beam of light through individual photoreceptor cells at the back of the eye, scan through the spectrum and find out what parts of the spectrum they're most sensitive to, and we think that's what colour they'd be most sensitive to. Doing that, we found that by surveying the retina, there was only one type present and we confirmed that using genetic techniques which is a way of screening what's expressed in the eye in terms of a protein or gene and we could confirm there was only one cone pigment there.
Victoria - Once you have made that discovery, did you immediately jumped to being able to take advantage of what you knew of the limitations in a shark's vision in order to camouflage people and prevent them from shark attacks? How did the work progress from there?
Nathan - That kind of connection was an application that's obviously always been there, but we're actually quite coincidentally approached by a company here who was interested in the space after the spate of shark attacks in WA. They sort of put their heads together and thought, "We must be able to design some sort of wetsuit that's harder for sharks to see or that scares sharks away." They found our group here UWA and so, it was very timely, based on the discoveries that we'd made.
Victoria - How do you test a shark proof wetsuit? It sounds like it could potentially be a very terrifying test.
Nathan - Well, we have lots of PhD students who are willing to swim with these suits on (just joking!). Really, it is very hard, in actual fact for all shark deterrents to test these things. Limited testing can be done in the lab, but ultimately, you have to take the product out into the wild, try and induce sharks to come up and investigate. Then you have to see how effective it is. We're at the very early stages of helping the company to test their wetsuit and it would be really nice to validate it and see if the science really works. There's likely to be no downside of the suit, but it would be really nice to know whether it decreases your chances.
Victoria - Does that really involve putting dummies in different wet suits underwater and inducing sharks to come and investigate and see what happens?
Nathan - Well obviously, there are ethical problems in putting either a person or even just an entire wetsuit, a mannequin in the water. So, we have to do it in a slightly more controlled way which is to use a piece of the wetsuit material, wrapped around something which doesn't resemble a human because obviously, we have to be very careful that we don't want to make any association between humans and food in an area where there are sharks.
29:51 - Muscular Dystrophy Treatment
Muscular Dystrophy Treatment
with Steve Wilton, Sue Fletcher, Murdoch University
A breakthrough for the disease muscular dystrophy now, thanks to two Murdoch 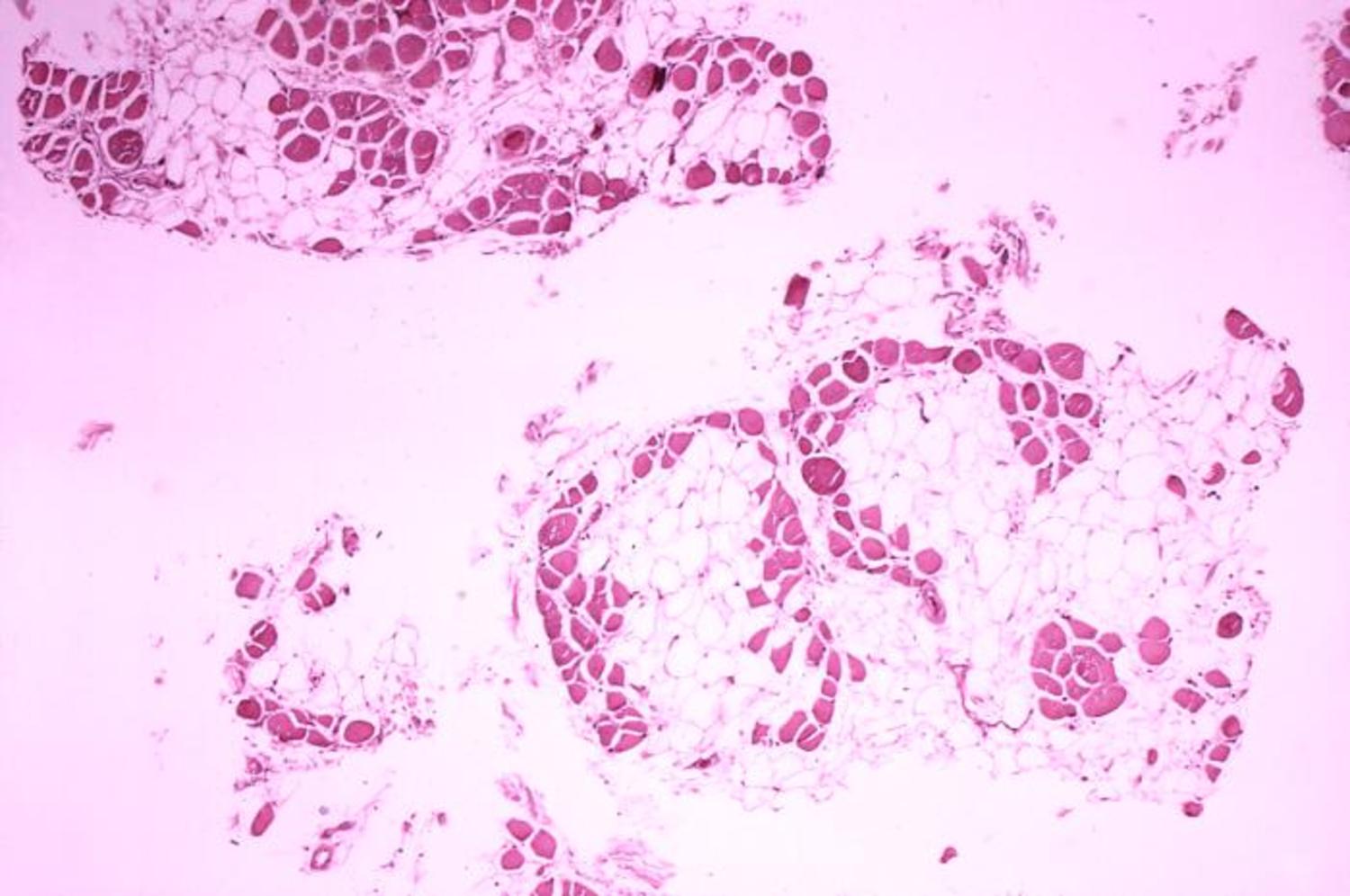 University researchers Steve Wilton and Sue Fletcher...
University researchers Steve Wilton and Sue Fletcher...
Sue - Duchenne Muscular Dystrophy is a life-limiting neuromuscular disease that results in losing the ability to walk by the age of 12 and they don't usually survive beyond their early 30's. The gene responsible for the disease is on the X chromosome, so it mainly affects boys but about 1 in 50,000 girls has the disease. Mutations in this gene prevent the synthesis of a protein called dystrophin which is a very important structural protein in muscle and that's in all muscles - heart, skeletal muscle, and in the smooth muscle of the gut.
Chris - So, when an individual has muscular dystrophy, what goes wrong in their muscles? What would a person notice about them?
Sue - Boys with muscular dystrophy do not reach the same motor milestones as other children. Typically, the boys are not walking until perhaps the age of 2. They don't climb stairs very well. They have to hold on to someone to climb stairs. They don't jump with both feet up off the ground and they don't run as other children do, they struggle to rise from the floor and that's very characteristic in this disease.
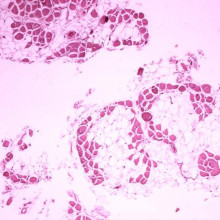
Chris - And if I were to take a little piece of muscle away and look at it under a microscope, how would it differ to a person who doesn't have muscular dystrophy?
Sue - Somebody with normal muscle, if you take a piece of muscle and you cut across the muscle, it looks like a bundle of straws. The muscle fibres are nice, regular, they're much the same size. When somebody has muscular dystrophy, the muscle is disorganised. It's full of fat and connective tissue and lots of little inflammatory cells chewing up damaged muscle. In advanced disease, it doesn't look like muscle at all.
Chris - Do we know why, not having that dystrophin protein working properly in the muscle makes that happen?
Sue - Dystrophin provides strength and stability to the muscle during muscle contraction. So, when the protein is missing, the muscle gets little tears when it's under strain, when it contracts. And gradually, inflammatory cells start to chew up the muscle because it looks like damaged tissue and the body cleans it up. So, the muscle is gradually replaced with fat and scar tissue.
Chris - Steve, what can we do about this?
Steve - One of the ways we've been trying to treat this disease is changing the expression of the defective dystrophin gene. The dystrophin gene is the largest known gene. It makes up almost 0.1% of our genetic makeup. 2.3 million letters in this one gene. It's been estimated that the enzyme that starts making the RNA copy takes 16 hours to get from one end to the other. It's like a book with 79 chapters. The first few chapters code for the attachment point at one end. The last few chapters are important at the other end. So, what happens in Duchenne Muscular Dystrophy, there's a spelling error somewhere in the chapters. And so, there's a part of chapter 16 that says, "Stop reading at this point." So, the shock absorber is made up to chapter 16 and then the rest of the message is lost. So, we have a defective shock absorber. It's not going to work. You're getting weak muscle fibres.
Chris - So, can we persuade muscle cells when they're reading this book to skip over the chapter that's got this instruction so that it would carry on through to the end?
Steve - That's is exactly what we're doing. We've designed genetic band aids that can stick at or near the defective chapter. And so that, imagine a book with the 79 chapters, it's sticking all the pages together of one chapter. So, it's chapter 15 is joined to chapter 17 rather than having chapter 16 which is the disease causing part.
Chris - Can you just explain exactly what these genetic band aids are? How do they work?
Steve - They're called anti-sense oligonucleotides. They're short genetic fragments that will stick to the dystrophin message. Now, the dystrophin message is called the sense strand and the fact that these are complimentary to the sense strand makes them anti-sense.
Chris - So, they're effectively the genetic mirror image of the gene that is in the cells that's gone wrong. So, how do they make the cell stick the chapters together?
Steve - What happens is that the cell machinery recognises parts of the gene message that have to stitch one chapter to the next. So, we're interfering with the recognition of a chapter. It essentially sort of tapes all those pages of defective chapter together.
Chris - So, when your genetic band aid is stuck on, it effectively crow bars the cell's machinery and says, "Ignore this bit and go on with the next bit."
Steve - I actually use the analogy of a spanner. We're dropping a spanner into this splicing machinery so it clogs up that part of the process and it skips over it completely.
Chris - Have you got to the stage where you've got evidence this is actually working?
Sue - Well, we've had closer than 20 years of animal experimentation and cell experimentation, showing that we can do this in a dish with cells and we can do it in mice. But a mouse is a very small animal. The longest muscle in a mouse is about a centimetre long. So, we have a lot of evidence showing that if we treat young mice, we can abrogate the onset of the disease. We can stop the disease process beginning in these mice by making the muscles and that's all the muscles, make dystrophin. So these mice, their muscle looks like normal muscle.
Chris - So, how do you get Steve's genetic band aids into the mice?
Sue - We inject them into the mice. It's very simple. We dissolve the compounds in saline, salty water, and inject them into the mice. And we've been working with a company that has taken this to human trials and they do exactly the same thing. They dissolve the anti-sense oligos in saline and they inject it intravenously over about an hour, and there are 12 boys have been treated. In the current clinical trials, there have been some previous trials. They have now been treated for over 80 weeks and they have stabilised. So, they measured the distance the boys can walk in 6 minutes and over 80 weeks, those who have been treated are essentially walking the same distance 80 weeks later. And the natural history of the disease tells us that these boys, many of them would've been expected to stop walking by now. And one of the clinicians working on the trials says that in the history of this disease, this is unprecedented.
Chris - And this requires regular therapy. They would come and see someone in the clinic say, what, every week for a top up of the genetic band aid.
Sue - At present, the boys are being treated every week. We don't know if this is the optimum treatment, but this is what clinical trials are for, to first of all prove that the drug is safe. Secondly, to prove that the drug does what it's supposed to do. In other words, preserve the muscle, restore muscle function, and ultimately, there will be additional trials to determine how often and how much of the treatment is required to keep the boys well.
36:50 - Healing Fractures with Sea Sponge
Healing Fractures with Sea Sponge
with Professor Ming Hao Zheng, University of Western Australia
Professor Ming Hao Zheng from the University of Western Australia is leading a 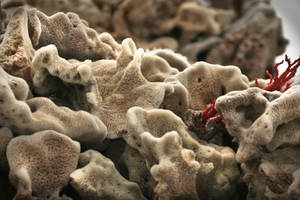 research project that began with a walk on the beach, when he kicked a dried up piece of sea sponge.
research project that began with a walk on the beach, when he kicked a dried up piece of sea sponge.
He immediately noticed how much its porous structure looked like human bone. And he's now created sea sponge implants to treat osteoporosis that are now undergoing animal trials, as he explained to Victoria Gill.
Ming - This sea sponge powder once implanted into a pouch of muscle, we can see on the inside of the muscle, they induce the sea sponge to become bone tissues. So, that tells us that these sea sponge have osteo conductivity to actually attract stem cells to form bone tissues.
Victoria - So, it's actually forming bone tissue in the animal's body that would never grow bone.
Ming - Exactly, yeah. So, that is what we can use because this scientific experiment proved that this sea sponge may be able to use in patient for the induction of bone formations.
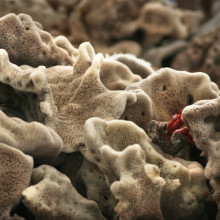
Victoria - We're looking now at a piece of the sea sponge and this is actually the same species that you kicked upon the beach in Margaret River. So, what did you notice about it that inspired your research?
Ming - The first thing I noticed about this - what I kicked away was probably a piece of animal bone that's left by dog, but after I look at it, no, this is sea sponge. So I said, "How come the sea sponge look like animals bone?" But the more I look at it, this is not animal bone. It's a sea sponge. So, I analysed them and found that the sea sponge first of all, their morphology is almost identical to the normal bone tissue and secondly, when we analysed this, the sea sponge contained collagen. We found that collagen is very conservative though evolution. So, in other words, what we're seeing of collagen in a sea sponge is probably very much identical from the collagen like we see in humans. So, as a result, this inspired me that the sea sponge may be bio-comparable, maybe about to be implanted into the human tissue.
Victoria - And it's this very porous structure that you can see these tiny little holes in it that made you think of that bone scaffold, is it?
Ming - Yeah. There are all these tiny holes and all these holes are connected to each other. It's very much like what we see in human bone tissues.
Victoria - You've brought some of the paste along. So, in this dish here, what do we have?
Ming - So, we took the sea sponge and processing in the lab and make this become a sea sponge paste. We now do this paste implant into an animal to examine osteo conductivity. Some of the study found that this sea sponge paste can induce bone formations in the muscles.
Victoria - Osteo conductivity, so that means, causing bone to grow, inducing bone to grow.
Ming - Yeah.
Victoria - So, it looks a little bit like wallpaper paste. It's peach and it sort of has these fine granules in it. It's got a rather sort of sticky kind of claggy consistency. What exactly is in there?
Ming - Well, what we do is that we process this. The key is to actually remove all the DNA in the cells from the sea sponge and then we just preserve the collagen component and some of the mineral components. And then we mix with some of the chemicals to actually make a paste-like structure that we can make a different shape according to the shape of the bone and then do the implant.
Victoria - The implant could be shaped for the fracture, for the damage, for the specific bone that you're using it for.
Ming - Yeah. Sometimes we can put it in as a strip or in some solid bone or we can make it injectable.
Victoria - So, from a simple walk on the beach to this, which could be used in human clinical trials within - how long do you think before this comes into the clinic?
Ming - At the moment, the key is that we have to finalise a so-called agriculture good manufacture practice to ensure that the starting material of sea sponge is not sea sponge they can pick up from the beach. It's actually a sea sponge that goes through a quality control production and then we can use in the patient and this is why it's holding me for so long, but I hope in the next 2 or so years. With potential partner, we're able to translate this idea into patient use.
Victoria - So, you'll get your steady supply of sea sponge and then hopefully, get this into the clinic, and it will be able to help heal some very problematic fractures.
Ming - Absolutely.
42:20 - Cells Between Life and Death
Cells Between Life and Death
with Garth Maker, Ian Mullaney, Murdoch University
What do you get if you mix a pharmacologist - a drug specialist - with a  chemist? Well if you are Murdoch scientists Garth Maker and Ian Mullaney then the answer is a totally new way of profiling how cells respond to toxic - or therapeutic - substances.
chemist? Well if you are Murdoch scientists Garth Maker and Ian Mullaney then the answer is a totally new way of profiling how cells respond to toxic - or therapeutic - substances.
They've been profiling all of the chemicals made by cells - both when they are healthy and when they are injured - in order to develop a way to find new drugs that can reverse the damage done by a stroke. But their system has also enabled them to look at pesticide chemicals we used to think were safe...
Ian - Well, one of the problems that's been interesting me for a long time is, what happens when cells get injured. It's clear that when cells get injured either by some sort of mechanical trauma or chemical, or rather exogenous trauma, the cells won't immediately die. They're go into a period of being sick and then into a protective mode. In stroke for example, the stroke infarct, the area of dead tissue, a round about that becomes an area called the penumbra which are cells which are susceptible to damage. What we think there is there's a window of opportunity for example to actually protect those cells.
Chris - So, there is this period after a cell is injured when it maybe destined to die if there's no intervention, but not necessarily. And if you can work out what's going on when it goes into this mode, upstream of dying, you may be able to turn the tables and reverse the death process.
Ian - That's right. What seems to happen is that the cell starts to release compounds when they've been compromised. One of the major ones that they releases is a compound called glutamate and glutamate is ultimately a cell killer. But it seems that in these early stages, the glutamate is actually acting as a protector.
Chris - Garth, you're trying to do this by looking at what chemicals are in cells and come out of cells during these injury processes and the recovery processes. How are you actually doing that?
Garth - The basic principle of metabolomic analysis is that we want to profile as many of these small molecule metabolites - things like amino acids, sugars, fatty acids in a single sample as we can. So, we take the cells, we extract the metabolites, and we then use a combination of chromatography and mass spectrometry techniques in order to profile not only identifying which compounds are present in the sample. But also, how the levels change from one sample to another, which we can then use to actually see which changes are occurring in a specific sample that are due to the treatment or due to the toxic event.
Chris - So, Ian hands you some cells which he has made sick and you can then put the cells through your analytical process and come up with a profile. Not just at that moment in time, but I presume you could look at different points in time to work out how processes biochemically are changing around these cells as they go through this injury process.
Garth - Absolutely and that's one of the key advantages of the technology is that we get these very large amounts of data that tell us exactly what's going on in a whole range of biochemical pathways at both multiple time points and also, over a range of doses as well. So, we can get a true picture of the pharmacology or the toxicology of these compounds.
Chris - So, what is this is actually showing you, Ian?
Ian - What we're able to do is to take that data and then we can look at the data produced from the healthy cells and then compare it to the data produced from the cells that have been insulted. What we then actually end up with is actually a pictorial representation of the data. It makes it very easy to see the changes then. In the graft that we've produced, we can see a little hot spot of important chemicals which give us a normal condition and then we can see a shift in the graft. By looking at the shift, that gives us an indication of the damage and then we can maybe try and reverse that damage by adding protective drugs, to see if we can take the shifted pattern back to the normal pattern.
Chris - So, it's not just a question of saying, "Can we look for drugs that will stop this happening?" But you could also ask, "Why do some drugs cause damage? Or even, do they cause damage?" Because if you've got a new drug no one has ever seen before, you could put it into that kind of assay and see if the cells produce a metabolic profile in Garth's tests that are similar to the ones that are produced by known toxic agents. If they do, you know immediately this new drug may be harmful.
Ian - That's right. We've got quite a load of data now looking at methamphetamine and we can actually show when we treat the cells with methamphetamine, we can see these characteristic shifts from the well cell pattern to the sick cell pattern. And that could be extended to other drug of abuse and that's something that we're interested in at the moment anyway.
Chris - Obviously, if you can work out why some things cause bad things to happen, that may give you an insight into how to make good thing happen.
Ian - Yes, that's right. One of the things that again, we're interested in as actually screening compounds as neuroprotectants. What we have is a number of compounds planned to investigate. We make the cell sick. We can look at the pattern of the sick cells and then try and reverse that to make the cells healthy again.
Chris - Sounds so obvious. Now we're all having the sort of conversation doesn't it, to think you put a chemist like you Garth with a pharmacologist like you Ian, and you can do this. Why is no one doing this?
Garth - The technology for metabolomics is really only taken off in the last decade. I think it's the beautiful thing that we get in science which is where we have people with different skills coming together and realising that actually, we can combine these new skills and new technologies in exciting ways to look at things that we haven't been able to previously do.
Chris - And is it just drugs Ian or could we look at other things as well? People are quite worried about environmental contamination, that kind of thing.
Ian - No. Of course, we can actually look at any factor which has caused damage in these cells. So for example, we have a number of projects looking at pesticides - short term and long term exposure and we find changes in the metabolite profile of these cells which mimic normal damage that we've seen in cells.
Chris - Dare I ask, which insecticides you're seeing damage from?
Garth - So far, we've focused on ones that people will be commonly exposed to, primarily Permethrin which is found in a lot of flea treatments for household pets and also Malathion which is found primarily in head lice treatments. And we see not only do we have biochemical effects at very high concentrations as you might see during an accident, an exposure, but also chronic low level effects, and effects where we see a single one off event that then has a lasting effect within the cell over an extended period of time.
Chris - These compounds that are in common use because we regard them as safe. You're saying that in your assays, you can detect changes in cells biochemically which mirror changes caused by known very bad toxins.
Garth - And obviously, there's going to be a threshold whereby this will see some low level biochemical effects which are not in the long term toxic. At certain levels of exposure over certain periods of time, we will pass that point and we will see long term toxic exposure due to these compounds and that's something that I think we don't have enough data as to the long term effects of these compounds which are so prevalent in our environment.
Chris - So, what are the implications then for these things that are ubiquitously used in the environment - the coatings on the insides of the tin can that baked beans come out of? There must be all kinds of chemicals that this sort of level of scrutiny has never been done for.
Ian - That's absolutely, right. I think one of the problems that I think they've faced in the past is that we haven't been able to detect the changes at the low levels that these compounds are present in these products. And it's only now with the technologies, the mass spectrometry in particular that we can actually find effects caused by varied levels of exposure to these compounds.
Chris - Obviously, we've talked about brain cells a lot, but if you could probably do this for any bit of the body, couldn't you Garth?
Garth - Absolutely and our next focus is going to be on liver cells since these are the cells that are primarily going to deal with a lot of these toxic or potentially toxic compounds as they enter the body and indeed any other cell type that we are interested in, we could culture the cells and look at the effects of them using the same system, absolutely.

50:09 - Why do we dream? Why do we have nightmares?
Why do we dream? Why do we have nightmares?
Hannah - Thanks, Elisa. So, is there a biological reason for dreaming? We turn to the sleep and dream expert, Dr. Valdas Noreika based at the MRC Cognition and Brain Sciences Unit in Cambridge. He starts by debunking some sleeping myths.
Valdas - You may think that your brain switches off when you sleep, but no. In fact, the regions of your brain that process what we see and feel are just as active when we dream as when we are awake. But instead of using external stimuli such as what we see or hear is brain regions process with things we've learned and remember during the previous days. This means that we dream. But because when we dream we don't consciously choose a single memory to process, different memories can merge together in a spontaneous and rather unsystematic way. This means, we can create whole new worlds and people in our dreams.
Hannah - Thanks, Valdas. So, our dreams are the result of processing memories which can happen chaotically to integrate people, places, and times to create an incoherent dream world. Do we know why these dreams sometimes turn into nightmares though?
Valdas - As well as the memory part of your brain being active during dreaming, the motion processing part is also active including the fear processing limbic system. This might partially explain why negative emotions and feelings are much more common in dreams than in waking life and result in nightmares because what's in our dreams depends on memories of what we experience when we are awake. It's perhaps unsurprising that you've be experiencing high levels of stress in your day to day life, you're more likely to have nightmares. While there is no evidence of universal meaning of different contents of dreams, we certainly have a personal psychological meaning by bringing up traumatic experiences or perhaps by simply reminding us of old friends.
- Previous Piquant Paper
- Next Why do we dream? And what are nightmares?










Comments
Add a comment