How do magnets multiply? What keeps an aeroplane in the air? How do wild animals avoid incest? It's open season on science questions in this week's Naked Scientists. We'll find out if oil extraction leaves a cavity, can cranberry juice cut urine infection rates and what happens when two lightning bolts collide? In the news, evidence of bipedalism in an early human ancestor, how oily fish helps avoid common causes of blindness and how smartphones are taking the pain out of cardiac rehabilitation. Plus, in Kitchen Science, the unexpected physics of a flying balloon.
In this episode
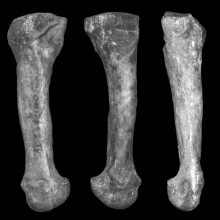
01:51 - Lucy (A. afarensis) had arched feet
Lucy (A. afarensis) had arched feet
Big news this week about the ability of Australopithecus afarensis to walk like a human. Researchers have found that this species of early hominin had rigid, arched feet. This means that afarensis would have spent a great deal of time walking only on two feet, which is just a step away from full bipedalism.
Researcher Carol Ward and colleagues from the University of Missouri have come to this conclusion over a fourth metatarsal (that's the Wayne Rooney foot bone for those that don't know). Found in Hadar, a well-dug fossil site in Ethiopia, this metatarsal is a perfect example of a bone which was lacking in the famous afarensis specimen known as Lucy.
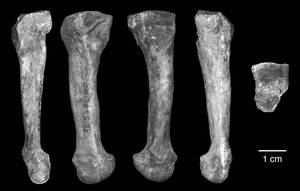 Publishing in the journal Science, the researchers think this bone points towards arched feet because its two ends are twisted in relation to each other. That means that one articular surface, where it meets the cuboid (that's one of the lumpy bones that makes up the body of the foot), sits at a different angle to the surface where it meets the phalanx (essentially in the first row of toe bones). This means that the foot was very unlikely to have been flat, and would have been quite well adapted to the 'push-off' motion that's required in walking or even running.
Publishing in the journal Science, the researchers think this bone points towards arched feet because its two ends are twisted in relation to each other. That means that one articular surface, where it meets the cuboid (that's one of the lumpy bones that makes up the body of the foot), sits at a different angle to the surface where it meets the phalanx (essentially in the first row of toe bones). This means that the foot was very unlikely to have been flat, and would have been quite well adapted to the 'push-off' motion that's required in walking or even running.
It fits in well with what we already know of afarensis' hips and spine, which do suggest quite upright walking: something which a chimpanzee can't do as efficiently. And walking on two feet between 3.7 and 2.9 million years ago should have made regular tool use possible. We don't know for certain that Lucy would have used stone tools but there was a study that came out last year in Nature, by Shannon McPherron and colleagues, which did seem to suggest that tool use was going on 3.4 million years ago. Previously it had been thought that tool use only started 2.5 million years ago, and was used by a much more human-like species of Homo than an australopithecine.

04:28 - Seeing inside the body
Seeing inside the body
A new way of seeing inside the body has been developed that uses normal light
Visible light is a great way of imaging and probing biological material, and it can be used in therapies such as photodynamic therapy where cancer patients are injected with a chemical that becomes poisonous only when exposed to light, minimising the side effects of chemotherapy.
 The problem is that despite the fact that flesh doesn't absorb that much light, it does scatter it very very strongly, which will have noticed this if you have ever shined a torch through your hand. Instead of the light going straight through your hand, it scatters repeatedly and the whole of your hand gently glows. This makes focussing light onto something deep inside your hand - say a tumor virtually impossible.
The problem is that despite the fact that flesh doesn't absorb that much light, it does scatter it very very strongly, which will have noticed this if you have ever shined a torch through your hand. Instead of the light going straight through your hand, it scatters repeatedly and the whole of your hand gently glows. This makes focussing light onto something deep inside your hand - say a tumor virtually impossible.
However if light paths are all reversible - so if you put a light bulb in the tumor, worked out how the light was leaving the body, and then sent light in the opposite direction, it would focus onto the tumor, the problem is of course that putting a light bulb in your body isn't exactly non-invasive.
Xiao Xu and collegues at Washington University have managed to get around this problem - they focus ultrasound onto the area of interest which will cause it to vibrate. They then changed the frequency of the laser light slightly, in time with the ultrasound. The light then scattered into and then out of the body, but the light which passed through the ultrasound focus will have its frequency slightly shifted by the doppler effect. They then stored the pattern of the doppler shifted light in a crystal as a hologram, and fired a much more powerful laser back through the crystal producing a reversed much more powerful version of the light which left the flesh coming from the focus of the ultrasound. So this second light pulse is focused by the flesh where you wanted it.
They have also managed to image structures in light by moving the focus of the ultrasound, which allows you to get information about the colour of structures deep inside you body non-invasively. Along with the possibilities for photodynamic therapy and imaging fluorescent markers inside the body, this could be a very useful technique.

07:21 - Omega-3 Prevents Common Forms of Blindness
Omega-3 Prevents Common Forms of Blindness
with Dr Lois Smith, Harvard Medical School and Children's Hospital
Chris - If you're a big fan, like me, of eating oily fish then you're probably doing your eye sight a big long term favour, because scientists at Harvard Medical School have discovered that the omega-3 fatty acids that are found in the fish can block the damage that's done to the retina by diseases like macular degeneration. They have also identified the gene pathway which is responsible for this protective effect and from Harvard, to tell us how it works, here's Lois Smith.
 Lois - The problem that I have been trying to solve is one of what's called retinopathy, which is abnormal blood vessel growth in the eye. This occurs in three major diseases: In premature infants, in children, there's retinopathy of prematurity. In this case, abnormal blood vessels grow and cause retinal detachment. That is, the retina, which is the light sensitive tissue in the eye, comes off the normal mooring, and children go blind. In the middle aged or working aged population, diabetic retinopathy causes the same problem. In the elderly population, there's a disease called age related macular degeneration. There's also abnormal blood vessel growth which can bleed and cause blindness. Taken together, these three diseases are a major cause of blindness.
Lois - The problem that I have been trying to solve is one of what's called retinopathy, which is abnormal blood vessel growth in the eye. This occurs in three major diseases: In premature infants, in children, there's retinopathy of prematurity. In this case, abnormal blood vessels grow and cause retinal detachment. That is, the retina, which is the light sensitive tissue in the eye, comes off the normal mooring, and children go blind. In the middle aged or working aged population, diabetic retinopathy causes the same problem. In the elderly population, there's a disease called age related macular degeneration. There's also abnormal blood vessel growth which can bleed and cause blindness. Taken together, these three diseases are a major cause of blindness.
Chris - And what are you doing in trying to understand what unites them in terms of the pathogenesis, the way these diseases occur?
Lois - There are two parts. One is, there's first loss of normal vessels, that causes oxygen starvation and then the oxygen starvation stimulates the production of chemicals that call in this pathologic or abnormal blood vessel growth. So, I've been trying to understand basic pathways that cause this disease process - both vessel loss and abnormal blood vessel proliferation.
Chris - And what's been the experimental method? What are you actually doing?
Lois - I've developed a method in mice and in this model system, we expose mice to high levels of oxygen and then bring them out into room air, and the blood vessels in the eye first disappear and then come back, roaring back in this pathologic form. Because it's in a mouse, we can do genetic manipulation and by doing that, and then subjecting them into this oxygen, we can find which gene pathways are involved in the disease process.
Chris - And presumably also, whether different treatments or interventions work...
Lois - Absolutely.
Chris - ...and in what context. In other words, which gene pathways those treatments, which we know exist but don't yet know how they work, we get an insight into how they do work.
Lois - Absolutely, that's true. In this case, I was very interested in looking at omega-6 fatty acids and omega-3 fatty acids. So the omega-6 fatty acids are the kind that are found in hamburgers basically, and the omega-3 are found in fish or a Japanese type diet. So we looked at the difference in these two diets to determine whether or not by changing the ratio of lipids that you ingest, whether or not that would have an effect on this proliferative disease - that is the production of these abnormal blood vessels.
Chris - Because if you look at the epidemiology, if you look at populations of humans, what they eat, and who gets what, people who do have a fish dominated diet tend to fair much better, and they're also at much lower risk of getting these retinopathies compared with people who are the hamburger eaters.
Lois - There's been one study in age-related macular degeneration that did suggest that and we have been working with that group at the National Eye Institute to provide the fundamental scientific basis for that process. Yes, that's true. And in the study, what we're looking for is what metabolite - that is what breakdown product really causes this beneficial effect and what are the enzymes, or what are the molecules within our body that create the breakdown product. Some of the enzymes are very, very commonly inhibited with over-the-counter drugs. There's one that's called Cyclooxygenase or COX and the COX inhibitors include aspirin and ibuprofen. So, these are drugs that many people take and we wanted to be sure that the metabolic products that cause the beneficial effect was not blocked by taking something as simple as aspirin.
 Chris - And put my mind at rest and tell me that it's not, please.
Chris - And put my mind at rest and tell me that it's not, please.
Lois - It's not. You can do both. You can take aspirin or ibuprofen and still take omega-3 fatty acids and have the beneficial effect of both.
Chris - Which is a massive relief, given the huge contribution aspirin is making to saving lives from stroke and heart disease, and maybe even preventing Alzheimer's disease as well. But if it's not impacting on those pathways, what are these fatty acids doing to prevent people getting retinopathy then?
Lois - It's going through a different pathway that's called lipoxygenase or LOX, and this pathway produces a specific metabolite called 4-HDHA which provides the beneficial effects. And interestingly enough, one of these effects is through an enzyme called PPAR-gamma which is what drugs to help improve insulin sensitivity in diabetes also activates. So the implication of this is that by taking an increased level of omega-3 fatty acids we're increasing insulin sensitivity in diabetes as well.
Chris - So now that you know this, now you know the pathway involved, does this mean that you can prevent the depletion of oily fish in the sea that we love to a certain extent by producing some kind of molecule which will be very targeted at this problem of retinopathy and can exploit the same effect?
Lois - That's exactly what we're interested in pursuing next and yes, that's our hope and expectation. Although it may still be better instead of depleting the fish in the sea to have algae make the omega-3 fatty acids that we can still take. It may be simpler to take the precursor than it is to take the metabolic product, but I think we can do it without depleting fish.
Chris - Can algae do that? How easy is it to get the algae to do what the fish do for us at the moment?
Lois - Actually, they are the original source because algae produce it and then the fish eat it, and we then eat the fish. So, it's quite simple to have them do that.
Chris - I think I'll stick with the oily fish. I think it's probably tastier than eating the algae, but fascinating. I just did not know that the essential omega-3 fatty acids come from algae, not the fish. That was Dr. Lois Smith, she's from Harvard Medical School and she published the work you've just been hearing about in the journal Science Translational Medicine this week.

14:13 - Greener method for making paint, glue, fixative...
Greener method for making paint, glue, fixative...
This week scientists from China have identified an environmentally-friendly way of making acrylic acid. Typically, acrylic acid is derived from propylene (so ultimately it comes from crude oil) and over the years it's become a very important industrial material: used in everything from paints, glues, fixing treatments and textiles. More than a billion kilos of the stuff are produced each year for industry.
 Weijie Ji and colleagues at Nanjing University have found an efficient way of making it from lactic acid, which most of us know as the acid which makes your muscles ache during exercise. But lactic acid is a better starting material than propylene, because it can readily be made by bacteria in huge vats. Reporting in ACS Catalysis, Ji and his team have cited a new catalyst which allows lactic acid to be converted into acrylic acid at low temperatures. And if you can promote this reaction at low temperatures, then it means less energy (or less expense) is required to keep the reaction going.
Weijie Ji and colleagues at Nanjing University have found an efficient way of making it from lactic acid, which most of us know as the acid which makes your muscles ache during exercise. But lactic acid is a better starting material than propylene, because it can readily be made by bacteria in huge vats. Reporting in ACS Catalysis, Ji and his team have cited a new catalyst which allows lactic acid to be converted into acrylic acid at low temperatures. And if you can promote this reaction at low temperatures, then it means less energy (or less expense) is required to keep the reaction going.
They call the catalyst an NaY zeolite, which means it's because it's a Y-type (called that because of its pore size), sodium absorbent. Cat litter is an example of a zeolite and during the two-stage process, what this one does is to dehydrate the alcohol from the lactic acid (C3H6O3) and produce acrylic acid (C3H4O2).
The zeolite in this case has pores which can hold and exchange sodium cations (Na+) in solution. Now, they actually add some alkali phosphates and reactants to the mix to get it going but what they think is happening is that the catalyst helps release a proton from the lactic acid and helps it react with the reagent, producing acrylic acid with an impressive yield of 58.4%
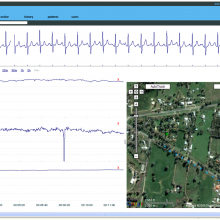
16:20 - Cardiac rehab by smartphone
Cardiac rehab by smartphone
Seeking to overcome the poor attendances at cardiac rehab classes by patients recovering from heart attacks and surgery, Brisbane-based scientists have brought modern technology to bear.
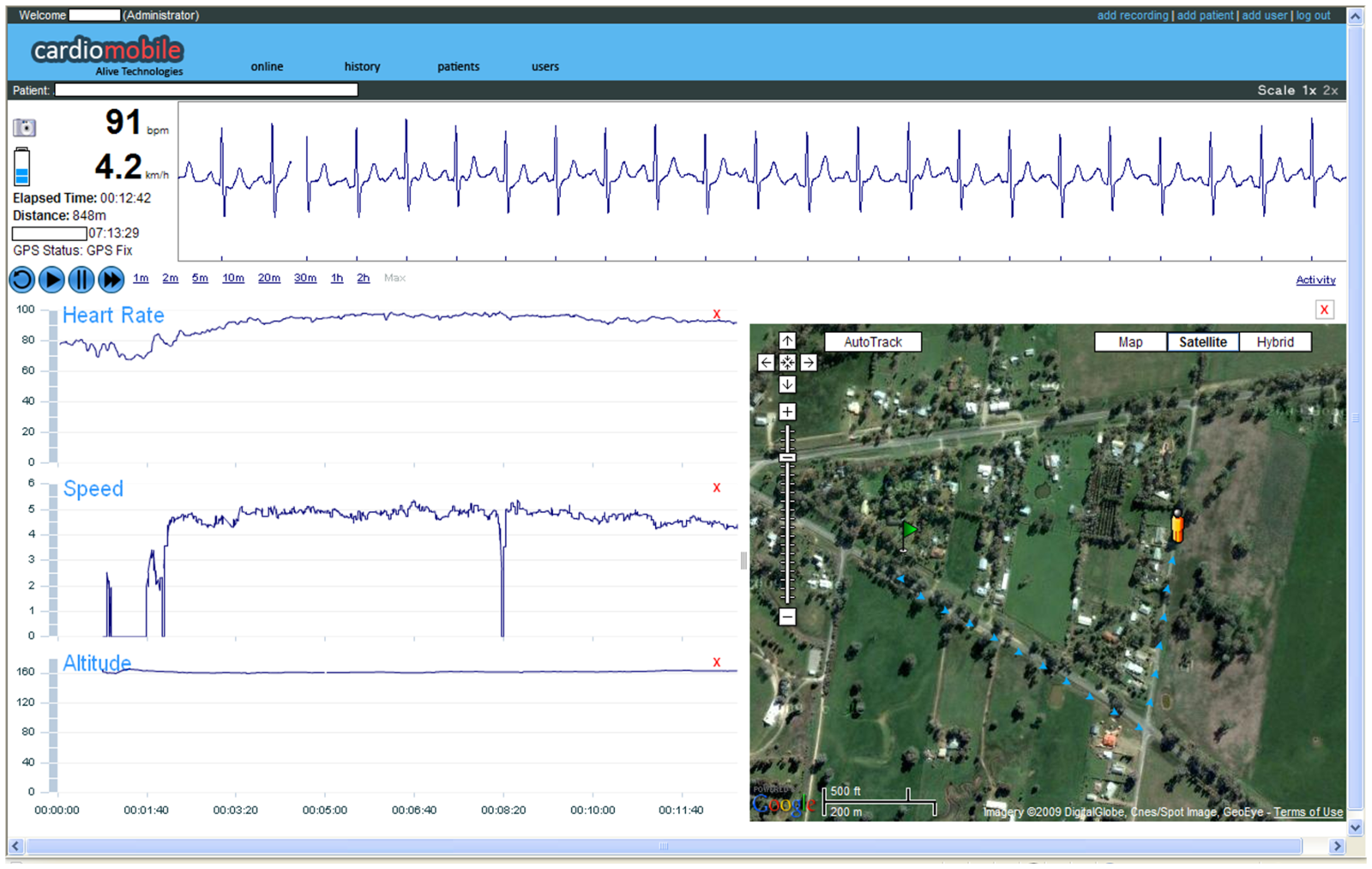 The system pioneered by Charles Worringham and his colleagues at Queensland University of Technology (QOT) and published this week in the journal PLoS One, comprises a pre-programmed smart-phone with built-in heart-monitoring software. The user simply turns on the device, attaches the monitoring pad and then commences their exercise programme, observed remotely by an exercise-trained lab support worker who follows their progress remotely, in real time, as the data is beamed back by the phone. A GPS system built into the phone is also used to record the rate at which the participant is moving.
The system pioneered by Charles Worringham and his colleagues at Queensland University of Technology (QOT) and published this week in the journal PLoS One, comprises a pre-programmed smart-phone with built-in heart-monitoring software. The user simply turns on the device, attaches the monitoring pad and then commences their exercise programme, observed remotely by an exercise-trained lab support worker who follows their progress remotely, in real time, as the data is beamed back by the phone. A GPS system built into the phone is also used to record the rate at which the participant is moving.
"The literature is stuffed full of critical publications saying that cardiac rehabilitation attendances are deplorable, but there's not one single solution being put forward," says Worringham. "So we decided to try to change things."
A programme of regular exercise and support from trained medical professionals following a heart attack is associated with significant health benefits including a reduced risk of a second event occurring. But in many countries, and especially in Australia where long travelling distances faced by rurally-based individuals can deter or prevent attendance at follow up clinics, patients often end up not benefiting from these interventions.
To find out whether their solution was practical, the QUT team initially enrolled 7 patients, including a country and western singer on tour and a carless farmer, in a six-week trial of the new gadget. They found that in all aspects, including exercise tolerance, mood and quality of life, the patients improved on-par with the results achieved by equivalent in-hospital programs. The participants also enjoyed the convenience and attraction of exercising outdoors rather than confined to an exercise bike or treadmill at an indoor gym facility.
This result could mark a big step forward for this field because even in countries with (allegedly) good transport links and a close proximity to healthcare, like Britain, only about 30% of patients actually attend cardiac rehabilitation. There therefore appears to be more than just a geographical barrier that this technology is capable of surmounting...

19:24 - Bendy superconducting cables
Bendy superconducting cables
Superconductors are materials whose electrical resistance drops to zero when they are cooled far enough, there are low temperature superconductors which normally work at below -263 celcius or 10 degrees above absolute zero, these are often conventional metals and are easy to bend but need immensely good insulation to keep them that cold, and there are high temperature superconductors which can work at 77 degrees above absolute zero, which is far easier to achieve. They would be ideal for carrying current but unfortunately but they are all brittle ceramics, similar physically to china, which makes making cables very difficult. The only way to make them at all flexible is to use very thin pieces of superconductor, making them reasonably flexible. Despite this most superconducting cables are still so brittle they have to be wound around large formers making them very very inflexible.
 Danko van der Laan has improved this significantly by using a slightly different superconductor called Gadalinium barium Copper Oxide or GBCO, whose superconducting properties are no better than other superconductors, but it degrades much more slowly when it is bent slightly. This has allowed him to wind his superconducting tapes around a copper former that is only about 6mm in diameter this along with the advantages of the GBCO means that the final cable is much more flexible. They have managed to sucessfully bend it around a diameter of 25cm whilst still being able to carry 1200A of current which is about half the current carried by a large electricity pylon. Of course to actually do this the cable would have to be electrically insulated and cooled, but it could still be far smaller, lighter and easier to install than current cables, making them practical for installing in ships and possibly even aircraft
Danko van der Laan has improved this significantly by using a slightly different superconductor called Gadalinium barium Copper Oxide or GBCO, whose superconducting properties are no better than other superconductors, but it degrades much more slowly when it is bent slightly. This has allowed him to wind his superconducting tapes around a copper former that is only about 6mm in diameter this along with the advantages of the GBCO means that the final cable is much more flexible. They have managed to sucessfully bend it around a diameter of 25cm whilst still being able to carry 1200A of current which is about half the current carried by a large electricity pylon. Of course to actually do this the cable would have to be electrically insulated and cooled, but it could still be far smaller, lighter and easier to install than current cables, making them practical for installing in ships and possibly even aircraft
Would water from 2000 ft up evaporate befoe it hits the ground?
Dave - Well to start off, I didn't have the answer in my head, so I thought I would do some science properly and actually do an experiment. So in the office last week, I got a lemonade bottle lid and a set of very accurate scales and I rigged up a fan next to it because I figured that we're interested in how fast the water is evaporating. If the drops of water from the shower head evaporate before they hit the ground, then they're never going to hit the ground, but if they get all the way down there, they will. So, I got some water in this lid, I put a fan across it. The fan probably wasn't really powerful enough, but it gives us a general idea what's going on, and I ran it for about 10 minutes. And in that time, about half a gram of water evaporated.
Chris - So that's half a ml.
Dave - Yeah, so half a millilitre of water and that was for a 28-millimetre diameter lid. If you work that all out, it comes out that you lose about 0.1 cubic millimetres of water per minute, per square millimetre of water.
Chris - So extrapolating that to Sophie's showerhead going over the cliff...
Dave - So then I've worked out the area of a raindrop, for different sized raindrops, and worked out how long it takes to evaporate. So probably, a normal to large size of raindrop is about 6 millimetres, so probably what you're getting from the showerhead. If you worked that out, it probably takes about 18 minutes for it to evaporate.
Chris - How long does a raindrop really take to fall 2,000 feet over a cliff?
Dave - Terminal velocity is about 10 metres per second, so it probably takes 70 seconds.
Chris - What fraction will evaporate then?
Dave - A tiny fraction. Maybe about an 18th of it, about 20th of it will evaporate.
Chris - But not zero. I mean, there is some evaporation.
Dave - Not zero.
Chris - Because there was a paper in Nature a couple of years ago where they were using oxygen isotopes to work out how much rainfall evaporates when rain falls from the sky, and something enormous like 40 or 50% of the rain that falls doesn't ever make it to the ground.
Dave - I could quite believe that. So I then thought I'd do some other calculations to work out what would happen if I have a smaller droplet - because, from a showerhead, you get the big droplets coming out but they also tend to break up into small ones. And around about 1-millimetre diameter raindrops falling off a 2,000-foot cliff, they'll probably just about evaporate before they hit the ground if the air was about the same as in our office, which is not necessarily a good assumption.
Chris - I'm not sure that's a good idea at all!
Dave - It would depend whether you do it in a desert or if you do it in Scotland where it's raining already. So, it very much depends, but certainly the small ones could evaporate depending on the weather.
Chris - There you go, Sophie. So if you have a very fine shower then a significant proportion could evaporate, but if you've got a really good quality power shower with big drop sizes, then a very small fraction of the water will evaporate on the way down.

Does oil extraction leave a cavity?
Chris - That's a really good question and it's one that actually, we've also had from Ady Yates who said "what do we replace pumped-out oil and gas with?"
I guess I can probably help both of you in one go here. A few years back when I first started doing all this, I thought oil existed underground in these big open caverns, almost like a coal seam - and when you take the coal away you're left with a big cave. But oil's different and, in fact, the best analogy I can think of to explain what it's really like is to imagine sticking a straw into a wet sponge and sucking water out, because that's essentially what the conditions are like underground.
When the oil was formed, it was lots of marine creatures and other organic matter; this got compacted on the sea floor below layers of sediment and the huge pressure heated everything up and cooked all these dead creatures into the soup which became the crude oil that we extract today. But that means that what that material is trapped inside is a porous rock, and I have to acknowledge on our forum, nakedscientists.com/forum, we had an answer to a similar question a couple of years ago which a geologist on the forum, JimBob, gave a very elegant answer to.
He points out that if you look at the rock that an oil well is drilling into, the porosity of that rock, in other words, the proportion of holes, is about 13%. So in other words, if you take the cross-sectional area that's holes as a whole proportion of the cross-sectional area of the piece of rock, about 13% is just empty space in the rock; and that empty space is filled with the oil. Now, he also says that that is about the same open porosity as concrete.
So, in other words, when you take the oil away, you're left with something which is equivalently strong already to concrete. So you're not leaving a big space. You're just taking the oil out from between all these little holes which are, to a certain extent, in continuity.
But then the next point to bear in mind is that this oil and gas that's underground is under extremely high pressure and that means that as you take the oil or gas away, largely under its own pressure, then other things will move in to displace it. And therefore, some water will move in from the adjacent rock and will also take up some of the space that's been vacated. In fact, when people call a well spent, in other words, they say that an oil well has become empty, actually, the amount of oil that's left behind can be as much as 90%, because the oil is very hard to get out.
To come back to Ady's question, what do we replace it with, well sometimes, you can help to get the oil out by pumping something else into the porous rock such as water to help the oil be pushed up to the surface because it floats on the water. The Norwegians have also got a technique where they pump steam in underground and the steam, being hot, can make the oil become runnier, so it's more likely to consolidate - join together - into big blobs of oil which are easier to get out.
And also, as Dave pointed out earlier when we were discussing this, sometimes they also put surfactants, things like washing up liquid, down underground and that helps the oil to have a lower surface tension so it can flow out of all these little holes more easily.
So, the bottom line is, you're sticking a straw into a sponge; the porosity of that sponge is equivalent to concrete so it's still very tough rock underground; therefore, you don't get left with a great big gaping hole, and therefore there are probably few seismic consequences as a result.
What is El Niño?
Diana - El Niño is usually defined as having occurred when you get a high air surface pressure on top of the Pacific Ocean and the actual surface of the Pacific Ocean increases in temperature. What happens as a result of this is you get the Humboldt current - that's the sort of nutrient-rich cold current of water - moving, and it moves away from the coast of South America.
That takes away all the nice happy fish, and it means that all the fishermen in South America lose a lot of their stock. And it also causes quite heavy rain and flooding in the southern parts of America. Conversely, you get droughts and sometimes even bush fires on the eastern side of the Pacific or where Australia is. This kind of thing can happen roughly every five years on average.
But as for the jet stream, we don't really know for sure, but it looks like El Niño can pull the polar jet stream slightly further south, which means that north America can suffer maybe more rainfall, maybe a bit more snow, colder winters, but we don't really know if it actually affects Europe, possibly.
Dave - Also, essentially you're changing the temperature of a great big area, a huge area, of the Pacific Ocean which will affect which area you're heating the air above, so it'll change the kind of global circulation of air currents slightly, which will have all these knock-on effects.
Diana - Exactly. It's a huge system and a hugely complex system as well and some people have even recorded maybe more droughts occurring in Africa during El Niño event, which is the other side of the world.
Does Increasing the Concentration of a Reactant Speed up an Experiment?
Dave - A reaction is normally to do with reacting two different substances together, so a type of molecule, we'll call molecule A and molecule B. If we just think about one molecule of molecule A, if you have a great big vat with only a few molecules of B in it, molecule A is going to wonder around gently throughout this huge vat, and because there's hardly any molecules of B in there, it's almost never going to meet a molecule B. So, it's very rarely going to react, so it's going to have a very, very slow rate of reaction. If you've got loads and load of molecule B, so billions and billions in there, it's hardly going to have to travel any distance at all. So the time it takes for a single molecule of A to react is going to be much, much less. So the greater concentration of B there is, the higher the rate of reaction. Similarly of course, the more of A there are, the more times this is happening at once, so the rate of reaction is going to increase. So, in general, the total rate of reaction is related to the concentration of molecule A times the concentration of molecule B.
Does cranberry juice prevent urinary tract infections?
Chris - Cranberry juice is very good for preventing and treating urinary tract infections. The reason for that is that there is a chemical in cranberry juice which down-regulates a chemical on the surfaces of cells which line the urinary tract - in other words, all the way from the kidney, right out to the outside world. It turns off, on the cells that line that pathway, a molecule which is a mannose sugar linked to some other bits of protein, which E. coli, the most common cause of urine infections, like to cling to. So, if you down-regulate the expression of that chemical on the cell surface, it gives you "anatomical Teflon". It makes you very slippery inside so the bacteria can't cling on; and as a result, they're more likely to be washed out every time we have a wee. And so, therefore, cranberry juice is very effective at reducing either the initial occurrence of a urinary tract infection, but also, it's very good at preventing it coming back and treating it as well.
How did blue eyes evolve?
Diana - I would say that actually, yes, we are all in the same family of apes. A common misconception is that people will say that we evolved from the chimpanzees or the gorillas, but of course, that's not true. We co-exist with chimpanzees and gorillas, who are also apes, but we have a common ancestor that existed several million years ago. Now, the species I spoke about was Australopithecus afarensis. That lived somewhere between 3.7 and 2.9 million years ago and almost certainly had quite different DNA to us. But, then again, humans have different DNA to each other. There's no way we can actually measure what the differences of DNA between ourselves and afarensis would've been because of course they're fossils, and there's not going to be much DNA left hanging around there, I don't think. As for the blue eye adaptation, I believe that mutated sometime around 10,000 years ago and of course, that mutation would've occurred in northern European farming populations almost certainly.
Chris - To add to this, we've also got the point that - because he makes a thing about eye colour - why don't we look at skin colour? Because chimpanzees and extant apes have pale skin, if you go back 6 million years to the ancestor we last shared with them, that also had pale skin. But the earliest generations of humans had dark skin because they were hairless and therefore needed to evolve some kind of protection against the sun. When they then leave Africa and come up to northern latitudes and low latitudes later, obviously, then they re-evolve to have paler skin because if you have that dark skin up here where the sun doesn't shine very often, you don't make enough vitamin D, and your bones suffer, and there's insufficient sunlight for the loss of folic acid, which is a reason to be dark in Africa, for that really to be a problem.
Diana - Exactly and this whole thing hinges on mutations, on people giving rise to new generations or people who were slightly different. And so, inevitably, you are going to get people with different shades of skin and different eye colours. It will happen eventually over a long enough period of time.
Chris - The evidence for that, just look at how different humans are across the whole surface of the Earth, from one geography to another; and there's the evidence that we're extremely genetically diverse, and we're all the same species. So, add a few more million years into the mix, and anything can happen.
Diana - Who knows what we'll end up looking like?!

37:15 - Celebrating William Ramsay
Celebrating William Ramsay
with Professor Alwyn Davies and Dr. Andrea Sella, University College London, and Dr. Susan Skedd, English Heritage
Chris - In London, Blue Plaques have been placed on buildings across the city for over 140 years to highlight where notable people in history have lived. This week, Meera Senthilingam went along to the unveiling of just such a plaque for a scientist whose work actually changed the field of chemistry...
Meera - I've come along to the chic area of Notting Hill in London and more specifically to number 12 Arundel Gardens, where the home of chemist Sir William Ramsay is being honoured with an English Heritage Blue Plaque. Now as the bagpipes may suggest, Ramsay was Scottish chemist but he was also a Nobel Prize winning one. He was awarded the prize for his work, finding a new group of elements to the periodic table. These are now known as the noble gases. These are a group of odourless, colourless elements that are mainly known for their lack of reactivity. And now, joining me here at number 12, to tell me a bit more about Ramsay's work is Alwyn Davies, Emeritus Professor at the University College London...
 Alwyn - Previously, in 1862, Mendeleev had classified all the elements into a very neat table and Ramsay's gases he found, he prepared, didn't fit into that table. He added a whole new group to it. So, from the fundamental chemical point of view, it was really world shattering.
Alwyn - Previously, in 1862, Mendeleev had classified all the elements into a very neat table and Ramsay's gases he found, he prepared, didn't fit into that table. He added a whole new group to it. So, from the fundamental chemical point of view, it was really world shattering.
Meera - What do we know then about Sir William Ramsay as a chemist at UCL, where he was Chair in Organic Chemistry?
Alwyn - His big characteristic was his devotion to practical work. He was a hands-on man. He wasn't a theoretician. He taught himself glass blowing. He designed and built his own apparatus, but he was prepared to tackle problems which other people would be just too scared to do. He finds two of the rare gases, helium and argon, and he guesses there's something in between. Now he said "here's a supposed inert gas and the whole world to find it in!" He does look in the whole world; He goes to Iceland, he looks in the volcanoes, he goes down to the Pyreneese, he gets meteorites and powders these, and he gets the gases from them. He looks for three years to find something that might not exist. Now that's a thing that most people would be too scared to do.
From the more practical point of view for magnetic resonance image scanning you need liquid helium, one of his gases, to get the strong magnetic field that you need. For doing much chemistry, you need an inert gas to stop the air reacting with chemicals. For that, you use argon. Argon goes into double glazing between the two panes for good insulation. Xenon ions are used for propelling spacecraft. Despite that chemical inertness, or maybe because of it, they do have many uses.
---
Meera - The Blue Plaque Scheme is currently run by English heritage and the historian that researched the case is Dr. Susan Skedd.
Susan - The Blue Plaque Scheme was started in 1866, founded by the Society of Arts. English Heritage has actually only been running it for the last 20 or so years. The scheme was inspired by the need to preserve some of the historic houses around London that were being torn down. So, to begin with, the scheme was about saving historic houses as well as commemorating the homes of notable people. It's not just about the person. It's about their formal home or workplace, and that's why placing a plaque for Ramsay is so important because it was here that he achieved his greatest works really.
Meera - So where does William Ramsay fit into all of this? What makes his home here in Notting Hill appropriate?
Susan - Well, William Ramsay was an outstanding chemist. He won the Nobel Prize for Chemistry, the first Britain to win that Prize, in 1904. He discovered not just one but five of the noble gases and really completed the periodic table of chemical elements.
---
 Meera - As well as Professor Davies, another organiser of today's event is Dr. Andreas Sella, also from University College London. Andreas, another interesting aspect of today's event is the fact that you've also managed to round up a group of cyclists and are yourself kitted out in your cycle gear upon a Brompton bicycle.
Meera - As well as Professor Davies, another organiser of today's event is Dr. Andreas Sella, also from University College London. Andreas, another interesting aspect of today's event is the fact that you've also managed to round up a group of cyclists and are yourself kitted out in your cycle gear upon a Brompton bicycle.
Andrea - Well it seems kind of crazy, what's this got to do with the noble gases? Well in 1887, Ramsay wrote to his sister, breathless with excitement, saying, "I go to college on a bicycle." And you think well, who cares? But if you think about it, the safety bicycle had only been invented a couple of years before. In fact, this is the year before Dunlop invented pneumatic tyres. So he was really on the cusp of new technology and he claimed to be able to cycle from home here in Notting Hill to college in 18 minutes. Now the streets were cobbled. He was using solid state tires. I mean, this would've been an incredible achievement.
Meera - Are you all attempting to race his 18 minutes? So today, the journey from here to UCL involves going through Oxford Circus, so lots of traffic, lots of traffic lights..?
Andrea - I think today really, we're going to be taking the scenic route and actually, we'll be pointing out along the way, other people who lived in the area. There were people like contemporaries, like Sir William Crookes, and so on. We're going to take it quite easy.
Meera - You've really drawn attention to the area of chemistry and chemists like Sir William Ramsay today, as you've managed to bring together scientists, historians, and cyclists all to one place!
Andrea - I think this really tells you about how deeply chemistry permeates our lives. In fact, modern bicycles rely hugely on chemical technology. Whether you're talking about lube, whether you're talking about brake pads, just the alloys, that make up the bicycles themselves. It's all chemistry.
Meera - Well I better let you get off as all the cyclists are waiting for you.
Andrea - Yup! Time to gather together and get the marshals...
Chris - And getting to UCL took them 40 minutes, so a little bit slower than Ramsay's time. That was University College London's Dr. Andrea Sella and before him, Dr. Susan Skedd from English Heritage, and Professor Alwyn Davies, also from University College London.
What keeps jets in the air?
Dave - I guess he means jets in a general aeroplane? Essentially, when an aeroplane moves through the air, if you have ever tried to walk from the back of the plane to the front of the plane, you'll have noticed it is always uphill. It's an awful lot more uphill than you're expecting. That's because the nose of the plane is pointed upwards, so the wings are pointing slightly downwards. This means that when air hits them underneath it bounces off and gets deflected downwards a bit, and also, due to an effect called the Coanda effect, the air on the top of the wing tends to stick to it and it also gets deflected downwards. So, the wing is pushing air downwards; Newton's laws mean that every action has an equal and opposite reaction, so the air is pushing the wing upwards and the plane stays up.
What makes mucus green?
Chris - We've all had those really nasty colds where the stuff you're coughing up - you can almost chew it because it's so thick and horrible - and it's a lovely vivid green colour. I actually had a patient who had a very bad pneumonia and I said, "Are you coughing anything up?" And she said, "Oh no" and then preceded to produce this huge blob of literally bright green stuff in front of me and she really had quite a bad pneumonia.
This is actually your body making this stuff. Mucus, in health, is colourless and it's produced by little cells called goblet cells which are in the epithelium - the layer which lines your airways - and this is rich in protein called mucin. When they secrete this substance over the surface of your airways, it attracts water from the surroundings and swells up, and becomes much more voluminous - maybe 600 times. So it swells up as it gets more water into it and it's very sticky. Its job is to trap micro-organisms, pathogens, bits of dust, and debris, that kind of thing, so it's a sort of cleaning thing.
But when you get an infection, which can be viral or bacterial, the infection can damage the cells that line the airways; this damage is intentional on the part of the pathogens. In the case of viruses, because they want to grow in the cells; in the course of growing in the cells, it kills them. In the case of the bacteria, the bacteria kill cells because if they kill a cell then all of the goodies inside the cell, the raw materials, can be liberated and the bacteria scavenge them and use them themselves.
But in the course of doing that, they create quite intense acute inflammation and this releases various inflammatory mediators which attract the immune system to the area, including a class of cell called a neutrophil. These neutrophils have something called a respiratory burst. What that means is they produce enzymes which produce free radicals of oxygen and these free radicals of oxygen destroy the bacteria. But in the process, they can also kill the white blood cell. These myeloperoxidase enzymes - that make this respiratory burst - contain iron as a cofactor, and it's the compounds of this iron, which are present in various oxidation states, that give the mucus its bright green colour.
So, healthy mucus isn't coloured, but when there's an infection going on and there are lots of white blood cells there which kill themselves with this respiratory burst, then the mucus gets this lovely vivid green colour. Yum!
How does magnetism multiply?
Dave - They will pick up some more bar magnets. If you have a single bar magnet, it's essentially a north pole and a south pole, and the force applied by those reduces with an inverse square - so if you double the distance, the force goes down to a quarter. However, because the north pole is the opposite of the south pole, if they're very, very close together, then the north pole is effectively reducing the strength of the south pole and vice versa. And so, if you get very, very close, it will be weaker. Conversely, if you make the bar magnet longer, then this actually moves the north pole away from the south pole, so they cancel each other out less. So, especially if you start off with a reasonably long bar magnet, it's not going to make very much difference to the strength of the magnetic field right next to the end of the magnet - a little bit but not very much. But it will drop away much more slowly with distance. And so, especially with the paper clips which are dangling quite a long way away, that might have a significant effect. For the ones which are close to the end of the magnet, it's going to have hardly any effect at all.

48:58 - Planet Earth Online - Dinosaurs Show their True Colours
Planet Earth Online - Dinosaurs Show their True Colours
with Professor Mike Benton, University of Bristol
Diana - Now here's a question... What colour are dinosaurs? It's an issue that puzzled palaeontologists since fossils of the creatures were first discovered. But now, thanks to modern science, dinosaurs are beginning to show their true colours.
Professor of palaeontology at the University of Bristol, Mike Benton showed Planet Earth podcast presenter Richard Hollingham a fossil of a 12-million-year-old bird.
Mike - So we have some specimens here...
Richard - So we're opening a locked cabinet, pulling out a drawer here.
Mike - So this is an astonishing little specimen....
 Richard - The sample fits in about the palm of your hand.
Richard - The sample fits in about the palm of your hand.
Mike - Yes, the palm of my hand, and this is a complete little bird. You can see that the bones are a kind of cream colour and almost chalky in appearance. These are the wing bones here, here's the back bone and the ribs there. I think here's the head, kind of bent back, almost over the top of the thing. And around it is a halo of fluffy, bluey grey material which are traces of the feathers, and you can see at the edges they become wispy. And so, we were then focusing on this dark gray material which is obviously remnants of soft tissue.
Richard - It is coloured. There are different shades of brown and gray. Can you say that they were the colours of the feather then?
Mike - No. I think that's been the question so far. There are many fossil birds and there are many fossil dinosaurs with rather obvious feathers around the edge and nobody can deny that they're feathers because in many cases, they have the detail of a wing feather with a quill up the middle and the branching barbs at the side. Others like these ones here are the sort of wispy down feathers, as we would call them, that cover the whole body area in the base of the wings and they're there mainly for insulation. But as you look at them, the differences in tone, the different shades of gray, you would be foolish to try and interpret those as original colour.
Richard - So, you got some of the samples that you put under the electron microscope. We can hear this electron microscope buzzing away behind us. If we look at the screen here, you've got one of the images when you're zooming in to the micron level here...
Mike - Yes, so this scale is 2 microns.
Richard - It's almost like a series of little rugby balls all jammed together.
Mike - Yes, they're sort of arranged higgledy piggledy, you can't see particular rows. These little structures, judging by their shape and size, we're pretty certain that these are what are called melanosomes. Melanosomes in feathers of modern birds are introduced at the very early embryonic stage of the feather as it sprouts from the skin and they come from the skin itself. So melanin is a well-known chemical that occurs throughout almost all organisms. Mostly, it gives a black colour and in the case of birds, it is produced within the skin and then it is packaged in a particular way before it goes into the feather because the keratin that makes the feather is rather like a plastic or it's quite a hard solid material. And in order to get the chemical in, it has to be packaged into these organelles.
Richard - So, a particular shape gives you a particular colour. Is that what you're looking for?
 Mike - This is what we're looking for and this is a remarkable fact that are across all modern birds and across indeed all modern mammals there are two main kinds of melanosomes. There are numerous others in between, but the two main end members are a sausage-shaped melanosome, that's about 1 micron in length, and at the other end, there's a spherical melanosome that's about half a micron across, or just slightly less. These two end members always correspond to black and ginger. So the long sausage-shaped one is black to dark brown and is called a umelanosome and the spherical one at the other end is a ginger colour, always in mammals and birds and the rest and it's called a phaeomelanosome.
Mike - This is what we're looking for and this is a remarkable fact that are across all modern birds and across indeed all modern mammals there are two main kinds of melanosomes. There are numerous others in between, but the two main end members are a sausage-shaped melanosome, that's about 1 micron in length, and at the other end, there's a spherical melanosome that's about half a micron across, or just slightly less. These two end members always correspond to black and ginger. So the long sausage-shaped one is black to dark brown and is called a umelanosome and the spherical one at the other end is a ginger colour, always in mammals and birds and the rest and it's called a phaeomelanosome.
Richard - So let's zoom back out from these samples you've got at micron level under the electron microscope to this dinosaur bird. What does it look like?
Mike - We looked at a number of different dinosaur specimens from China, but the one we were particularly keen to study in detail is called Sinosauropteryx, and it was a small turkey-sized animal with a long thin tail. But the reason we chose it was because it has the most primitive feather-like structures and we thought if we can not only determine the colour, but also determine whether these are feathers or not then we're doing something important in terms of the evolution of the group. So we had two things to do and we looked at some of the dark coloured ones and found that they were full of phaeomelanosomes, the ginger giving melanosomes. So we believe that shows two things: first of all, that these simple bristle-like filaments actually are some kind of a feather and that's important, and secondly, they have to be ginger. So we can reconstruct this dinosaur at least with a ginger in white-striped tail, like a barb or spoke.
Chris - Dianasaur or dinosaur?
Diana - Ginger power! Yes! Revealing the coloration of dinosaurs, millions of years after they died. That was Mike Benton from the University of Bristol and he was talking to Planet Earth podcast presenter, Richard Hollingham

Bashing balloons - why balloons suddenly stop.

Do wild animals avoid incest?
Diana - There are a few adaptations. One interesting one is hyenas. Apparently, female hyenas will only breed with males that have been recently born into their group or who have just recently joined it. And male hyenas will only move to new groups of females to breed. So they've got a sort of social setup which helps to avoid incest. But, on a more sort of scientific level, lemurs actually use scent. They use pheromones on their naughty bits to detect who is the most closely related to them and who's the farthest sort of relation. The females who will actually smell the males and decide "he seems perhaps a bit of a close relative. I'm not going to go there." but I think Chris, isn't there also something about mice?
Chris - Well, mice are intriguing. The explanation for mice is that the genes which are concerned with smell are found on the same part of the genome as the genes that control how the immune system decorates cells so they can recognise each other. The idea is that if mice smell alike, they probably have a very similar immune system as well. The problem with this is that if you have an immune system that's too similar, it shows you're very genetically related and so, if you have your smell system programmed to recognise someone who's very closely related to you, it means it's a bad idea to mate with them.
People have done experiments: If you take a male mouse and a female mouse, and they're brother and sister, if you force them and you put them in a caged environment where they haven't got any choice, then they will mate with each other and have offspring. But if you introduce a second mouse, so you've got the mouse's brother and then another male mouse that's unrelated, then the female mouse will preferentially mate with the one she's not related to. And if you introduce the second male mouse after the mouse is already mated and is already pregnant, she can abort the pregnancy and then mate with the new mouse. So, they are very strongly trying to avoid incest at all cost, it would appear.
Diana - Yeah, that's interesting. I think it's probably worth mentioning but sometimes it happens the other way. The ancient Egyptian royal family actually used to promote incest because they thought it was a good thing.
Chris - But were there consequences? Presumably there were.
Diana - There were, and Tutankhamun, of course, famously had a club foot as a result.

What would happen if 2 lightning bolts hit each other?
Dave Ansell answered this question...
Dave - A lightning bolt is essentially a very, very large spark!
This happens when you have a very, very large charge building up on something. It is so large that it can rip the electrons off air molecules, and then that suddenly means that air goes from being an insulator to a conductive plasma.
A plasma has free electrons moving around and therefore it conducts electricity very well. That means the path has become very, very conducting; so it kind of attracts all of the other little lightning bolts and all the other charge from around it.
All that charge runs down one single lightning bolt all the way down to the ground.
I think that lightning bolts hit each other in the sky all the time and essentially, they just pick the path of least resistance and all the current from both of them would go down the path of least resistance to the ground.
Chris - And they carry a huge current: 20,000 amps or so, I read, and it's enough to heat the air in the area of the lightning bolt to about 30,000 Kelvin, or six times the surface temperature of the Sun, which is why you get this huge expansion in the shockwave.
Dave - Which is the thunder, of course!
Where is the best place to run a marathon?
Chris - If you're running a marathon, you're presumably in a race with lots of other people and so therefore, it is, relatively speaking, a level of playing field for everybody. The difference, I would argue though, would be the altitude. If you were to go up a mountain and run, if you live up a mountain and you train regularly at altitude, then your body undergoes all kinds of adaptations in order to make you tolerate that high altitude much better; for example, you make more haemoglobin, the red stuff in your blood that carries oxygen around the body. You also have muscles that are much better able to tolerate lower oxygen tension and they make other chemicals, including one called 2,3 bisphosphoglycerate. (I know this because I've been teaching medical students at the university just recently!) This goes inside red blood cells and makes them give up oxygen from their haemoglobin more readily. So, if you went in a marathon with some people from Kenya who all live and train at high altitude, which is why they always do very well in marathons all over the world, you would be competing as someone who doesn't live at that altitude; you also don't have the same genetic ancestry that makes that beneficial for you, and therefore, you would be at a disadvantage compared with someone who does.

Can blind people, or blind-folded people, walk in a straight line?
We put this to Jan Souman, formerly of the Max Planck Institute for Biological Cybernetics in Tubingen, Germany... Jan - It is a very interesting question. I guess most people intuitively would say that blind people will be better at walking a straight line than sighted people when blindfolded; Because of course, blind people have been used to not seeing all their life and therefore probably developed some strategies of coping with that handicap. It actually turns out that blind people are not better at that than sighted people.
So people have done two kinds of studies, one kind of study is just exactly that test - just to have people walk in a straight line while either blindfolded or being blind and see how well they do and it turns out that blind people do not do better than sighted people who are blindfolded. And the other test that has been done is have people walk in a curved path by holding on to something that guides them on this curved path and then have people judge whether they're curving to the left or to the right.
Again, it turns out that blind people are not better at that than sighted people who are blindfolded. The problem for blind people is so big that sometimes it happens that when blind people try to cross a wide street or multiple lane street, that they end up at the same side of the street where they started from. So they actually walk half a circle while trying to cross the street.
It's actually not that surprising if you think about it, that blind people are not better at walking a straight line than sighted people because the brain of people trying to walk in a straight path while blindfolded are blind only has internal information, only information that comes from the body itself, from the sense of balance, the vestibular organ, from the muscles and the tendons in the body and so on. And all those cues only gives information about the relative changes in walking directions. So with every step, it basically tells the brain whether it's still going in the same direction or veering a little bit.
Diana - So why might this be?
Jan - Because the signals are noisy signals, they're biological signals so there are some kind of noise in those signals. There will be small errors in those signals and those add up over time and therefore, you end up walking in circles because that's just the accumulation of errors over time. That works the same way for blind people as for sighted people who are blindfolded. So that might be a possible explanation of why people walk in circles when they get lost or when walking blindfolded, also why blind people are not better at it than blindfolded people.










Comments
Add a comment