 It is often said that oil and water don't mix, but if you have ever done any washing up you will know that you can make them mix by adding a detergent, which allows the water to dissolve all those stubbon stains. This sort of thing is often done in chemistry, possibly to allow some catalytic nanoparticles to a liquid they wouldn't normally dissolve in. This means that they can catalyse a reaction. The problem is then retrieving your expensive nanoparticles, because once you have made them mix really well stopping them is difficult. Normally this is done by using expensive centrifuges, spinning the mixture and causing the denser nanoparticles to sink to the bottom.
It is often said that oil and water don't mix, but if you have ever done any washing up you will know that you can make them mix by adding a detergent, which allows the water to dissolve all those stubbon stains. This sort of thing is often done in chemistry, possibly to allow some catalytic nanoparticles to a liquid they wouldn't normally dissolve in. This means that they can catalyse a reaction. The problem is then retrieving your expensive nanoparticles, because once you have made them mix really well stopping them is difficult. Normally this is done by using expensive centrifuges, spinning the mixture and causing the denser nanoparticles to sink to the bottom.
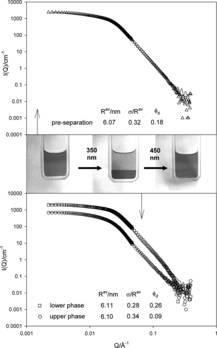
But Chemists based in the University of Bristol have developed a chemical which could do this. Normally it works like a detergent allowing droplets of water to dissolve in oil but if you shine ultra violet light onto it suddenly it ceases to be a detergent and dissolves in water again. The water droplets are now insoluble and will clump together and become easy to seperate again.
The chemical is a normal surfactant molecule which they have added
azobenzene to, which will put a kink into the molecule when exposed to ultra violet light changing its properties.
The really elegant feature is that if you illuminate it with visible light, the surfactant goes back to its original shape and properties and starts to work again, so you can recycle your nano particles again and again.










Comments
Add a comment