A technique for producing anatomically accurate replacement bones using an individual's own cells has been developed by scientists in the US.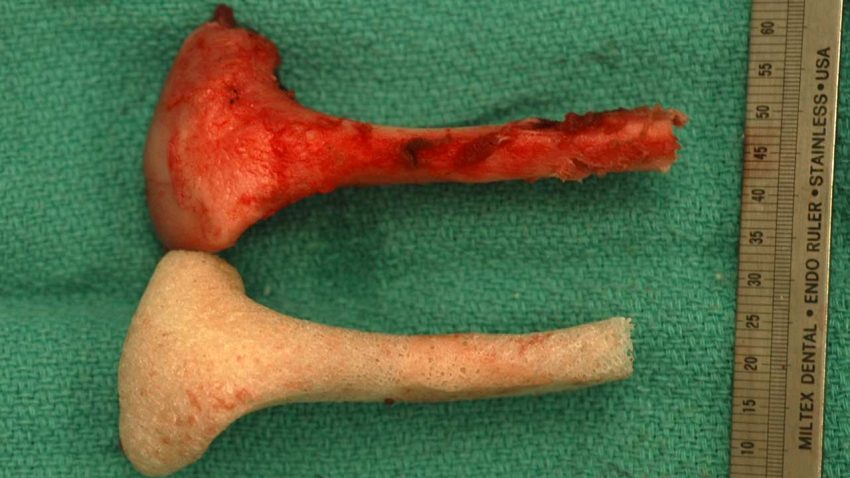
There are many instances, whether through trauma, cancers or congenital problems, where patients need new bones.
A number of techniques have been tried over the years to generate replacement bony parts. Most common is the harvesting of bone from elsewhere in the body.
Although this means patients receive their own tissue, the practice is painful and limited by the volume of bone that can be harvested and difficulty in fashioning the components into the required anatomical configurations needed to achieve a good cosmetic and functional result.
Now Columbia University's Sarindr Bhumiratana and his colleagues, writing in Science Translational Medicine, have developed a new strategy that produces replacement bone parts replete with the patient's own bone-forming cells.
Working with pigs, which are a good model for humans, the Columbia team first made CT scans of the animals' jawbones before removing a chunk of the bone and joint.
Next, a section of the thigh bone (femur) from a cow, which had been treated to remove all of the native cells, was milled using a computerised system to produce a replica of the bone defect in the pigs' jawbones.
Stem cells harvested from adipose or fat tissue collected from the animals' backs were then seeded into the bony replicas and incubated for 3 weeks in a bioreactor vessel containing nutrient growth medium.
Adipose-derived stem cells have a natural tendency to turn into bone-producing cells called osteoblasts, particularly when they find themselves in a bony environment like the de-cellularised jawbone replicas made by the team.
The stem cells swarmed all over the replicas and, once they were implanted, they remodelled the bone to stitch it almost seamlessly into place in the animals' jaws, repairing the defect introduced earlier.
Tested at six months, and compared with control animals that were given no grafts or grafts lacking any adipose-derived stem cells, the bone in the treated animals was strong and pliable with similar characteristics to a native jawbone.
A steady process of re-modelling of the bone was also taking place, progressively replacing the cow femur material with the pig equivalent.
According to the Columbia scientists, "this translational study demonstrates feasibility of facial bone reconstruction using autologous, anatomically shaped, living grafts formed in vitro, and presents a platform for personalised bone tissue engineering."










Comments
Add a comment