Ingredients
 | 6 or 9% Hydrogen Peroxide - you can buy it at most chemists |  | Liver |
 | A splint or thin piece of wood |  | Matches or a lighter |
 | A Jar |
Instructions
Warning - Hydrogen peroxide is a weak bleach, it will irritate the skin, and you really don't want to get it in your eyes, so wear some form of eye protection. You are dealing with burning objects which may burn quicker than you expect. This experiment should be done by an adult.
Pour about 1-2cm of hydrogen peroxide into a jar.
Add some potato, what happens?
Try the same thing with a small piece of liver.
Try lighting a splint and then blowing it out so it is still glowing, and putting it into the jar.
Result
You should find that the hydrogen peroxide sits there quietly until you add the potato when it will bubble gently. When you add the liver it should froth violently and if you carefully feel the jar, you should notice that it gets warm. The splint should relight when you put it in the jar, showing that you have made some oxygen.
Just to make it a bit more interesting we thought that we would try to use this principle to power a
lemonade bottle rocket, using the oxygen produced to pressurise the bottle. Do not try this at home, you are pressurising bleach, it could spray everywhere!
Thanks to Ian Russell for the idea.
Explanation
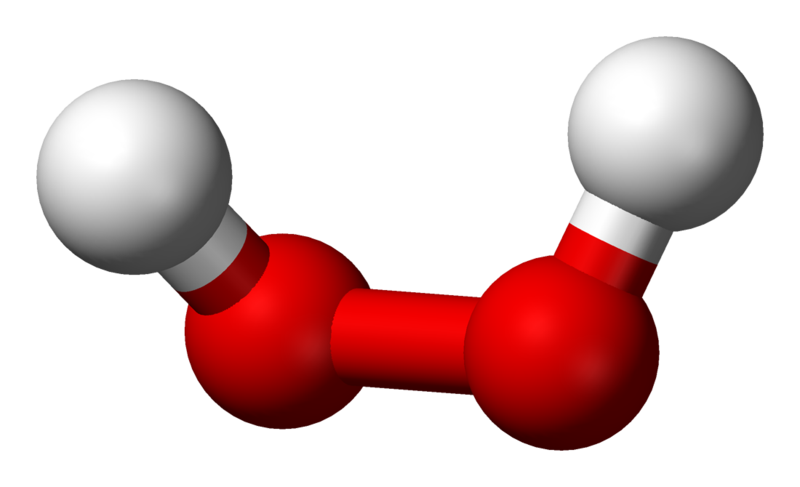 |
| Hydrogen Peroxide © Benjah-bmm2 |
Hydrogen peroxide has a molecule very like water but instead of being H2O it has an extra oxygen molecule to form H2O2. This extra oxygen is the reason that hydrogen peroxide acts as a bleach and an antiseptic. The Hydrogen peroxide can react with many different organic molecules oxidising them, which is equivalent to partially burning them. Many coloured molecules are particularly susceptible to oxidation, so they tend to be damaged first so a material will loose its colour long before it is seriously damaged.
If you add hydrogen peroxide to a living cell it will Oxidise molecules at random, causing all sorts of biochemical chaos and seriously damage or kill the cell which is why hydrogen peroxide and other bleaches are very good antiseptics. This is also why most biological cells have mechanisms to get rid of the hydrogen peroxide, which is made as a biproduct of several metabolic processes, as quickly as possible. One of the mechanisms is using an enzyme called catalase.
Hydrogen peroxide will decompose into water and oxygen in sunlight, very slowly over a period of days, so it is sold in dark brown bottles to keep the light out. However catalase is an enzyme - a biological catalyst which speeds up the reaction thousands of times without being used up itself causing oxygen to be released fast enough to form bubbles.
2H2O2 | -> | 2H2O | + | O2 |
| Hydrogen Peroxide | -> | Water | + | Oxygen |
 |
| Catalase © ProteinBoxBot @ Wikimedia |
The liver is used to remove toxins from the body so there is a particularly high concentration of catalyase there so liver will decompose hydrogen peroxide particularly quickly.
Why does the splint relight?
Burning is just the reaction of a fuel with oxygen, the more oxygen there is the faster the reaction, so the faster and hotter the fuel will burn. A glowing splint is still burning slowly, so when you put it into the oxygen the reaction speeds up so it gets hot enough to start burning with a flame again.
How do catalysts speed up the reaction?
Catalysis is a hugely complex subject and still somewhat of a black art, but one way that catalysts, particularly biological catalysts (enzymes), can work is by holding molecules in the right orientation. This helps because for a reaction between two molecules you need them to collide, with enough energy and in the right orientation. If the catalyst holds them in the correct orientation, this can hugely increase the likelyhood of them reacting.










Comments
I don't suppose you could
I don't suppose you could recommend the amount of h2o2 that would be required to propell a 1L bottle
Add a comment