Why did a Laser Make My Nuts Glow?
Can you electrocute weeds? Why do teeth go wobbly? And which cells last a lifetime? In this bumper edition of the Naked Scientists, we tackle your pressing science questions and find out how the shuttle manoeuvres in space, what makes wounds itch, whether reverse osmosis can make moonshine and if static can stick a cat to a wall. Plus, how diamonds deal death to tumours, cooperation in the elephant world and an update on the Japanese earthquake situation. We also hear how a hairy leg can help you bend water to your will, and Diana discovers why potato peelers never need sharpening!
In this episode

- Why don't potato peelers need sharpening?
Why don't potato peelers need sharpening?
We put this to Tony Atkins, Emeritus Professor of Engineering at the University of Reading... Diana - It turns out that it doesn't need sharpening, simply because it doesn't need to be sharp...
Tony - There is something called the critical crack opening displacement which means how much have you got to stretch the end of the crack before the crack will carry on propagating? If you're using a knife to get rid of the skin or the peel, then you have to wedge open the material at the end of the blade, at least as much as this property called the crack opening displacement. I was surprised to discover this magic displacement that you have to achieve for potato is actually much bigger than you would have to do for meat or cheese. Now the implication of that is, that to cut meat certainly and cheese, you really need something very sharp whereas with potato, because this property value is big anyway, you can say that, "Well, why bother to have something which is sharp?"
Diana - A potato peeler doesn't need sharpening because it still works well even when it's blunt. But there's also something about the angle of cutting which makes using a less sharp tool even easier.
Tony - What is much more interesting about cutting is the whole business of why, if you take a knife, however sharp it is, you can cut, but it is so much easier if you introduce some horizontal reciprocating motion. That turns out to be a very, very interesting problem that I've solved and it goes like this: If you're say, cutting something that requires work to do it, which is force times displacement, and you say, "Okay, well if I put a bit of work in sideways, clearly I won't require as much work pressing down."
That is true, but when you do the sums, you get a strange non-linear coupling between the forces. Meaning that the slightest horizontal movement reduces the vertical force considerably and that's why it's so noticeable. What this also goes on to is that it means that the overall forces required to cut are less - if you have this slice and push it together, and that means that you don't damage the surfaces that you cut.
That can be commercially very important if you think of buying salads or something containing melon in a supermarket. In the normal way of cutting them, you damage lots of cells adjacent to the surface that you've cut and these weep out liquid. So this shelf life is improved if you do the cutting properly.

01:45 - Earthquake Update
Earthquake Update
This was a discussion held in the evening on Sunday, 13th March...
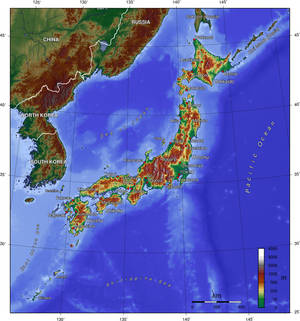
Sarah - First of all, just to recap on the situation. On the 11th of March, a massive major earthquake struck off the eastern coast of Japan. It was measured at an 8.9 magnitude and it was a mega thrust earthquake which is the type that you get where you have a subduction zone, where you have one tectonic plate being squeezed underneath another one. It hit about 130 kilometres east of Sendai which is in Honshu, which is the big main island, and the epicentre was in the Japan Trench, which is where you have the Pacific plates subducting under parts of the Eurasian plates. Although the earthquake was actually massive, what caused a lot of the damage was the 10-meter wave tsunami, so it's caused huge amounts of damage in the country.
Chris - A part of the reason why Japan suffers so many quakes is because they're not unusual there, are they? They're getting hundreds a year. It's because it's at the uniting point of three major plates.
 Sarah - Exactly. It's the meeting between the Philippine, the Pacific, and the Eurasian plates. So there's a lot of chances there for various different source of earthquakes. So the ones where you have plates sliding along each other or subducting underneath each other which is what happened in this case.
Sarah - Exactly. It's the meeting between the Philippine, the Pacific, and the Eurasian plates. So there's a lot of chances there for various different source of earthquakes. So the ones where you have plates sliding along each other or subducting underneath each other which is what happened in this case.
Chris - Dave, what's actually going on is you've got huge amounts of material moving or trying to move. It gets to a point where it can't move because you have a plate boundary and therefore, energy is getting stored up, and eventually gives and unleashes all that stored energy from many, many years of movement, but all at once.
Dave - Yeah, that's right. Essentially, it's a huge amount of elastic energy. It was an 8.9 Richter scale earthquake, that's equivalent to 300 million tons of TNT going off all at once. It's an immense amount of energy.
Chris - Is that unprecedented? Have we had one that big before?
Dave - It's certainly in our top 10 earthquakes since we've been measuring them in the last 150 years, so it's a really, really, really serious earthquake.
Chris - Could you talk us through the mechanism of how we ended up with the tsunami. What actually would have provoked that?
Dave - Fundamentally, because you've got one plate being pushed under the other one, so as the strain energy is building up, the plate is getting bent downwards, and effectively, it's pushing up the other plate and all of a sudden, it jumps and the Pacific plate jumps downwards and the Japanese plate jumps upwards. That lifts up billions and billions of tons of water by - I'm not sure what the throw was in the earthquake, but it's probably going to be a few meters. All of a sudden, you've got a huge amount of water, several meters higher that it should be. That creates a great big wave. It then moves in towards the coast. As any wave runs up the coast, it gets higher and higher, and higher. Out in the deep ocean, you don't really notice them, but as they run up the coast, it becomes higher and higher, and higher, and it will cause really big damage.
 Chris - Is that because you've got lots of waves catching up with each other or is it because the energy would be dissipated across a very deep ocean, but as it gets into shallow water, now you've got the same amount of energy, but in much less water, so you can move that water through a much greater distance to dissipate the same amount of energy?
Chris - Is that because you've got lots of waves catching up with each other or is it because the energy would be dissipated across a very deep ocean, but as it gets into shallow water, now you've got the same amount of energy, but in much less water, so you can move that water through a much greater distance to dissipate the same amount of energy?
Dave - Yes, essentially, it has a huge amount of momentum. When it's in deep water, it converts that into height and so, it gets higher and higher. Also, the really big thing is that it has a very, very long wavelength so you get a huge amount of water rather than a short wave, you've got water rushing up and then rushing down. It's like a tide coming in and out very quickly.
Chris - Which is why it's so devastating. Sarah, what about the nuclear threat? Where are we with that?
Sarah - It's Fukushima Nuclear Power Plant that are having problems at the moment, but I think the key point to make is that a lot of people are probably concerned because you see that today, there were pictures of people being tested for radiation and a small amount of people have tested positive, but I think a lot of people are concerned, could this be another Chernobyl? Could there be a really serious risk to the local population? I think in general, the answer seems to be no, just because of the type of reactor that it is. It's cooled by water. If something like this happens, you have all these control rods that get put down into the nuclear source which absorbs all the neutrons that they're releasing, and helps to stop the energy being produced, but you do still have delayed neutron precursors, and they still keep releasing neutrons for a certain amount of time after this which means you do still get heat building up, so they still need to cool the reactor after it's been shut down.
Chris - And the fact that they're having these problems is that because the cooling isn't working?
Sarah - I think they were having some problem with the water cooling, but I'm not sure what the situation with this.
Dave - I think essentially, they should have pumps pumping water around it which is keeping it cool. The system is designed so that even if the power goes off and the reactor stop producing power, it should have backup generators. I think the big problem they've had is their backup generators have failed which means it stopped pumping water around which means that it's been overheating, and causing all sorts of subsequent problems.
Chris - If the worst case scenario does occur and there is an explosion, how likely is it that there'll be widespread contamination?
Dave - There's already been an explosion high up, but that wasn't a nuclear explosion by any means. It even wouldn't get a nuclear explosion anyway. Hydrogen was being generated and that hydrogen has build up in the top of the building, and they haven't vented it properly, and therefore, the hydrogen just exploded. It's a very, very serious situation, but at the moment, the amount of radiation which is being released is not good, but it's still less than an x-ray, for the people who have been exposed certainly outside the plant itself.
Chris - And also, I think it's worth bearing in mind that Chernobyl was along while ago and the Japanese technology is way in advance of what we have today.
Dave - This is still a '70s reactor which was built in the early '70s.
Sarah - But the key point here is that the moderator isn't made of graphite which is essentially combustible, which is what the problem was in Chernobyl. It set fire because of heat whereas the moderator here is water. Obviously, it's not going to catch fire, so it's less of a problem. And also, these plants are designed - every possible thing that could go wrong, they think of that and think right, "How can we deal with that?" So they are designed to be as safe as possible.

07:45 - Diamonds - girl's best friend, cancer's worst enemy
Diamonds - girl's best friend, cancer's worst enemy
Scientists have added a sparkle to the arsenal of anti-tumour agents by successfully demonstrating that diamonds can augment the cancer-killing properties of some drugs.
Writing in Science Translational Medicine, University of California San Francisco scientist George Chow and his colleagues have demonstrated the effective, safe use in mice of tiny diamond particles dubbed "nanodiamonds". These bodies are octahedral in shape and, at 10 nanometres across, roughly equivalent in size to some small viruses. They are made by firing beams of electrons at diamond dust and can be "functionalised" by washing them in acid, which electrically charges the faces, making them sticky.
 Drug molecules can then be bound to these adhesive facets, permitting the nanodiamond to act as a carrier for the drug molecule. This makes the drug much more difficult for cancer cells to pump out, overcoming the reason why most chemotherapies currently ultimately fail, because tumours turn on efflux pumps that protect the cell from the harmful effects of the drug.
Drug molecules can then be bound to these adhesive facets, permitting the nanodiamond to act as a carrier for the drug molecule. This makes the drug much more difficult for cancer cells to pump out, overcoming the reason why most chemotherapies currently ultimately fail, because tumours turn on efflux pumps that protect the cell from the harmful effects of the drug.
The research team tested their diamond concept by binding the nanodiamonds to the anti-cancer agent doxorubicin, which kills cells by damaging DNA. Cancer cells grown in a culture dish, including cells expressing the kinds of molecular pumps that make tumours chemotherapy-resistant, were killed when the new preparation was administered. Tests also confirmed that the nanodiamond-coupled drug was retained tenfold longer in the cells compared with doxorubicin administered on its own.
The researchers also successfully tested the agents on mice with liver and breast tumours. Injected via a vein in the tail, the nanodiamond preparation gained access to and was retained within the cancerous cells where it led to significant survival enhancements compared with control animals.
Significantly, the nanodiamond formulation also showed lower rates of systemic toxicity, allowing higher overall doses of drugs to be delivered to tumours. However, this proof of principle is only the first step. Now it needs to be shown that diamonds really are a girl's (or boy's) best friend by demonstrating that the technique is equally safe and effective for humans.

11:05 - The DNA that makes us Human
The DNA that makes us Human
with Dr David Kingsley, Stanford University
Chris - Also this week, scientists at Stanford University in California have discovered how certain changes in our DNA have sculpted the evolution of human specific traits and those in particular that set us apart from our closest relatives. Dr. David Kingsley is the co-author on the work which is published this week in the journal Nature. Hello, David.
David - Hello there.
Chris - Thank you for joining us on the Naked Scientists. Could you tell us David, first of all, what was the approach? How did you try to work out genetically, how we have changed in such a way that we can do what we can do, whereas animals like chimpanzees are still hanging around in trees?
 David - One of the big breakthroughs now is that we have the complete genome sequence of both ourselves and many of our closest relatives. So the starting point for this research was to line up all the letters in the human genome with the letters in related organisms, and then look for places where the human sequence was different. That's been done before. People have looked for cases where individual letters had changed in the human sequence. This study took a different approach. We looked for block sub-sequences that were completely missing from the human genome, although present in chimpanzees, and highly conserved across many other organisms. So the fact that they're present in the highly conserved means they're likely to be functional. The fact that they're completely missing from humans suggests that they might contribute to interesting differences in our lineage compared to others.
David - One of the big breakthroughs now is that we have the complete genome sequence of both ourselves and many of our closest relatives. So the starting point for this research was to line up all the letters in the human genome with the letters in related organisms, and then look for places where the human sequence was different. That's been done before. People have looked for cases where individual letters had changed in the human sequence. This study took a different approach. We looked for block sub-sequences that were completely missing from the human genome, although present in chimpanzees, and highly conserved across many other organisms. So the fact that they're present in the highly conserved means they're likely to be functional. The fact that they're completely missing from humans suggests that they might contribute to interesting differences in our lineage compared to others.
Chris - And once you did this analysis, what genes started popping up as important and in other words, present and strongly conserved in the chimps and their relatives, but totally missing from us?
David - We found over 500 locations in the human genome where we were missing information highly conserved in other animals. One interesting feature of that list of 500 locations was that they were almost all places where, rather than changing the products of genes, you would change the regulatory information surrounding the gene. So you would alter how much they're expressed or exactly where those genes turn on, but you wouldn't destroy the product of the gene itself.
Chris - So that's the point because people have gone looking for a lot of differences previously and trying to ask what sets us apart from our closest relatives. In fact, it's not just the genes that are changing. It's the things that turn them on and off.
David - Exactly and we know that exactly that sort of change has a very important role in evolutionary differences in other organisms, so to find that sort of list in the human genome was highly motivating to try to track down the molecular basis of particular human traits.
Chris - What about the really critical ones, the things we're really interested in, things like forebrain expansion? We know that we have a very big pre-frontal cortex - the bit of the brain that does executive function and planning, compared with many of these lower animals. Did you get any insights into how that happened from your work?
David - Yeah. I think that's a great example of a really interesting human trait and it may seem paradoxical to try to explain how brains expanded by looking at a list of sequences that were missing from the human genome. But in order to look for exactly that sort of event, we searched the list for genes whose normal function is to repress or limit how much cells grow. So if you lose information from a gene whose normal function is to limit cell divisions, the loss of that regulatory information may in fact lead to expansion. We found a great example of that in the list. There's a gene called the tumour suppressor gene whose normal function is to limit cell division and sure enough, one of the lesions that had happened in the human genome was to eliminate a switch that normally causes that gene to turn on in a special growth layer of the developing brain.
Chris - Did the research also inform any of the reasons why we get certain things that we'd rather not get? In other words, does it explain why we have certain weaknesses that our close relatives don't have?
 David - Well, the list of molecular losses we think can explain both some gain traits like brain expansion, but also some loss traits in the human genome. We found some molecular switches next to a gene that controls structures that are lost in humans including sensory whiskers. We no longer have those. There's also structures in mini-organisms, mice, chimps, and other animals called penile spines and those are also missing in the human lineage, both controlled by a switch that has been deleted from the human genome.
David - Well, the list of molecular losses we think can explain both some gain traits like brain expansion, but also some loss traits in the human genome. We found some molecular switches next to a gene that controls structures that are lost in humans including sensory whiskers. We no longer have those. There's also structures in mini-organisms, mice, chimps, and other animals called penile spines and those are also missing in the human lineage, both controlled by a switch that has been deleted from the human genome.
Chris - Why don't we have those spines?
David - That's a great question. It's important to remember, this was a "How Study" not a "Why Study" so we know the molecular basis of losing those. The "Why," it might be good to lose those - there's lots of speculation about the potential functions of penile spines in other organisms. They're used to try to remove copulatory plugs that males leave in the female reproductive tracts. So if multiple males are competing for fertilisation, they can be a structure that helps ensure the fertilisation of one male compared to another. They're also sensory. They increase stimulation in males. They may increase stimulation in females, but that could either be a good thing or a bad thing. It could be painful. In fact, some people have speculated that penile spines may inflict a certain amount of tissue damage and make a female less interested in reproduction with another male.
Chris - So by losing them then, we've ended up in a situation where actually it favours us having long term relationships in a monogamous fashion.
David - That's right. So lots of organisms where the penile spines are present, multiple males are competing for a brief period of fertility with a female and the female is only interested for a short amount of time. In humans, female sexual receptivity is extended and we tend to form long term pair bonds, which is a change in social structure within the human lineage that's associated with a whole series of anatomical changes, including the loss of the penile spine.
Chris - David, we have to leave it there. Thank you. It's amazing to think how molecular genetics can then inform how we behave as a society, and how we decide how we're going to have a monogamous partner or not. David Kingsley from Stanford University. You can read the work this week in the journal Nature.

17:35 - Tiny cameras
Tiny cameras
Silicon chips have been getting smaller and smaller over the years, and this has made wiring them up more and more difficult. Most complicated chips are made on a large silicon wafer up to 18 inches across, then cut out of the wafer to produce individual chips. These are mostly wired up by making contacts on the surface and turning them over and putting them onto a connector. This is fine for most purposes but for camera chips this is a big problem, as the light then can't get to the sensors. This means that they have to cut the chips out of the large wafer, then connect wires to the edges which for a small sensor is difficult and fiddley especially for a very small chips and so expensive.
 A group from the IZM Fraunhoffer Institute has come up with a solution to this, rather than connecting to the front of the silicon wafer, they have used a small wafer, effective drilled holes through the wafer to the back, and filled them with conductor creating what is called a via. This means that they can connect the wires onto the back of the wafer before it is cut up. It also means you can stick lenses on the other side.
A group from the IZM Fraunhoffer Institute has come up with a solution to this, rather than connecting to the front of the silicon wafer, they have used a small wafer, effective drilled holes through the wafer to the back, and filled them with conductor creating what is called a via. This means that they can connect the wires onto the back of the wafer before it is cut up. It also means you can stick lenses on the other side.
Then you can just cut up the wafer to produce finished cameras. This has enabled them to make complete cameras that take up a volume of 1mm by 1mm by 1mm, with about 600 000 pixels, and they are made in a way that should be very cheap. They are suggesting using them in endoscopes and as they are so cheap they can be essentially disposable avoiding the difficult problem of disinfecting electronics before putting them in the next patient.

19:30 - Elephants show Cooperation
Elephants show Cooperation
Researchers have shown that elephants are able to cooperate just as well as chimpanzees, to retrieve a tray of food that can't be retrieved by one individual alone.
Elephants live in complex social groups, and are known to show cooperative behaviour like looking after other mothers' young.
As you might imagine, there is a lack of behavioural studies on elephants because they are so large and potentially quite dangerous. This study, which was published in PNAS by Joshua Plotnik and his colleagues, used a group of docile elephants at the Thai Elephant Conservation Centre in Lampang, Thailand.
The researchers modified a cooperation task test that has been in use studying primates since the 1930s. It involves having a tray with food on it, on the ground separated from the pair of elephants being tested by a fence. In order to bring the food into their reach, the elephants had to pull on the two ends of a rope attached to the table. If just one pulled on their end of the rope, it would pull the rope around the table and out of the other's reach. But if they both pulled on the ends of the rope together, the tray would move towards them and they could reach the food.
So the experimenters carried out three tests. After training the elephants individually to be able to pull the rope to get the tray, they tested the six pairs for cooperation. After the first test, which showed that when the two elephants were released into their roped off compartments, they would go forward, pull the rope together and get the tray, the question is 'well are they just doing that because we trained them to pull on a rope to get the tray', so they carried out further tests. The first was 'delayed release', where the elephants were released separately, and the first would have to wait for their partner to arrive before they could pull the rope. All the elephants showed a high success rate in learning to wait for their partner. The third scenario was to ensure the elephant was not just using the arrival of the second elephant as a cue to pull their rope, rather than understanding that their cooperation was what would bring the food. Here, one end of the rope was colied up by the tray of food, so there was no way to retrieve the food - if the elephants understood this, then the one with access to their end of the rope shouldn't bother pulling. And this is what happened, showing the elephants really were learning to cooperate to retrieve the reward.
One quite amusing thing they found was that some of the elephants in the group came up with novel ways to retrieve the food. One of them had to actually be excluded from the trial as she learned to stand on her end of the rope, so that when her partner pulled on their end, the tray moved towards them, but she had not had to do any work!
Do any cells last a lifetime?
Chris - There are lots of cells that you do replace on a minute-by-minute basis. There are other cells that you replace, never! In other words, they do have to last a lifetime. A good example of these are some of the brain cells. Although you can produce new brain cells during life, and that was a discovery made in the last 10 or 15 years, the vast majority of the brain cells that run your brain throughout your life, you have to make last a lifetime. One of the reasons why neurodegenerative diseases like Parkinson's and Alzheimer's disease are a problem is because once the nerve cells in certain parts of the brain die off, they're not replaced. So brain cells are a good example of a cell that lasts a lifetime.
Another one is some muscle cells. Let's take the heart as an example. Another reason why a heart attack is bad news is because when the heart is injured by a heart attack, there's an interruption of the blood flow to a territory of the heart so you will therefore lose muscle cells there, those cells in humans at least, and other high animals are not replaced. They are replaced instead by just fibrous tissue and scar tissue, so you lose physical muscle tissue, and this means the heart loses its ability to pump so well.
People used to think that fat cells were something that lasted a lifetime and that if you overfed the baby, the baby would make far too many fat cells when it was little, and this would be carried through the rest of its life and give it an increase risk of obesity. But in more recent years, there was a lady called Kirsty Spalding who's at the Karolinska Institute in Sweden, and she actually carbon dated fat cells, and found that they last about 12 years, and so, you make new ones on a roughly 12-year basis. So you make a fat cell, it will last an average of 12 years, then you can make more fat cells.
So the answer is, your body is a mixed bag. Some cells are made and they're killed off, and replaced very, very regularly, very, very rapidly, blood cells last 120 days for example, others do have to genuinely last you a lifetime.
How does the space shuttle manoeuvre?
Dave - Okay, to start off from the beginning, the two forms of rocket on the space shuttle are working very differently. The main engines on the back of the rocket are burning a mixture of oxygen and hydrogen. So we've got liquid oxygen and mix it with hydrogen in a big fuel tank. It's then piped into the engines. It's then burned, it heats up to 2,000 or 3,000 degrees centigrade, to form water. Essentially, you get very, very hot water shooting out at the back. It's very, very clean. There's almost no solids in it and water doesn't glow particularly in visible light, so the flame is almost invisible.
The solid rocket boosters on the other hand are burning a completely different fuel, they're burning aluminium and then they put an oxidiser on there as well. It's all mixed up and it's solid, and that burns, and it gets very, very hot and it produces an awful lot of thrust. It's kicking out a lot of solid particles and solid particles, when they get hot, they glow. It's a bit like if you cook, it gets very hot, it glows red hot. So these particles coming out of the bottom of the boosters are going to be glowing very brightly. They produce a lot of light and it's also coming out of much bigger holes. Everything is a bit less focused and a bit less clean.
How does the space shuttle manoeuvre? When it's in the take-off phase, so when it's going up, the main engines are gimballed. That means they can change the direction over them and so, if it's starting to go too far over the right, they kind of tip the engines around a little bit and it pushes the bottom of the rocket around. The main engines do most of the directional work of the shuttle as it goes up. They can throttle the engines a bit which would affect where it ends up, so it can do the boost, but the most control is by gimballing them.
When it gets actually up into space and it's manoeuvring just gently up there, it's dropped off all the main engines. The main engines don't work so they've got a few minor little thrusters which just act like very small rocket engines. They throw out stuff one way and it gets pushed the other way. And then for re-entering, they've got some slightly larger engines, not as big as the main ones, which can produce enough thrust to lose enough energy so they come down and hit the atmosphere and end up landing.

Can you electrocute plants?
Sarah - It's very noble of him to not want to use chemicals. I think the important point here is, yes, you could electrocute a plant, but not in the same way that you would electrocute a human.
So the reason that we would die by being electrocuted is because it will stop the heart because the heart is full of little tiny cells, called myocytes, that have their own electrical rhythm.
But if you zap them with electricity, they start just firing madly and don't beat in rhythm.
The rhythm is led by the sino-atrial node, which keeps them beating at a regular time. The whole process keeps the blood going around your body.
But if you have a load of electricity going through you, that stops happening.
Obviously, plants don't have a heart, so they can survive.
In fact, if you get a tree that's struck by lightning - this is one of the reasons that you shouldn't stand by a tree in a lightning storm - they can actually explode!
The sap inside the cells boils with the heat of the lightning and can make part of the tree blast out.
But, depending on the extent of the damage, the tree can actually survive that and you do see "scars" down the sides of trees that are still living, and then they just sort of healed over.
So you could kill the cells by frying them with electricity so that they got so hot that they died, but you'd be more likely to accidentally kill yourself, in the process, than the plants.
So I think if you're going to heat them with so much heat that they die, you might as well use a flame thrower.
Chris - Or even a garden spade and fork, better still. That might be a better idea!

Why did a blue laser make my nuts glow?
Chris - There was a paper that was published by some researchers in Athens about 7 or 8 years ago and they were looking at things like olive oil. They also looked at some other nut oils. Their motivation for looking at the fluorescent property of oils was because they wanted to try to come up with a test in order to prove that a sample of oil was authentic, because lots of people sell virgin olive oil for example, and often, it's a mixture of horrible old stuff with a few other bits and pieces to make it look nice and green, and vibrant as though it is [the real stuff].
So, they wanted to know if they were to shine light into the sample, would it interact with the light in a certain way, in order to produce a fingerprint fluorescence pattern or something that they could use as a marker. And they were really surprised to find, yes, it does, and so, I think what's going on with your glowing nuts is that when you shine the blue laser light in, there are molecules in the nuts, specifically, there'll be oil molecules which absorb the energy in the blue light, and they then re-radiate or re-emit the energy shifted towards the green end of the spectrum. That's what these people found in Athens, that these oils soak up the energy - and especially vitamin E - soak up energy in the blue end of the spectrum, and re-emits it in the green. So it soaks up energy at about 450 nanometres bluish and re-emits at about 525-530 nanometres that's green. As a result, you get this nice colour glowing coming through. So I think that's probably what you're seeing going on. Sarah...
Sarah - Does that mean that if you used a red laser, you wouldn't get the same effect?
Chris - Probably, because the molecules are sensitive to the wavelength of blue light and that's how the energy is inputted in the first place. But then after the laser is turned off, there is enough energy still in the molecule that it then, when it relaxes back to its original unexcited state, re-emits the energy at the green wavelength, and that's what happens
Why do healing wounds itch?
Chris - There are special nerves in the skin, and this has only been discovered in fairly recent times as in the last few years, but there are itch specific nerve fibres in the skin, and their job solely is to signal to the spinal cord that an area of the skin is being irritated in a way that we would perceive as itchy.
Those nerve cells are activated in a number of ways. One of them is mechanically. So, if you have an insect crawling on you for example, the insect crawling over your skin elicits the right kind of stimulus that those nerve cells are interested in, and they are triggered. So you are therefore paying attention to that bit of skin because there might be a bug which is biting you, which might be about to give you malaria or something. So that's the first point, it's mechanical.
The nerve cells are also sensitive to chemicals and there are certain chemicals which when you put them in your skin, they're irritants. As a result, they make you think, "Oh! I've got an itchy patch of skin again." You pay attention and you brush away the irritant chemical.
Now when you have a wound, the wound closes by cells around the margins of the wound, proliferating, in other words, growing and they then migrate from the margins of the wound, down into the base of the wound. They actually follow the electrical gradient, a guy in Aberdeen discovered in the last 5 years or so that the inside of the wound is at a different voltage than the margin, and the cells flow down this electrical gradient, so they know where the base of the wound is. They then unite with their cellular counterparts, and stitch themselves into place. Then they start to contract, contractile filaments which pull the wound closed.
So as they do that, they're eliciting a mechanical stress which the itch sensitive nerves will respond to and at the same time, there are various other factors which get released in a healing wound, chemicals which provoke healing in the wound, but also, upregulate the activity of these itch sensitive nerves.
So therefore, a wound that's closing up will feel itchy for mechanical and chemical reasons which are precisely the reasons why those nerve cells get stimulated in the first place. So that's the reason.

How is cosmic microwave background radiation left over from the Big Bang?
Dave Ansell answered this...
Dave - That's a really interesting question. As far as we know, the universe is effectively infinite. The Big Bang wasn't an explosion in one place in space and stuff moving out in the rest of the space. It was the fact that all of space was just a lot smaller. So, everything was a lot closer together. Because the universe was infinite, however far back you go, there'll always be some more universe further away, so the light could've travelled from somewhere just slightly further away - so it can travel through more universe and get to us.
The other thing with the cosmic microwave background radiation was that it didn't date from the very beginning of the Big Bang. It's actually light given off when electrons joined up with protons to form hydrogen atoms and joined up with helium nuclei to form helium atoms, and that released a lot of x-ray or ultraviolet light, and that happened about 380,000 years after that very, very violent beginning of the Big Bang. So by that point, the universe was actually, already quite big. We just happen to see that out 13 or 14 billion lightyears away. So, that's the edge where light is travelling towards us from. As we wait in a billion year's time, we will see that light coming from another billion lightyears further away. And so, as far as we know, if the universe is infinite, we'll keep on seeing it forever because the light will have just travelled further, so, for a longer time.
Chris - I mean the point is that, as you say, the universe was created everywhere all at once in those early days. So every bit of it is emitting radiation, and therefore, as it expands and grows, and then 13 billion years later, here we are. We are seeing light which is coming from one side of it, the opposite side to which we are. And so, as a result, there is still stuff coming our way because it was coming from everywhere all at once!
Dave - And as far as we know, it will keep on coming...

37:46 - Planet Earth Online - Water Repelling Soils
Planet Earth Online - Water Repelling Soils
with Stefan Doerr, Swansea University
Sarah - Now, if you've ever neglected a houseplant (I know I have, I'm not very good at keeping houseplants alive!) you'll know this problem; when you finally come to water it, the water doesn't soak into the soil, but it forms beads all over the surface. On a larger scale, these hydrophobic soils can cause major problems. To discover how, Planet Earth podcast presenter, Richard Hollingam has been to meet a scientist who loves the rain...
Richard - Well I really couldn't have picked a better day to investigate the effects of moisture of soil. I'm in the grounds of Swansea University with Stefan Doerr and you've got a water bottle here. Why do we need a water bottle when it's raining quite so much?
 Stefan - Yeah, that's a good point. Well, I'm just going to add a little bit more water to two different soils here on campus. One is under a conifer tree and the other one is under a deciduous tree and I'm just going to show you the effect.
Stefan - Yeah, that's a good point. Well, I'm just going to add a little bit more water to two different soils here on campus. One is under a conifer tree and the other one is under a deciduous tree and I'm just going to show you the effect.
Richard - Sweep away the leaves...
Stefan - Yup, so we've got a nice smooth soil surface here under the conifer tree, and look at that, it's actually not going in. It's beading up.
Richard - That's really weird. It's almost like mercury. As if you poured mercury on the surface, not that you're allowed to do that anymore. As if you poured mercury on the sort of the beads of water - on a soaking wet day?
Stefan - Well exactly. Everywhere else, it's soaking wet. Let's just move to somewhere else and let's put some water onto what I would say a normal soil. If we pour some water on it, we get what we normally expect to get, the water infiltrates fairly rapidly although the soil is already fairly wet from the rain we had now for probably 24 hours.
Richard - Which raises the question, why is it not soaking in, under that conifer tree?
Stefan - Well, exactly. I mean, that's what we call soil hydrophobicity or soil water repellency.
Richard - Now you've been investigating this, not outside, but actually in the lab.
Stefan - To really understand the phenomenon, we basically have to go into the lab. We have to investigate it in quite some detail, and at a range of scales as well.
...
Richard - Even inside, you can't get away from the rain. This is the rainfall simulation laboratory which consists a large steel frame, a bottle - a bucket if you like - on top, and a box which is simulating the rainfall. So it's dropping in drops through a wire mesh onto two different types of soil.
Stefan - On the right hand side, we have the wetable soil and the rain is causing little craters in the loose soil surface. It's loose material and it's soaking up very nicely. And on the left hand side, it's not forming craters, and as we've seen outside, the water is just sitting on the surface, just like mercury. We want to be able to predict when soil becomes hydrophobic, under which conditions, when does it actually switch to a hydrophobic condition? So for example when we have drought periods in the UK or elsewhere, and the soil become relatively dry, hydrophobicity tends to switch on with all these implications. As you can see now here on the rainfall simulation plot, the water is just running off the surface and that for example may be something that could've contributed to the devastating floods in 2007. There's evidence that the soils weren't particularly wet. In 2007, we had a very dry spring in April, a very, very dry period, followed by an obviously very, very wet period, but the soils didn't actually soak up the water everywhere as they should've done. So we think hydrophobicity may have well played a role.
Richard - Now you've been studying this, haven't you - at the microscopic level, well actually beyond the microscopic level.
 Stefan - Yes and what we've done in recent years, we've looked at the chemistry of those compounds that actually cause water repellency. What we do is we use a technique called atomic force microscopy. That's basically a microscopy that's not using light or x-ray. It's in a sense using a microscopic, or rather a nanoscale, little tapping device that scans across the surface of a soil particle, and that's basically at a scale you couldn't actually see with a normal microscope.
Stefan - Yes and what we've done in recent years, we've looked at the chemistry of those compounds that actually cause water repellency. What we do is we use a technique called atomic force microscopy. That's basically a microscopy that's not using light or x-ray. It's in a sense using a microscopic, or rather a nanoscale, little tapping device that scans across the surface of a soil particle, and that's basically at a scale you couldn't actually see with a normal microscope.
Richard - So you can see what type, well "see". Perhaps we shouldn't use the word 'see.'
Stefan - No, that's correct, yes.
Richard - But you can detect the individual molecules and what effects they're having.
Stefan - Yes. In a sense, you can't actually visualise the molecules themselves- that would be pushing it, but what we can see is, at the nanoscale basically, how the organic coating on the soil's surface varies. If we have a wetable soil surface, it looks quite different than on a hydrophobic surface and the physical properties at that particular level. So for example, are these compounds - do they form a smooth layer or do they actually look more like a landscape with craters and little globules of organic material, and the latter is actually the case.
Richard - Do they come from something living then?
Stefan - Yes, absolutely. I mean, it's absolutely clear that these compounds are organic, they are derived from living organisms in a sense. It could be just upgraded from plant leaves. For example, plant leaves often repel water, they're waxy. But it could also be from microorganisms like fungi or bacteria that actually produce hydrophobic compounds.
Sarah - Stefan Doerr at Swansea University, investigating the effect of hydrophobic soils.

What biological process causes flowers to wilt?
Sarah - Well there are two different things that are going on here. There's the argument of why plants wilt, so in the case if you have a house plant, and why if you don't water it, it wilts. I'll address that first.
Usually in plants, the cells within the plant are what are known as turgid, which means that they're absolutely stuffed full of water which keeps moving into them by osmosis. They're very rigid which is how plants are able to support themselves. But if they're not able to get enough water, water will move out of the cells and the cells become sort of floppy or what's known as flaccid and that is why the plant is no longer able to support itself and the leaves go all floppy and wilted and soft. So that's what happens if you don't get enough water, but there are actually some other reasons behind cut flowers wilting. So obviously, if you don't water them, the same thing will happen, but also, it can be because they run out of nutrients because obviously, they're no longer attached to roots that are getting any nutrients as well and there can be a build up of bacteria, and fungi, and things on the end of the cut surface.
But also, when you cut flowers, you cut them on your work top or whatever and then you put them in water. Because of the water tension within the xylem vessels (which are the vessels that go up and down a plant, carrying the water around), if you cut the stem, it sucks in a bubble of air into the xylem. If you then put the stem in water, it stops more water from flowing up the xylem. So that actually can be a real reason why they wilt. Some florists recommend that you cut the stems of flowers underwater which will keep the water just a little droplet on the end whilst you put them in the vase and will help them to stay alive for longer.
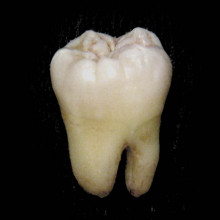
Why don't teeth fall out immediately?
Chris - I have to thank my friend Steve Jones who's a dentist at Broomfield Hospital in Essex, whom I phoned up about this one, just to make sure I had the appropriate knowledge.
He tells me that the reason teeth get wobbly when they're your deciduous teeth - your first teeth - is because the secondary dentition, the adult teeth grow in from underneath and the growth of the adult tooth erodes the root of the juvenile deciduous dentition, and that releases the tooth from the interface with the bone, making it obviously less tightly bonded in the first place.
Plus, part of that loss means that you don't have an interface between the gum - or what's called the periodontal ligament - and the connective tissue of the jaw. The teeth are connected via these tough fibrous strands, the periodontal ligaments, to the surrounding jaw tissue. As that becomes eroded away, then you've got less and less connections holding the tooth in place, so it becomes wobblier and wobblier.
I said to him, "Should you do the thread around the tooth, slamming the door trick?" He said, "No, probably not. Probably better to just let it fall out naturally." So there you go.

Can ethanol be purified by reverse osmosis?
Dave - I don't want to comment on the legality of this so let's stick to the science!
What is reverse osmosis in the first place? This is used quite often for purifying water, to get salt out of it. Essentially, you have a membrane, which will let water through but not salt, for example.
So, if you apply pressure to the side with the salt water, the water molecules are forced through the membrane but the salt can't get through. So you end up with more salty water on one side, and fresh water on the other side.
Essentially, what the questionner is attempting to do is get a membrane which will let water through but not alcohol; then apply pressure to it, and that pumps the water out leaving a stronger solution of the alcohol behind (and all the other stuff in there).
I imagine it would end up tasting very, very strong, because very few of the taste molecules are going to get through so it's going to end up with a very, very flavoursome brew.
I also think it depends on the membrane. I've looked up that some membranes are better at this than others. You'd have to pick one which is particularly good at noticing a difference between the alcohol and the water. And if you get one which works, if you pick the right membrane, there's no reason why it wouldn't work...

Why do we see blotches after looking at lights?
Sarah - Well this is very similar to the effect that you get when you're standing there at a party and someone takes a photo, and you get those spots in front of your eyes from the flashes and you just can't see anything.
It's because of something called photo-bleaching and it happens to the cells in your retina which is the light sensitive bit at the back of your eye. It's populated with cells called rods and cones, and these are full of light sensitive pigments.
Normally you get light as particles called photons coming into your eye; these stimulate the cells by changing this light sensitive pigment, which is called retinal, into a slightly different form and they stimulate the cells to send electrical impulses to your brain.
But, if you get a really bright light - like a flash or if you look at the sun for a long time or even just a really bright light bulb - this sends the cells into overdrive and they get really overexcited, and it takes them a little while to calm down. So then when you look at a normal level of light, like a wall, you get these kind of black spots where you've been looking at light where the cells have been over stimulated.
Chris - So, you have lost the photo pigment in that particular area temporarily because it's been broken down by the photons of light hitting it, and when it regenerates, it comes back then you see normal again, but while it's regenerating up the pigment because of the overexposure, you see a less intense spot, so you therefore see a dark spot. It's worth noting actually that the retina is less active when light shines on it than in the dark. It's in fact deactivated by light rather than activated by light. It's rather paradoxical, isn't it?
[Note added in proof - you should never look directly at the Sun. The lens in your eye focuses light onto the retina in the same way that a magnifying glass can focus the Sun's rays to a spot and cause things to burn. You can burn a hole in your retina leading to permanent sight loss...]

Bending water - static attraction

Why does warm water feel scalding to cold hands?
Chris - This is called adaptation and under normal circumstances, the nerve cells which are carrying sensory information from our peripheries into the core of the body, into the central nervous system, to inform the brain as to what the environment is like, they are very rapidly adapting.
What this means is that they are largely interested in how things are changing. They're not so interested in the static situation. That's because if you didn't have something that quickly got bored, and stopped signalling the absolute level of something, then you'd suffer from sensory overload. So they tend to signal change.
Now if you have got your hands very cold, then there's very little increase in the temperature in your hands and, therefore, the nerves that signal warmth are largely very inactive. And the ones that signal cold are firing off saying, "It's cold and I'm getting colder."
When you then put your hands indoors or under warmish water, the amount of firing in the warm fibres now goes through the roof because where they were previously not very responsive, they've gone from going, "I'm very, very cold so I'm not going to react" to suddenly, "Wow! The temperature is increasing very, very rapidly."
And it's actually the change that they're interested in, and so they fire intensively, and that's why you experience this sensation that things are much, much hotter than they are.
Dave - There's also a lovely experiment you can do with this, completely confusing your body. If you put one hand in a very hot water, one in a very, very cold then put them both in the middle, it's entirely confusing. One hand feels hot, one feels cold in the same water.
Chris - It's actually in our book, Crisp Packet Fireworks. We actually explained how to do that experiment if you want to have a go.
Could you stick a cat to a wall with static?
Chris - Well fundamentally, we think there's no reason why not but the amount of energy you would need would probably, at a voltage level, be lethal!
- Previous Diamonds Deal Death to Tumours
- Next Trading the Oceans










Comments
Add a comment