Iceland provides a fertile breeding-ground for genetic studies, as the population is relatively isolated making it easy to find genes involved in specific syndromes. The researchers pinned down FLAP by looking for people who had suffered heart disease or stroke. By comparing 700 heart patients with their unaffected relatives they found a region of DNA on chromosome 13 which was common to many of the patients. A further 1600 patients helped them to narrow down the region, pinpointing the FLAP gene. They found that certain versions of FLAP doubled the risk of both diseases in Icelandic populations, while another FLAP variation doubles the likelihood of a heart attack in British people. But what does FLAP do?
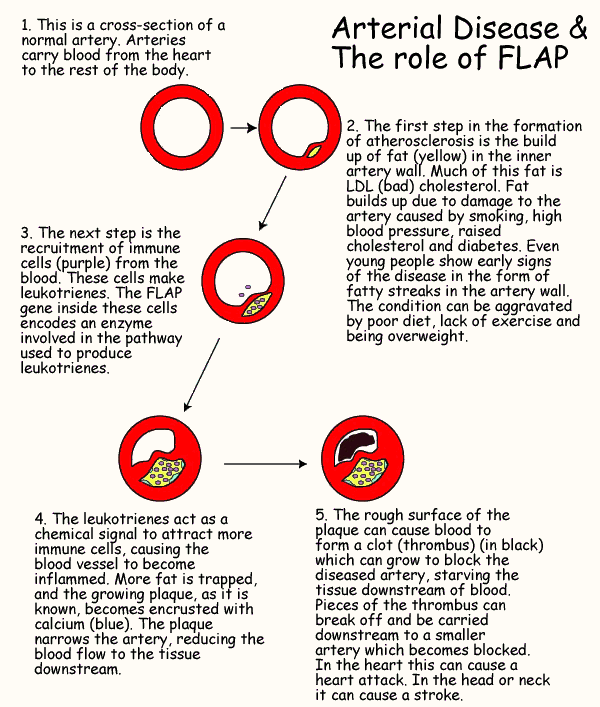
Initially, FLAP was fingered by scientists investigating asthma. They found that the FLAP gene made a protein with a role in producing leukotrienes, chemical signals secreted by cells in the immune system. When someone has an asthma attack, immune cells in the lungs produce leukotrienes which cause the airways to swell so air cannot get in and out properly. Although they seem different, asthma and heart disease are both ailments involving inflammation (swelling). Recent research has shown that the artery-clogging disease atherosclerosis involves inflammation of the blood vessels. When the wall of a blood vessel is damaged, immune cells cluster at the site of the injury and produce leukotrienes. These chemicals recruit more immune cells which help to build up the fatty deposits which are typical of atherosclerosis (see the figure below) and which progressively block the artery. These diseased regions in blood vessels are prone to developing blood clots (thrombi) which can block important blood arteries in the heart, causing a heart attack, or break loose and lodge in the brain, causing a stroke.
When the scientists investigated patients with the heart disease-prone version of FLAP, they found that their cells made more of these inflammatory leukotrienes than normal people. The researchers think that these high levels of leukotrienes could predispose people to developing atherosclerosis. Experiments by other researchers have also shown a link between high levels of leukotrienes, overactive FLAP, and atherosclerosis. The scientists in Iceland are now carrying out a clinical trial to find out whether an anti-asthma drug, that works by blocking the action of FLAP, can also have an effect on atherosclerosis.
The discovery that FLAP, and the associated leukotrienes, may play a role in the development of heart disease and strokes is very exciting. The research points towards leukotrienes as being important targets for new drugs designed to combat cardiovascular problems, either by blocking the effects of leukotrienes on arteries, or by blocking the production of these molecules. It also shows how important genetic studies will become in assessing our risk of disease, for better or for worse. Perhaps we will see the introduction of a test to identify people with a dodgy FLAP gene, which will ensure they get suitable preventative therapy. Lifestyle factors such as smoking play a big part in heart disease, so those at increased risk could be warned in advance to stay off the cigarettes, lose weight, eat healthily, or take up sport. But such genetic screening technology leaves people open to exploitation by health and life insurance companies. Would people with risky FLAP genes be made to pay higher premiums even though it is only a factor in heart disease and not a sole cause? As we discover more about the hidden secrets in our genes, and their links to disease, we will have to answer these ethical questions as well as tackling the scientific ones.










Comments
Add a comment