You've probably heard of DNA, but what do you know about RNA? As well as being the molecule that shuttles information from our genes into our cells, it also plays a huge number of other roles in all cells, from bacteria and viruses to tiny worms, plants and humans. Plus we delve into quadruplex DNA, wonder about the wolf genome, speculate on skin colour and our gene of the month has a literary twist.
In this episode
01:07 - Dr Julie Claycomb - Understanding RNA
Dr Julie Claycomb - Understanding RNA
with Dr Julie Claycomb, University of Toronto
Kat - Everyone's heard of DNA, or deoxyribonucleic acid to give it its full name - the blueprint of life that encodes the information that tells our cells when to grow, what to be and when to die. But there's another important player in the world of molecular biology, and that's RNA, or ribonucleic acid.
It's becoming increasingly clear that RNA plays a vital role in many different biological processes in all kinds of organisms, from viruses and bacteria to insects, worms, plants and mammals. Tiny RNAs can even control the activity of our genes - something that researchers are now trying to exploit to come up with treatments for cancer and other diseases. I spoke to Dr Julie Claycomb at the University of Toronto to get the low-down on these mysterious molecules.
Julie - RNA is a cousin of DNA. It's a type of nucleic acid and it's really the go-between between the instructions to make proteins, which are your DNA in the nucleus of your cell, and proteins. So, DNA is turned into RNA, a copy of itself and then the RNA is translated into proteins which make up all of you - your skin, your hair, everything.
Kat - We think of this as the central dogma of molecular biology that DNA encodes RNA and these makes proteins, but now we know that RNA is a little bit more complicated. What do we know about RNA now?
Julie - So, in addition to this RNA that's the go-between between DNA and proteins, we have these itsy-bitsy RNAs, these small RNAs that are utilised by the cells to regulate the expression of those bigger messenger go-between RNAs.
Kat - What do we call these little RNAs?
Julie - Yes, so these little RNAs are called small interfering RNAs or microRNAs and it just depends on where they've originated from what we call them. So, the cell makes these microRNAs that regulate gene expression.
Kat - So, the instructions to make these microRNAs, they're still in our DNA as well.
Julie - Absolutely, yeah. These microRNAs are encoded in our genomes and in fact, they're encoded in the genomes of lots of different organisms ranging from plants all the way to humans. So, this is a very evolutionarily conserved mechanism for regulating gene expression.
Kat - What are they normally doing in our cells?
Julie - So normally, they're regulating the levels of gene expression. What they can do is that they can actually cause the inhibition of generating a protein from that messenger RNA.
Kat - So, like switching it off.
Julie - They turn it off basically. They inhibit it from becoming what it's meant to instruct which is the making of a protein. In addition in some instances, these small RNAs can induce the degradation, the altogether destruction of those messenger RNAs. And again, keep them from being turned into a protein or keep them from being expressed as we say.
Kat - Sometimes we might think about a gene that's making protein and then suddenly, we have this whole level of regulation on top of that. So, are these really playing an important role in how our genes are switched on and off?
Julie - Yeah, absolutely. These microRNAs have been implicated in cancer. They're very important for normal development and there are a number of instances in which microRNAs becoming misregulated or mutated leads to aberrant gene expression and the onset of cancer. So, they're absolutely a very key player in regulating gene expression throughout development and in our normal cell division.
Kat - So, if these microRNAs are helping to control which genes are on and off, what's controlling them?
Julie - So, microRNAs are transcribed just as normal genes, mRNAs, are. They're expressed in the cell using the same types of machinery and we still have a lot to learn about what temporally controls which microRNAs are expressed when and in which cell types because there are different types of microRNAs that are expressed in different cell types. So, they're expressed in much the same way as most of your mRNAs, your normal protein coding genes are. They're then processed by a number of other protein machines to make them their small size which is about 20 nucleotides on average.
Kat - So they get chopped up and then go and do their job.
Julie - Yeah, absolutely. They get chopped up. They start as a much longer - we call it a transcript - a much longer molecule which is then processed into several iterations, several shorter molecules. And ultimately, the small RNA is bound by a protein called the Argonaut protein which is the effector molecule, the machine that actually causes mRNA degradation or causes the inhibition of the production of proteins.
Kat - I remember when I was doing my PhD back in the day, this was some time ago, that this field really started to take off because they first really found this was going on in little worms, which is what you work in. Tell me a little bit about the history of where we've come from since those first discoveries.
Julie - Plants and worms have both been very, very important in the key discoveries that led us to understanding how RNA interference and microRNAs work. Basically in plants, one of the things they first discovered was that if you added a colour gene to make the plants more purple, lo and behold! The plants turned less purple. So, there was a process on-going in the plant cells that shut off gene expression and this process came to be known later as co-suppression. In C. elegans, what was found, there were several different discoveries. One of which was in the early '90s that there were these small RNAs - they didn't know they were small RNAs at that time - that led to defects in developmental timing. Subsequently, through a lot of different studies, people identified microRNAs, the loci on the chromosomes encoding these small RNAs, and through technological advances, we've done a lot of identification of these microRNAs and other small RNAs in the worm.
Kat - So, we know that these short RNAs are really important for normal life, normal growth, normal cells to function. But now, we're starting to exploit them. Tell me about some of the things we can do to use this knowledge for our benefit.
Julie - Yes, so surely you can imagine a situation if we could selectively shut off genes that were bad for us, that were deleterious to us that we could then somehow eliminate or reduce disease, right? So, for instance, cancer is often caused by the misexpression of a number of genes which then lead to overdivision of the cells, overproliferation of the cells. If we could turn off those genes that tell a cell to divide and divide, and keep dividing, then we could potentially stop cancer progression. So, as you might imagine, RNA interference and these small interfering RNAs are a very hot topic of interest in therapeutics. So, if we could find a way to exploit them then we could potentially develop new therapeutic mechanisms.
Kat - That was Dr Julie Claycomb from the University of Toronto.
08:26 - Quadruplex DNA
Quadruplex DNA
with Nell Barrie
Nell - Well, the first thing we've picked out is a really interesting story about quadruplex DNA which sounds like this big new, exciting thing, but in actual fact, not quite as new and exciting as some of the reports made out.
Kat - It was like, "Oh my God! It's the new DNA helix. Everything we know is wrong." It's not.
Nell - DNA 2.0.
Kat - People have known about this for quite a long time and in fact, this isn't actually the first time they found quadruplex DNA in humans because they did that in 2011, the same group. But what is quadruplex DNA?
Nell - So, this is a type of DNA, so that instead of being two halves curled into a nice little spiral - how everyone sees it in cool little 'sciency' bits of films and on TV.
Kat - The double helix.
Nell - Exactly. It's curved around. It's almost like a circle, so it's got four sides to it. So, it's kind of a square-ish circle sort of formation and it's to do with what that particular section of DNA is actually made up of. So, if it has a lot of guanine in it, it could be that that's what makes it curl up into this particular shape.
Kat - So, it's the physical properties of the chemicals that are making up that stretch of DNA and it turns out, there's a lot of these guanine, or G-rich, sequences in telomeres at the ends of our chromosomes. And funnily enough, that's obviously where they found them and they've done a really nice technique. This is a research from Shankar Balasubramanian and Giulia Biffi. Tell me a bit about what they actually did to spy on this DNA in cells.
Nell - So, you mentioned that we have already seen evidence that human cells can have this kind of quadruplex DNA and we already knew that some other forms of life - so small organisms, single-celled organisms - might have it. But now, we know it's in human cells too. We did already have evidence of this, but what the new research has done is actually looked at when the quadruplex DNA is produced. So, which stage of the cell cycle, when it appears, when it disappears, and they've been able to tag it with a fluorescent marker so they can essentially watch it appearing, disappearing, moving around.
Kat - And when is it mostly turning up?
Nell - So, it's in a phase called S-phase. 'S' stands for synthesis and essentially, what's happening then is all the DNA in a cell is being copied. And this is quite interesting because in terms of relevance to cancer, it's a stage when a lot of cancer drugs will act on the cell and they'll stop various things from happening in the cell at that point in time.
Kat - So, that's also quite a good opportunity to intervene as well if you wanted to maybe get drugs that stuck to the quadruplex DNA or interfered with it in some way. That would be pretty good.
Nell - Exactly, so the idea here is that if it turns out that quadruplex DNA is playing a role in driving cancer cells, making them grow, making them divide, it could be a chance for people to develop drugs that perhaps latch onto the quadruplex DNA, block it from doing whatever it's up to in the cell, and that could be a new way to tackle cancer cells.
Kat - Or even if it's not driving them, if it's just there, if it's maybe some kind of marker that happens to be there more in cancer cells - we don't really know. It's very interesting. That came out in Nature Chemistry this week from the team at the University of Cambridge.

11:39 - Dog domestication
Dog domestication
with Nell Barrie
Kat - Another story that you highlighted to me was some research from Eric Axelsson and his team in Scandinavia and the US, and this is in Nature. And this is about dogs because I love dogs. We always have some kind of animal genetics thing. What's this about dogs?
Nell - So, I think this is really interesting because essentially, what it's doing is using genetics to go back and try and find evidence to help answer the question about how dogs might have evolved. Obviously, this is a really tricky question because we don't have a lot of evidence. There's no written records, we haven't got any scientists from 14,000 years ago who've published paper on what happened. But instead, we can go back, we can look at the DNA of dogs and we can figure out how that's changed compared to wolves on the assumption that dogs evolved from wolves. So, these scientists have looked at the genetic code of wolves and dogs. They've compared it in loads and loads of detail to see exactly what those differences might be.
Kat - What can it tell us about the relationship of dogs to us and how they evolved?
Nell - So essentially, what they found is there were two main areas where there were obvious differences between the dog DNA and the wolf DNA. One of those was in various brain pathways, so that could be to do with the way that dogs' behaviour is clearly quite different from wolves. The other one that I really was interested in was looking at starch metabolism. So essentially, they found that dogs tend to have many more genes that allow them to metabolise starch in their cells. And this links in quite nicely with the theory that dogs may have evolved by taking advantage of all the waste produced by human societies. So, you can imagine a group of hunter gatherers perhaps. They leave these waste dumps wherever they go, so they have bits of food they don't want anymore, bits of crops maybe if they're farming, and dogs could be able to take advantage of that. So there might have been wolves who'll go, "Okay, here's some food I could eat." The ones who are better able to digest the starch, to digest all these kind of waste food would've been more successful and perhaps could've gone on to evolve into these other species.
Kat - And presumably, with the evolution in the brain as well and giving them characteristics that might make them more amenable to hanging around with humans. It's not a done-deal. It's quite controversial because from what I know, there's another theory that dogs evolved because humans were sort of harnessing wolves for hunting or using wolves for hunting.
Nell - Exactly. I mean, I just like the idea that you could maybe come up with an idea for which of those theories was more accurate using genetics and exactly, you can look perhaps as well at the behaviour because we know that if you breed dogs, you can quite quickly select the ones that are more tamed and are more docile. And there's a theory that that's exactly what happened with wolves. So, if people were using them for hunting, it would be much easier to have wolves that were less aggressive, had better behaviour. And if you ended up selecting those, that could change their behaviour every time.
Kat - I saw an article about this describing dogs is basically more like puppies. They're just grown up puppies and we see these in many species. For example, the human face is basically a baby monkey face and the idea that in behaviour as well, you can preserve those juvenile characteristics into adults.
Nell - Yeah, exactly and that so some of these genes that they were looking at, there's some evidence that that could be maybe what they're doing. But clearly, it's really difficult because we've got all those evidence from a long, long time ago, from thousands of years ago, but there's not a lot to go on in terms of what we know about what happened when. And there's also this theory that dog domestication could have happened multiple times in different ways, in different places. So, it's not necessarily true that there's one answer to that question.
Kat - Well, that's one "ruff" idea. Anyway, thanks very much. That's Nell Barrie science writer.
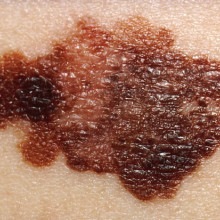
15:09 - Gene mutation in melanoma
Gene mutation in melanoma
Writing in the journal Science, German researchers have revealed a previously unknown genetic route to melanoma - the most dangerous form of skin cancer. Around one in ten cases of melanoma is caused by an inherited gene fault, so the researchers started by studying a family where 14 members were affected with the disease.
They discovered that they all carried a gene mutation that causes overproduction of a protein called telomerase, the so-called immortality enzyme that helps protect the ends of chromosomes as cells divide. Then they looked at the telomerase gene in non-hereditary, or sporadic, melanoma and found that nearly three-quarters of the samples they looked at also had faults that increased the production of telomerase. And all these gene faults carried the signature of damage caused by excessive exposure to UV radiation from the sun or sunbeds.
The researchers believe their results prove that faults causing overactive telomerase are a key driver in melanoma. They were also surprised to find it, as the genetic faults in melanoma have been extensively analysed, but somehow this was overlooked. They also hope that their discovery will lead to more effective treatments for the disease, as therapies that block telomerase are already being tested in clinical trials and could be beneficial in melanoma patients.

16:25 - Gender gene clues to heart disease
Gender gene clues to heart disease
As we approach Valentine's day, much is made of the difference between the behaviour of men's and women's hearts in the romantic sense. But in purely biological terms there's little difference between these organs, except for small differences in the electrical signals they produce. Now researchers at Washington University in St Louis have managed to track down some important molecular differences in human hearts from both sexes that could lead to more personalised treatment for cardiac problems in the future.
Publishing their results in the journal PLoS One, the researchers looked at samples taken from 34 human hearts from people that were either receiving a heart transplant following heart failure, or whose healthy hearts had been rejected for transplantation, and looked at nearly 90 important genes involved in heart rhythms and other functions. They found major differences in the activity levels of many key genes in the atria - the chambers at the top of the heart - rather than the lower chambers, or ventricles. Overall men had higher levels of activity of nearly all the genes, while women with failing hearts had unusually low levels of gene activity - in some cases less than half of that seen in men.
The scientists hope their findings will shed light on how best to treat men and women with heart failure.
17:33 - Storing stories in DNA
Storing stories in DNA
And finally, enormous library archives of magnetic tape or other media could one day be a thing of the past if a new data storage technique takes off - storing information encoded in DNA. Publishing in Nature, the team from the European Bioinformatics Institute encoded several different files in DNA, including a text file comprising all of Shakespeare's sonnets, Watson and Crick's 1953 paper on the structure of DNA, a photo, and a short excerpt from Martin Luther King's famous "I have a dream" speech. If kept under the right conditions - namely a cool, dark environment - DNA can last intact for thousands of years and the costs of synthesising it are coming down all the time.
Although it's not going to replace a hard drive or USB stick any time soon, experts think that long-term DNA-based information storage could become feasible within the next decade.
18:41 - Dr Susan Gottesman - Bacterial RNAs
Dr Susan Gottesman - Bacterial RNAs
with Dr Susan Gottesman - Centre for Cancer Research, Bethesda
Kat - Here's Susan Gottesman from the Centre for Cancer Research in Bethesda, Maryland, who's an expert on bacterial RNAs. I asked her to explain more about how small RNAs can control the activity of genes in bacteria.
Susan - So, in parallel both in bacteria and humans, some of them have the ability to decide which of the messengers actually get translated into the protein machinery. So they pair with the messages, and then can change their fate, so they can make them disappear from the cell when they otherwise would be there. They can make them more active than they would otherwise be. And so, the cell has a capacity for sending in new signals to these small RNAs which are then regulating these much bigger messages and that's important for development, it's important for cancer, important for disease, it's clearly important for bacteria to respond in a proper way to their host, and cause disease.
Kat - So, how are these small RNAs made?
Susan - So, that's a little bit different between bacteria and animals because animal cells have a very complicated process. In the bacteria, they look just like some of the other genes. They have promoters, they're made of small pieces, and then they fold up and they can work. In the higher animal cells they're made in the nucleus, they have to be folded up, they have to be cut, they have to be sent out of the nucleus, and then processed down. In each of those steps, in some place at which the cell can say, "Okay, I don't really want this or I do want it now."
Kat - And in bacteria that you study, what are these small RNAs up to? What sort of processes are they controlling?
Susan - Well, the more we study them, the more we find out about what they're doing. So, if I tell you what I know now, there'll be something new. We know they regulate whether the bacteria can swim from here to there. They regulate whether the bacteria have enough iron inside them to grow properly and how it gets that iron. They regulate whether the bacteria know to continue growing or stop growing so, almost everything. Every decision the bacteria has to make, and bacteria have to make a lot of decisions, the RNAs are in there, sticking in their two cents.
Kat - And what do we know about how this system has evolved because RNA and DNA kind of growing up in parallel?
Susan - That's really a very interesting question and I don't have an answer to it, so it's one of the things that I really like to play with, trying to figure out where these RNAs [came from] - are they ancient or are they newly evolving? They are evolving quickly. So, I think now that we know that they're there, we know where to look for them, we can start to understand what they do in the more complex behaviours. Let's just stay in the world of bacteria, how the bacteria interacts with its host, how it causes the disease, did that bacteria ever send RNAs to talk to the animal cells or vice versa. A lot of those things that we think would be really interesting that we don't know about yet. So, lots yet to look at.
Kat - That was Dr Susan Gottesman from the Centre for Cancer Research.
22:05 - Professor Joan Steitz - Viral RNAs
Professor Joan Steitz - Viral RNAs
with Profesor Joan Steitz - Yale University
Kat - Joan Steitz at Yale University has worked on RNA for more than three decades, and specialises in the small RNAs produced by viruses. I started by asking her how RNA research has grown in importance over recent years.
Joan - Our cells, I think the latest numbers were something like 2,000 of these microRNAs are known to be made by our cells in various tissues at various stages of development. And they have huge impact on the actual output of the genes in terms of the protein - huge expansion in terms of the appreciation of RNA, and its regulatory roles in the whole process of DNA makes RNA makes protein.
Kat - So, we've got the messenger RNAs that tells cells what proteins to make, we've got all these microRNAs controlling things, we've got tRNAs that help to build proteins, and all the RNAs that are part of the machinery of cells. What's your favourite one? What's the one that you're working on?
Joan - I don't know that I have favourite ones, but I do like the RNAs that are in the so-called 'snurps' (snRNPs) that are part of the splicing apparatus, because we worked on those for a very long time. At the moment, we're working on viruses and it turns out that many of the viruses that infect us also make non-coding RNAs in order to manipulate the cells that they live in and help them with their lifecycles. And the really fun thing about viruses is that viruses don't really sort of make things up. They get things from their host and then they adapt them to the ends of the virus. And so, viruses can make non-coding RNAs that you look at it and you say, "Oh, I know what that RNA does" because you know what the host RNA that looks like that does. But it turns out that viruses are very clever and in many cases, they've completely changed with that RNA might do into doing something completely different that fits with what the virus needs it to do. So, that's providing lots of insights into the evolution and how some of these control mechanisms came to be and how versatile they are.
Kat - You mentioned evolution and I know that it's usually thought that RNA evolved before DNA and the fact that we're discovering so much more about these RNAs and what they do, and that they're involved in viruses, are we almost getting a window on what our previous biological world was like?
Joan - I think you're absolutely right, yeah. I don't believe in a pure RNA world. This is just personal. I think that RNA has always existed with helper peptides or proteins that bound to them, and made them more functional for the various functions that they're carrying out. But I think we are getting, especially now with massive databases and being able to compare and contrast what one sees in one organism or another versus what one sees in a particular virus or another that we're getting a whole new view into the possible ways that things could've evolved.
Kat - That was Professor Joan Steitz, from Yale University.

Why are there different skin colors?
Louise:: This month's question is from Agum in Canada who asks, "Why are there different skin colours? How did that happen?" And to answer this question, I spoke to Dr. Aylwyn Scally from the Department of Genetics at the University of Cambridge.
Aylwyn:: Skin colour is like lots of human characteristics, one of these things which is predominantly determined by your genetic content. Another way of saying it is that it's determined by your ancestry, so we know the people whose ancestry comes from Africa or regions close to the equator tend to have darker skin colour and people further away in higher latitudes will have lighter skin colour. And that is something which we've recently been able to actually identify in terms of which parts of the genome and how much of our DNA is actually involved in determining skin colour.
We can say quite a lot now about when that might have happened, when these changes might have occurred, and then speculate about the reasons why they might have occurred. But there are still questions that we don't understand; we don't have the answers to. One of which is really the basic thing is, exactly why have these genetic changes taken place, what has been the benefit for people living in northern latitudes for them to have lighter skin colour? We have ideas about that and they're related to protection of the skin from UV light. That's what people have speculated about for a long time, but we don't really have a very clear cut example or reason for why that might be the case.
Alternatively, there's also the idea that when your skin is heavily pigmented, it means it protects you against UV light, but actually, you need certain amount of UV light to synthesise vitamin D in your body. So, when the amount of UV light you're getting is lower, say, when you're living in a northern climate, then that means that it's beneficial to actually increase the amount of UV light and therefore, that could be a reason why people have evolved lighter skin. We were able to look around across the whole genome and look at different regions and see where these changes have taken place, and we can see that in Europeans, it seems to have happened quite recently - recently in terms of human evolution that is. So, within the last 20,000 years or so, that's sometime around since the last Ice Age. And you can also see lighter skin colours elsewhere around the world, in Asia, but the genes involved are not necessarily the same ones. We can identify different genetic changes that you find in east Asians who also have lighter skin than Africans, our ancestors.

28:19 - Gene of the month - Amontillado
Gene of the month - Amontillado
with Kat Arney
And finally, our gene of the month is Amontillado, a gene with a rather macabre twist. Horror fans may know 19th century writer Edgar Allan Poe's story "The Cask of Amontillado", where an unwitting victim is lured into a wine cellar with the promise of a precious vintage sherry, then bricked up alive by the murderous protagonist. Rather like this unfortunate literary end, fruit fly larvae with a faulty Amontillado gene cannot hatch, and remain trapped in their pupae to die.
In contrast to many of the genes we've featured in this podcast, the mammalian versions of Amontillado were actually identified first in the form of a protein called PC2, which chops up hormone precursors to make finished hormones. The victims of PC2 include a substance called pro-opiomelanocortin, which can be processed in a variety of ways to make lots of different useful hormones, none of which give you a predisposition to liking sherry.
- Previous Our ancient ancestors, deep sea worms
- Next What is Love?










Comments
Add a comment