Heat Shrink! - Why rubber bands get shorter when you heat them.
Ingredients
 | An elastic (rubber) band |  | Something to hang it from (you can use something smaller!) |
 | A hot air gun or hair drier |  | A weight (you can use something smaller!) |
Instructions
Hang your rubber band from the hook.
If you stretch a rubber band it starts off quite easily and then it gets more difficult. You want to add weight to the band to stretch it until it starts to get more difficult to stretch.
Watch the weight very carefully, you will probably have to keep your head in one place by putting it on the table and sighting the weight onto something behind.
Heat up the rubber band to see what happens.
Result
You should find that as you heat the elastic band the weight lifts up. As the band cools down again it will stretch (lengthen) again.
Explanation
Most materials expand in all directions as they heat up and unstretched rubber is no exception. But owing to its molecular structure, stretched rubber behaves very differently. Rubber is a polymer made up of long chains of repeating units. At certain points these chains are chemically attached to one another, which is known as cross-linking.
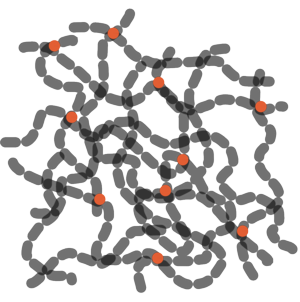 | Rubber is made up of long polymer molecules, joined together in some places. |
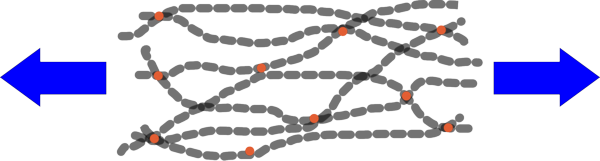 | Stretching the rubber causes the polymer chains to become straighter. |
Normally the molecules are randomly arranged and so appear very wiggley. But if you stretch the rubber it will tend to straighten out the chains. Then, as you heat up these chains they gain energy and begin to vibrate more violently, causing the chains to shorten again. You can demonstrate this for yourself with a piece of chain dangled from a hand. If you shake the chain it becomes more wiggley and therefore wider and shorter. This is most obvious if you compare the height of the bottom of the chain above the floor when it is moving compared with when it is held still.
A rubber molecule is the same so, when it is stretched, the more you heat it up, the shorter the rubber gets!
What has this got to do with crisp packets and shrink wrap?
The plastic film in crisp packets and shrink wrap has a similar structure to rubber. When packets and pots are made the material is stretched into a thin film. The big difference is that the plastic freezes below about 100°C and the polymer molecules cease to be able to move across one another. But if you heat the plastic up above about 100°C the polymer chains are freed and can shrink back to their original unstretched states. This is why crisp packets shrink if you put them on a fire, and how they shrink the plastic in shrink-wrapped goods.
- Previous Steam Powered Can Crusher
- Next Fizzes from peroxide










Comments
This helped with my project
This helped with my project however the website name got me in trouble
Add a comment