Ingredients
We used a large "fire piston" this is an ancient fire lighting device from SE Asia. The one we used is basically a large transparent bicycle pump, but with nowhere for the air to get out at the bottom.

Some cotton wool is teased out and then placed in the bottom of the cylinder. The plunger is then pushed down incredibly hard.

Result
When the plunger is pushed down very fast the cotton wool gets immensely hot and bursts into flames. This was used in the Burma, Malaysia Borneo region as a way of starting fires. These were made of horn or bamboo and would light a small piece of tinder. It is thought that Rudolf Diesel saw one of these and it gave him an idea...
Explanation
It takes a large amount of energy to compress the air in the cylinder. This energy is transferred to the air, the only way a gas can hold this energy is as heat, so the gas heats up. The gas inside the cylinder is compressed to about 1/20th of its original size and theoretically could be heating up to about 400°C although it is probably a little less than this.
This high temperature and the fact that the oxygen in the air is at a much higher pressure means that the cotton wool can start reacting with the oxygen in the air, and burn. The cotton wool has a large surface area so burns incredibly quickly and is ash by the time the piston is back to the beginning.
What has this got to do with Rudolf Diesel?
Rudolf Diesel invented (possibly unsurprisingly) the Diesel engine. This works similarly to a petrol engine, it mixes air and fuel in a cylinder, ignites it and uses the resulting explosion to move a vehicle along.
A diesel engine uses the same principle as the fire piston to ignite the fuel.
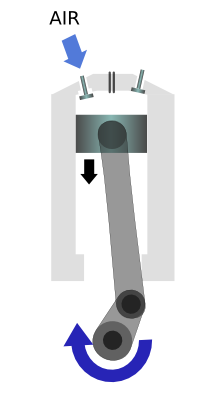 |
Air is sucked into the cylinder as the piston moves down |
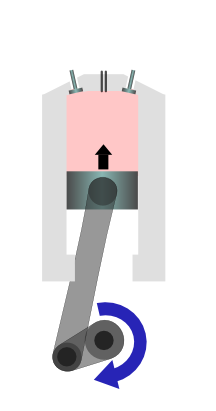 |
The air is compressed, so it gets hotter. |
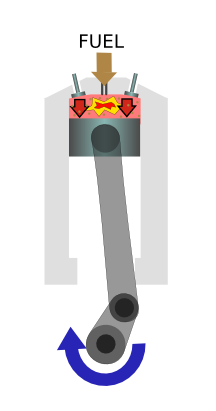 |
The compressed air gets very hot and fuel is injected, which ignites. The explosion drives the piston back down, producing power. |
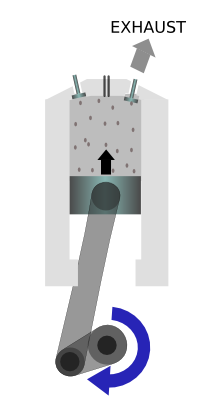 |
The exhaust gases are then pushed out of a valve. |
Why does the gas get hotter when you compress it?
Gases are made up of molecules; these are small groups of atoms. In a gas they are flying around at high speed, colliding with each other and the walls. Normally, when they collide with a surface, on average, they will leave it with the same energy as they hit it. But what happens when that wall is moving?
If the wall is moving towards the gas molecule (like when the piston is compressing the gas), it will tend to bounce off faster than it approached the piston. This is the same reason that a tennis ball will bounce off a racket faster when the racket is swung towards it.
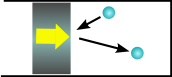
The moving piston means that the air molecules bounce off faster, and therefore have more energy, so the gas heats up.
Similarly, if the piston is moving away from the gas molecules, it will bounce off more slowly and lose energy. So a gas cools when it expands.
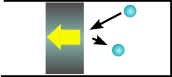
This is why an aerosol can gets cold when you spray it (the gas is expanding against the air and getting colder) and is the same principle that most fridges use to keep food cold. They compress a gas at the back of the fridge and it increases in temperature. It then loses heat in the thin pipes at the back of the fridge. The gas is then allowed to expands inside the body of the fridge, cooling it down.
- Previous Lemonade Volcano
- Next Electric Slime










Comments
Add a comment