Repairing Nerves and Roaring Lions
In this NewsFlash, we get the latest from the National Cancer Research Institute's conference in Liverpool, find out why removing genes can help repair neurons, and why clearing out old cells can help reduce diseases of old age. Plus, how nicotine primes the brain for cocaine addiction, and how the lion got it's roar.
In this episode

00:17 - The National Cancer Research Institute's Cancer Conference
The National Cancer Research Institute's Cancer Conference
Kat Arney offers a roundup of the latest news, and what to expect from the National Cancer Research Institute's annual cancer conference, held this year in Liverpool...
Kat - This is the National UK cancer conference, so it's really big and there's loads of stuff. There are loads of doctors, nurses, scientists, cancer patients. All sorts of people connected with cancer research are coming here today, and over the next couple of days, to talk about the latest developments, find out what's going on and set up collaborations. It's the 10th anniversary of the NCRI, the National Cancer Research Institute, and that's the umbrella organisation for all the cancer funders in the UK. So it's shaping up to be a really big and impressive conference.
 Already we've had some stories coming out of the conference. There's been some news about increasing rates of cervical cancer among young women in their 20s. Now we have a very good screening program for cervical cancer and it prevents many deaths by detecting dodgy cells before they actually become cancerous. And initially, this screening program did cause a drop in cancer rates in young women, but the rates are on the rise again. The majority of cases of cervical cancer are caused by HPV, the human papillomavirus, and this is usually a sexually transmitted infection. So scientists maybe think that a lack of safe sex in young women might be the cause. Be safe out there ladies and help to reduce your cancer risk!
Already we've had some stories coming out of the conference. There's been some news about increasing rates of cervical cancer among young women in their 20s. Now we have a very good screening program for cervical cancer and it prevents many deaths by detecting dodgy cells before they actually become cancerous. And initially, this screening program did cause a drop in cancer rates in young women, but the rates are on the rise again. The majority of cases of cervical cancer are caused by HPV, the human papillomavirus, and this is usually a sexually transmitted infection. So scientists maybe think that a lack of safe sex in young women might be the cause. Be safe out there ladies and help to reduce your cancer risk!
Also, there's a strong focus here at the conference on personalised medicine, and we'll be talking a lot more about that later in the show. This is about testing tumours for specific faulty genes or molecules, and finding out which drugs are most likely to be effective. This is really moving towards treating cancer as a molecular disease, not just a one-size-fits-all thing. So instead of saying, "Well, you have bowel cancer or you have breast cancer..." testing someone's tumour and saying "Ah! You have this, this and this faulty gene, so you need this, this and this drug."
We'll be talking later in the show to Professor Chris Marshall, who has done a lot of work over the past 30 years into cell signalling, and trying to understand how these signals go wrong and how we might target them to treat cancer. He's just done an excellent talk this afternoon all about that.
We've also got a very interesting talk going on about chemoprevention, from John Potter [Prof. John D. Potter, University of Washington, Seattle, USA and Massey University, Wellington, New Zealand]. This is the idea of preventing cancer using drugs. We've been very successful with preventing things like heart disease using statins, but much less successful with cancer. There is some evidence that a breast cancer drug called Tamoxifen, and now new drugs called aromatase inhibitors might help to prevent breast cancer in women at high risk. But there haven't been many other successes like this. So he asks "why aren't we being successful, do we need to completely rethink our ideas about how to prevent cancer?"
So it's looking like it's going to be a week packed with talks, posters, networking events and there's even art exhibitions exploring issues around cancer and how it effects people. It's shaping up to be a fantastic few days, and me and my team from Cancer Research UK are going to be covering all the top stories on our blog, so
check it out to get all the breaking news.
Ben - In some ways, it feels like we're on the cusp of a sea change in attitudes towards cancer, because of personalised medicine, because of gene therapies. It really does seem like we're in a very exciting time.
Kat - It's a really, really exciting time. If you look at when the human genome was sequenced, just a decade ago, it cost so much money - hundreds and hundreds of thousands of pounds - and it took ages. And now we can sequence three cancer genomes in a week for a cost of around one thousand dollars. So we can actually really start to analyse people's tumours, figure out what's gone wrong and work out the right drugs to treat them. So there's a lot of optimism in the field now of cancer researchers and cancer doctors.
04:05 - Remove Genes to Repair Nerves
Remove Genes to Repair Nerves
Deletion of two regulatory genes allows damaged neurons to grow back in a sustained way. This could point the way to new drug targets that encourage nerve regrowth.
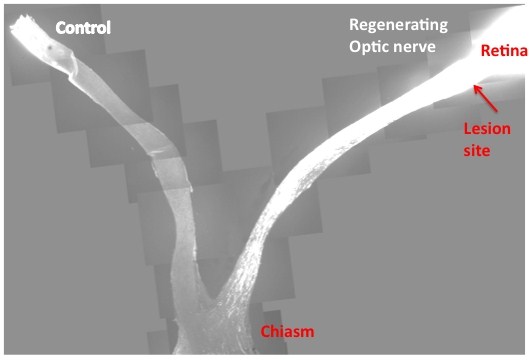 |
| This slide is the composite images from multiple sections that cover both control and injured eyes, optic nerves and the chiasm. In contrast to the control, where no regenerating axons pass the lesion site in the left, PTEN/SOCS3 double deletion allows massive optic nerve regeneration and many axons reach the chiasm. Credit: Fang Sun |
One of the major problems with repairing or re-growing damaged cells from the Central Nervous System (CNS) is the relatively huge distances the cells need to extend in order to meet their target. There are two stages of neuron growth in development: De novo outgrowth and connection in the developing embryo, followed by a long period of "networked growth", where the axons - the long slender projection that carries the electrical signal from one neuron to the next - extend, maintaining their connection at either end. As an animal grows, this second stage can result in axons far longer than anything created by the novel growth in the embryo.
If adult CNS neurons are damaged, however, the de novo methods kick in to attempt a repair. However, this can be slow and unreliable, and doesn't really offer the sustained repair and regrowth required to regain pre-injury performance.
Writing in Nature, Zhigang He from the Children's Hospital Boston and colleagues report that deleting or suppressing two regulatory genes - called PTEN and SOCS3 - enabled adult retinal ganglion cells from the optic nerves of mice to sustainably regenerate a damaged axon.
PTEN, or Phosphatase and tensin homolog, acts as a tumour suppressor, regulating cell growth and replication, while SOCS3, Suppressor of cytokine signaling 3, regulates cell signalling. If you remove either gene on its own, you see a little improvement in neuron regeneration, but deleting both genes together has a remarkable effect, a more than tenfold increase in the number of regenerating cells.
The researchers then looked at the pathways downstream of these genes, and identified a number of factors that were only activated in damaged nerves if these two genes were deleted. This suggests that PTEN and SOCS3 are suppressing growth factors and pathways that promote axon growth, and although each gene acts on different pathways, there is some synergy between them.
Understanding these factors could now provide a target for treatments that would encourage the rapid and healthy regrowth of damaged nerves and achieve functional recovery.

06:33 - Clearing Old Cells Prevents Old Age Diseases
Clearing Old Cells Prevents Old Age Diseases
Dr Jan van Deursen, Mayo Clinic
Most cell types in our body are being constantly replenished, but we still get old. A sub population of cells are said to undergo senescence - chemical controls kick in and stop them from dividing to produce new cells. When we're young, these are then cleared out by the immune system, but as we age they start to build up in our tissues.
Now, researchers at the Mayo Clinic in Rochester suggest that these cells may play an important role in diseases of old age, as Meera Senthilingam found out when she spoke to Jan van Deursen...
Jan - So we basically set out to test whether senescent cells, which are really cells that accumulate in some mammalian organisms over time were suspect of contributing to age-related disease. So we wanted to test whether that was really the case. All we did essentially was design an animal model in which we could clear these so-called senescent cells. We have two approaches - one approach was to do it lifelong, start at an early age and keep doing it for the rest of the life of the animal and then the other way was to first have the animal age so that the age related disabilities and disorders would be present and then clear the cells, and see whether there would be any beneficial effect of their clearance.
Meera - And whereabouts are these cells actually found? Are they throughout the body?
 Jan - Yeah, they're found in most tissues and organs. They are really thought of as cells that are accumulating as a result of an anti-cancer defence mechanism. So if a cell becomes damaged to a degree that it is likely to develop into a tumour cell, this process of cell senescence is activated. And basically, what it is, it makes these cells stop proliferating as a way to prevent the formation of tumours. The side effect is that you start accumulating these cells in tissues and organs, and they're not just innocent bystanders at that point. This process of senescence really changes the profile of proteins that these cells produce. They start producing and secreting inflammatory cytokines, growth factors, proteases that chew up parts of the surrounding cells. So basically, the secretory components of senescent cells make the life of neighbouring cells much more difficult. So although the senescent cells are relatively small in numbers, maybe a couple of percentage of the total number of cells in a tissue or organ, their negative effects are quite widespread.
Jan - Yeah, they're found in most tissues and organs. They are really thought of as cells that are accumulating as a result of an anti-cancer defence mechanism. So if a cell becomes damaged to a degree that it is likely to develop into a tumour cell, this process of cell senescence is activated. And basically, what it is, it makes these cells stop proliferating as a way to prevent the formation of tumours. The side effect is that you start accumulating these cells in tissues and organs, and they're not just innocent bystanders at that point. This process of senescence really changes the profile of proteins that these cells produce. They start producing and secreting inflammatory cytokines, growth factors, proteases that chew up parts of the surrounding cells. So basically, the secretory components of senescent cells make the life of neighbouring cells much more difficult. So although the senescent cells are relatively small in numbers, maybe a couple of percentage of the total number of cells in a tissue or organ, their negative effects are quite widespread.
Meera - And how did you set about removing these cells?
Jan - We took advantage of the knowledge that tumour suppressor gene p16 is really expressed in most of these senescent cells. It actually is thought to play a role in the conversion of a normal cell to a senescent cell. So we use that to make an artificial gene, kind of a suicide gene driven by that same p16 promoter. So whenever p16 is expressed, we would also express this suicide gene. But the suicide gene is present but it needs to still be activated by a drug. So, senescent cells that express this suicide gene will still not die until we would expose the cells to a synthetic drug.
Meera - And what were your key findings then with regard to the presence of these cells and their effect on particular age-related disorders and which disorders did you see an effect with?
Jan - We needed to test this experimental system and we did that on a so-called progeroid mouse model. This is basically mice that have low amounts of this protein BubR1 and they age about five times faster than a normal mouse. So now the phenotypes in this accelerated ageing mouse that display ageing related disorders are sarcopenia, which is basically muscle wasting, also the loss of fats, both fat deposits in the body and subcutaneous fat, and the subcutaneous fat loss typically gives us the wrinkled skin. The third hallmark of ageing was formation of cataracts which is seen in about 25% of people above 65. So we looked at these three aspects because we know that in those ageing processes the gene p16 was expressed, indicating that in those age-related processes, perhaps p16 positive senescence cells will play a role. We saw that lifelong clearance really can either prevent or delay those age-related disabilities. But when we treat late in life, when those disorders were already present. We could hold them or slow them down from that moment on.
Meera - What are the hoped applications of this then?
Jan - A lot of work needs to be done, but this mouse model can be used to really try to figure out what would be the best strategy of removing these cells. Would that be continuous or would that be once every or once every so often? Would it be advantageous to do it late in life? We agree that lifelong treatments are not really desirable but could also imagine that this is more like a spring cleaning that you do every so often.

12:43 - Drug Addiction, Polluted Cyclones and Roaring Lions
Drug Addiction, Polluted Cyclones and Roaring Lions
Amir Levine, Columbia University; Amato Evans, Virginia University; Elena Oancea, Brown University; Sarah Klemuk, University of Utah
Nicotine as a gateway to Cocaine Addiction
Being a smoker may increase your chances of also becoming addicted to  cocaine.
cocaine.
Working with experimental animals and also data from human subjects, University of Columbia scientist Amir Levine and his colleagues found that nicotine alters the activity of a gene called FosB, which has previously been linked to addiction. These changes increase the likelihood of developing a subsequent cocaine dependency.
Amir - We saw an increase in the different behavioural paradigms that are related to addiction. Finally, we looked at a certain gene that's called the FosB gene, that has been shown to be very important for addiction and we saw that when we give nicotine first and then cocaine, there is an enhancement in the expression of the FosB gene. And the final step is that we discovered that nicotine basically opens up chromatin and that is how it primes the brain to the effects of cocaine.
---
Cyclone Risk increased by pollution
High levels of pollution are
increasing the intensity of cyclones over Asia.  Carbon-rich brown clouds over the Arabian sea, which have grown six-fold since the 1930s, are cooling the water surface, resulting in a drop in vertical wind shear - the difference in wind speed and direction - between the upper and lower parts of the atmosphere. This increases the efficiency with which storm systems can form, making super-cyclones, with wind speeds exceeding 185 kilometres per hour, much more likely to form. Amato Evan, from the University of Virginia, led the study published this week in Nature.
Carbon-rich brown clouds over the Arabian sea, which have grown six-fold since the 1930s, are cooling the water surface, resulting in a drop in vertical wind shear - the difference in wind speed and direction - between the upper and lower parts of the atmosphere. This increases the efficiency with which storm systems can form, making super-cyclones, with wind speeds exceeding 185 kilometres per hour, much more likely to form. Amato Evan, from the University of Virginia, led the study published this week in Nature.
Amato - What we're really showing is that human activity can actually change this massive atmospheric phenomenon, but what it also says is that because these aerosols reside in the lower part of the atmosphere, if emission stops, this effect would essentially reverse in a timescale of a couple of months. The relevance of these findings is that although we have in this way, changed the climate in such a way that creates very powerful storms, it's not irreversible.
---
Seeing the Light
Skin can 'see the light' to protect us from UV radiation. Elena Oancea and colleagues from Brown University has found that a light- sensitive chemical called rhodopsin, normally found in the retina, is also present in melanocytes, skin cells that produce the suntan pigment melanin. The cells use the rhodopsin to detect UV rays and then switch on melanin production in under an hour. Previously melanin production was thought to occur only after a few days in response to shorter wavelength UVB radiation, which can damage DNA.
sensitive chemical called rhodopsin, normally found in the retina, is also present in melanocytes, skin cells that produce the suntan pigment melanin. The cells use the rhodopsin to detect UV rays and then switch on melanin production in under an hour. Previously melanin production was thought to occur only after a few days in response to shorter wavelength UVB radiation, which can damage DNA.
Elena - So, if a small amount of initial UVA exposure increases the skin's defence to UVB, it's really important not to have for example UVB only sunscreens. The other thing is that if this is a protective response and we have identified the molecules that mediate this response that leads to melanin production then we can activate the pathway artificially and increase the skin protection.
---
New Chemical Elements
Three new chemical elements have been officially named by the general assembly of the International Union of Pure and Applied Physics - IUPAP. Elements 110, 111 and 112 in the periodic table have beennamed darmstatdium, roentgenium and copernecium respectively. They were named by physicists from around the world and are now officially part of the periodic table.
---
The Key to  a Roaring Success
a Roaring Success
The secret of a lion's roar lies in the
shape of its vocal cord, new research has shown. A lion can generate sounds as loud as 114 decibels, equalling that of a jet engine taking off. Scientists had believed that the loud, low-pitched roar was down to the weight and presence of fat within the animals' vocal cords. But now, by analysing samples of lion and tiger vocal tissues, Sarak Klemuk's team at the University of Utah identified the key features to be stretchiness, pliability and square shape of the vocal folds themselves.
Sarak - Little lung pressure is really needed to set these vocal folds into vibration. The Panthera vocal fold is a very square shape. The mechanical properties, along with that square shape allows a lion to generate a very loud roar at a very low pitch.The researchers equated the sound to that of a baby's cry. Both sounds demands attention; but lions use it as a scare tactic to keep intruders away, rather than a call for help...










Comments
Add a comment