Getting to grips with neuropathic pain
Interview with
Neuropathic pain is a debilitating nerve condition in which patients can experience excruciating pain in the absence of any obvious tissue injury. Clearly, the pain pathways concerned must be more active than they should be, but no one actually knows why. Now, a new paper by Stephanie Ratte and Steven Prescott who are both at the University of Toronto, has shown by using drugs to tweak the activity of cultured neurons that it's not simply one single condition. There are lots of ways that nerve cells can become over-excitable. 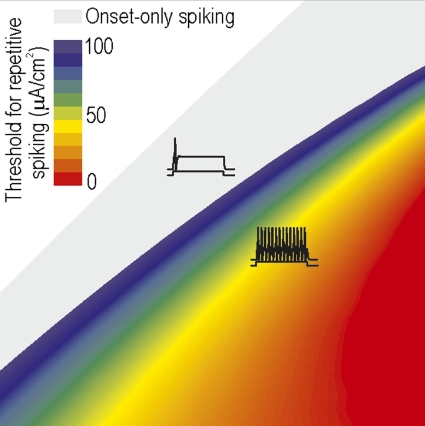 They spoke to Chris Smith...
They spoke to Chris Smith...
Steven - Neuropathic pain is pain caused by damage to a dysfunction of the nervous system. It can result from nerve injury or certain types of diseases - shingles or diabetic neuropathy. Problem is that pain signals arise abnormally from within the nervous system or that they're amplified within the nervous system rather than being accurately relayed from the skin or muscle where they normally originate. So, we were really trying to understand how that originates from changes in the nervous system in the nerve cells themselves.
Chris - So Stephanie, how have you gone about trying to make this attractable problem?
Stephanie - Excitability of nerve cells that we know changes in neuropathic pain is regulated by ion channels. These ion channels are proteins in the cell membrane that control the movement of ions in and out of the cells. So, ion channels are directly responsible for controlling degeneration of spikes and neurons. And spikes are the electrical signals that neurons use to transmit information in the nervous system. After injury, many neurons start producing more spikes than normal. We know from previous work that there's many ion channels that are upregulated or downregulated after injury. But the connection with the hyperexcitability is more of a correlation. So researchers have looked at neurons after injury and they've observed that the density of different channels in the membrane is changed. But that doesn't mean that those changes in the ion channels are actually what causes the hyperexcitability of the neuron.
Chris - So, can you disentangle that?
Stephanie - So, we decided to take a different approach than previous studies. And so, rather than triggering the changes in ion channels by injuring the neurons, we took healthy neurons and we try to reproduce those changes in ion channels in those healthy neurons. So this way, we could see what the impact of the individual ion channel changes would be under cellular excitability.
Chris - And you did that by adding various drugs in things in order to manipulate the ion channels artificially to produce that spiking activity.
Stephanie - That's right.
Chris - Do you know if that directly translated into a phenomenon that would be equivalent to neuropathic pain syndrome in a human?
Stephanie - Well, we don't know because we're looking at individual neurons. We're not looking at the entire system and we don't have a response. We don't know if it's actually causing pain or not, but what we do know is that the changes that we apply to the neuron causes or does not cause the hyperexcitability or the increase in spikes. So, that is all that we can say, but these increasing spikes has been observed in neuropathic pain.
Chris - So, when you pharmacologically manipulate these cultured nerve cells, you can end up with spiking activity that is suspiciously similar to what we see in the in vivo situation when an individual has a neuropathic pain state.
Stephanie - That's correct.
Chris - Your deduction would therefore be that the ion channel changes come first and then the pain state comes secondary to that.
Stephanie - That would be the assumption, yes.
Chris - So Steve, given what Stephanie has been saying in these observations, this therefore suggests that when we see a neuropathic pain state, actually, there could be multiple different underlying mechanisms all happening at once, triggering the observed phenomenon in any given individual patient.
Steven - Right. One way of looking at this is that there seems to some sort of tipping point in the neurons. So, we can artificially upregulate one sort of channel or artificially downregulate another sort of channel, and not necessarily have any effect on excitability until we reach some sort of threshold at which point the excitability of this neurons sort of dramatically changes. And this neuron now fires really like gang busters after we've reached that threshold. And the second finding is that there's actually many different ways of reaching that tipping point. And that has some important implications. What it tells us is that there are in fact many different individual changes sufficient, in and of themselves. There's no single change that is uniquely necessary. Ideally, from a therapeutic perspective, what we really like to do is to hit that one target, that one ion channel that is both necessary and sufficient. The thinking that we're starting to develop is that it's not so much a problem with any one ion channel, but rather, with how they are all being regulated. This really comes down to the idea of plasticity and specifically something called homeostatic plasticity. And maybe the magic bullet lies in backing up a step and asking, what has gone wrong with this regulation or with this plasticity that there may be certain molecules involved with that, that has somehow coordinately misregulating different ion channels?
Chris - So Stephanie, do you think you need to go hunting in the genome and ask what is being expressed differently in cells that are now locked into this hyperexcitable state to see if there's a genetic switch that could be thrown to reverse them back to their baseline?
Stephanie - Yes. Well, it's interesting that you would ask this because I was just talking this morning with Steve in saying that I thought we should search for collaborators who want to do this. I do think that there will be something out there to do in order to try to find this kind of upper level control that maybe is what goes wrong and starts changing all their ion channels or start resetting the set point in the abnormal state and try to control that so that we can reset the set point in a normal state.
- Previous Gene editing and repair kit
- Next Gene of the Month - Tempura










Comments
Add a comment