Just the bare bones this week as we find out how exercise strengthens the skeleton and how new scanning techniques can help to pick up osteoporosis earlier and inform its management. We also try out a new gadget for measuring the force muscles can apply and, in the news, discover what a self-healing tumour can tell us about common cancers, evidence that mammalian hearts can repair themselves and a new laser-based tool for diagnosing melanoma. Plus, how the bones of people who died up to a hundred years ago are helping scientists to combat chronic back pain by building a computer model of the backbone...
In this episode
- Video - Learning from Old Bones
Video - Learning from Old Bones
| ||
For a full transcript of the interview click here.
01:24 - Self-healing skin tumour could shed light on more common cancers
Self-healing skin tumour could shed light on more common cancers
"Self-healing" is not a word that people usually associate with cancer. But researchers studying a very unusual and rare form of skin cancer that can clear up by itself have learned some lessons that could potentially lead to new treatments for the disease in the future.
It's a rare hereditary cancer called multiple self-healing squamous epithelioma, or MSSE for short - it only affects a handful of people every year in the UK, and most of them can trace their ancestry to a single family from Scotland. The disease causes large flat tumours on the skin which heal up of their own accord, leaving disfiguring scars but no other lasting effects.
Because of the inheritance pattern of the disease, scientists know that it must be caused by a fault in a single gene, but the identity of this gene has been a mystery for more  than 40 years. But now an international team of researchers led by Cancer Research UK's Dr David Goudie in Dundee have found the culprit. The team have published their findings in the journal Nature Genetics this week, and their discovery could shed light on more common types of cancer as well.
than 40 years. But now an international team of researchers led by Cancer Research UK's Dr David Goudie in Dundee have found the culprit. The team have published their findings in the journal Nature Genetics this week, and their discovery could shed light on more common types of cancer as well.
The researchers looked at DNA samples from around 60 people with MSSE, and compared them with DNA from more than a hundred of their relative who were unaffected. They focused in particular on a region of DNA that had previously been shown to harbour the likely gene, narrowing down their search until they found the gene TGFBR1.
This gene encodes a receptor protein, which receive signals from a protein called TGF beta. These signals tell cells to divide, or to stop dividing, and the researchers were able to prove that carrying a faulty version of TGFBR1 caused people to develop MSSE tumours. Intriguingly, faults in TGF beta signalling have been found in many types of cancer.
TGF beta signalling is very interesting to cancer researchers, because it can be both a 'goodie' and a 'baddie' in the disease. In the early stages of more normal cancers, it tells cells to stop dividing, acting as a brake on tumour growth. But after a while, it switches sides, and TGF beta signals tell cells to divide and grow faster, fuelling the growth of cancer.
But in the case of MSSE, the opposite seems to happen. Cells start off growing out of control, forming the characteristic skin tumour, but then they stop growing and go into reverse.
TGFBR1 is just one of a family of receptors that are involved in receiving TGF beta signals. The researchers think that the pattern of signals must be changing as the tumours grow and then heal, allowing the 'good' side to come forward.
Although MSSE is a very rare and unusual cancer, this discovery could shed light on other, more common, types of cancer, because we know that TGF beta signalling is involved in many types of cancer. If researchers could figure out exactly how the changes in TGF beta signalling make MSSE tumours heal up, then maybe it could pave the way for future treatments for more common types of cancer. So this is a nice example of how research into a very unusual disease could have a much wider relevance.
Goudie, D. et al (2011). Multiple self-healing squamous epithelioma is caused by a disease-specific spectrum of mutations in TGFBR1 Nature Genetics DOI:
10.1038/ng.780
04:49 - Getting to the heart of cardiac repair
Getting to the heart of cardiac repair
A surprising discovery suggests cardiac self-repair may be possible. Prevailing wisdom suggests that mammalian hearts like our own lack the capacity to self-repair if they are injured. This is in stark contrast to other  animals, including fish, that can regenerate up to a quarter of their heart muscle if it is injured or removed. But now scientists have discovered that the newborn mammalian heart also initially has the ability to do the same, but only for a short while.
animals, including fish, that can regenerate up to a quarter of their heart muscle if it is injured or removed. But now scientists have discovered that the newborn mammalian heart also initially has the ability to do the same, but only for a short while.
Writing in Science, University of Texas Southwestern Medical Center scientist Enzo Porrello and his colleagues removed about 15% of the muscle from the apices of the left ventricles of 1 day old mice. After forming a blood clot to seal the wound at the site of the injury, the mouse hearts re-grew completely within 21 days, without the development of scar tissue and with the formation of new muscle tissue and blood vessels; functional tests carried out at two months also confirmed that the recovered hearts were beating completely normally.
The recovery was achieved, the scientists found, through the proliferation of surviving muscle cells elsewhere in the heart; newly-produced heart cells migrated to the site of the injury where they then wired themselves into the regenerating tissue to replace the lost muscle. However, when the same procedure was attempted on slightly older, 7 day-old mouse pups, there was no such regenerative effort, the injured hearts developed significant scar tissue and failed to repair themselves. This intriguing result shows that the mammalian heart is capable of self-repair similar to the hearts of simpler species like zebrafish but, for some reason, this capacity is lost soon after birth.
Encouragingly, however, it also suggests that if scientists can uncover the reason underlying this switch from a pro- to a non-repair state, using a model system like this, then it might be possible to trigger the same restoratative pathway in adult human victims of cardiac diseases.
07:25 - Old Bones Help Treat Back Pain
Old Bones Help Treat Back Pain
with Jane Reck, EPSRC; Dr. Ruth Wilcox, Leeds University; Dr. Kate Robson Brown, Bristol University
Kat - Also in the news this week, scientists have been using old bones to develop new treatments for chronic back pain. [It's estimated that] 80% of us will suffer from a bad back at some time in our lives, but the condition is hard to treat because the causes of it are so varied. Now, researchers have come up with a new way of testing out new treatments with a little help from our ancestors, as Jane Reck has been finding out...
Ruth - The data that we need, we can derive from these older bones. So, although the bones are old, what we end up with in the model is a simulation of a living spine.
 Kate - I, as an anthropologist, am providing access to data and providing access to analytical techniques which are going back, into bio-mechanical engineering.
Kate - I, as an anthropologist, am providing access to data and providing access to analytical techniques which are going back, into bio-mechanical engineering.
Jane - The unlikely combination of old human bones and the latest computer modelling techniques, are being used to develop new ways of treating chronic back pain. Spines from around 40 skeletons, housed in various museums and anatomy collections are being analysed in the research, which is taking place at the Universities of Leeds and Bristol. The computer modelling side of the work is led by Dr. Ruth Wilcox at Leeds.
Ruth - We're using computer models, the same kinds of modelling processes we'd use in aeronautical engineering or in civil engineering to analyse structures, but using those types of principles to model the spine, and to simulate how a treatment would work within a spine.
Jane - Ruth says, they're carrying out the work using the latest imaging techniques.
Ruth - Microcomputer tomography scanning is imaging a specimen in three dimensions. So it's similar to the type of techniques that we have in a hospital called a CAT scan. In a hospital, a patient is imaged by basically being x-rayed from different angles and then using that to build up a 3D image of the patients. This works in the same kind of way, but we're doing it at much higher resolution so that we can see the individual strands or struts of bone within the vertebra. And that data, we then put into our computer models so that our computer model simulates this variation in bone across an individual vertebra but also from one patient to another.
Jane - The bone remains are being gathered and scanned at the University of Bristol's Department of Archaeology and Anthropology by Dr. Kate Robson Brown. Kate explains why they're using such old human remains.
Kate - Most of the time at the moment, we get material from the more elderly sections of the population. And in order to understand the range of variation within a normal population, we need to have information from younger adult age groups. By using older collections of dry bone; these are macerated, which means cleaned, dry skeletons from those collections, it allows us to investigate bone morphology in age groups which are not accessible using recently donated material. One of the skeletons we're looking at is that of an adult female. She was quite little. She was around 5'2" when she died. She was probably in her late 20s or early 30s. We have her complete skeleton. This was a skeleton that made it's way into the collection we think in the 1930s, and this particular individual has a very clean, dry skeleton, so there's no evidence of osteoarthritis, there's no evidence of bone cancer, anything like that. We can't tell from her skeleton what she died of, but it was certainly nothing that is represented on the bones itself.
Jane - Ruth says, the new computer software will speed up the process of clinical trials, for testing out new treatments for chronic back pain.
Ruth - The idea by the end of this project is that we'll have these models up and running. A company could come in with a design for a new product and we could simulate how it would work across this population of different spines, so we could reduce the risk when it does go into the clinic, and prove beforehand that in our computer models, that it's behaving as it should. The good thing about the computer models is that we can use them over and over again, so we could test lots of different products on the same computer model whereas if we're doing this in the laboratory, then we'd need a new spine, a new donated spine each time we wanted to test the new treatments out.
Jane - Kate says the work could lead to tailor-made medical treatments and even provide new insights into how our ancestors evolved.
Kate - It would surely be great if we could be in a position where surgical interventions could be assessed for us as an individual against a model of our own bones. We're moving in that direction, but research like this is helping to provide the baseline data on which those developments can be made. One of the really exciting developments that's arising out of this, for me as a Biologic Anthropologist, is being able to apply this type of modelling technique to fossil bones, for which there is no other way of testing them. So for example, we could take a fossil skeleton that's 3 million years old, scan a vertebra, make a model of it, and actually test some locomotory strategies to see whether that individual in life walked around on two feet or four feet. That's a major development for evolutionary anthropology.
Jane - The research is funded by the Engineering in Physical Sciences Research Council, the computer modelling software is the first of it's kind for back conditions. It should be available for testing out newly developed products and treatments in the next few years.
Kat - Well, that's making me want to sit up straight with just listening to that. That was Jane Reck from the Engineering and Physical Sciences Research Council.
| Learning From Old Bones - EPRSC Video |
13:19 - Laser technique offers optical biopsy
Laser technique offers optical biopsy
Scientists have developed a new tool that could significantly improve the sensitivity of melanoma screening techniques.
Currently, the majority of melanoma diagnoses are made clinically by eye,  but even visualisation by a skilled dermatologist can miss up to 15% of lesions. Moreover, studies have also found that pathologists can fail to agree on a diagnosis in 14% of biopsy samples. As a result, doctors tend to act cautiously and "when in doubt, cut it out", leading to a high false-positive rate and over-treatment of benign lesions.
but even visualisation by a skilled dermatologist can miss up to 15% of lesions. Moreover, studies have also found that pathologists can fail to agree on a diagnosis in 14% of biopsy samples. As a result, doctors tend to act cautiously and "when in doubt, cut it out", leading to a high false-positive rate and over-treatment of benign lesions.
Now clinicians could soon have a new weapon with which to combat the problem. Writing in Science Translational Medicine, Duke University researcher Thomas Matthews and his colleagues have found that a laser technique called "pump-probing" can be used to detect the quantities of different forms of melanin in a skin sample. Using 42 biopsy specimens, which included a mixture of benign and melanoma lesions, the team exposed the tissue to two brief bursts of laser light of two slightly different wavelengths (colours).
The first burst of light (the pump) sensitises one form of melanin - called eumelanin, which is darker and increases in concentration proportionally in malignant cells - to the second short laser blast (the probe) delivered shortly afterwards. This enabled the team to quantitate the relative concentrations of eumelanin and its lighter phaeomelanin relative in their samples. Comparing the levels of each enabled the researchers to correctly identify all of the genuinely-malignant specimens amongst the samples studied, although the team acknowledge that they have not tested their system against the subset of non-pigmented melanomas that don't make melanin.
However, for the majority of malignant melanomas, which are pigmented, the team point out that, apart from having the potential to dramatically improve current clinical protocols, at the laser powers they have been using, the system could readily be adapted to do diagnoses in-situ on a patient without needing to take a biopsy first.

17:27 - Planet Earth Online - Roman Recycling
Planet Earth Online - Roman Recycling
with Caroline Jackson, Sheffield University; Harriet Foster, Norfolk Museums and Archaeology Service
Kat - Planet Earth podcast presenter Richard Hollingham has been asking what have the Romans ever done for us? Well, apart from building baths, sewers in cities and education, it turns out they were also into recycling. And to hear how, he met up with Sheffield University's Caroline Jackson and Harriet Foster from the Norfolk Museums and Archaeology Service...
Caroline - What we've got are six vessels. Three drinking vessels, there's two jugs and a bottle, and they're all late 4th century.
Richard - Now we've got here a modern glass which I am allowed to touch. Actually, you look at this mass-produced modern glass and you compare it with these from almost 2000 years ago, Caroline.
Caroline - That's right.
 Richard - These glasses could almost be mass-produced because those three glasses, pretty much identical. You can imagine those in a box at a supermarket or something.
Richard - These glasses could almost be mass-produced because those three glasses, pretty much identical. You can imagine those in a box at a supermarket or something.
Caroline - Well that's right. They actually were mass-produced in a way. Glass production, certainly by the 4th century was something which was prolific. Glass is found in most archaeological contexts. It's all blown glass, this is something which came in certainly in the Roman period. The Romans invented glass blowing, something that perhaps wasn't around in earlier periods. So, to produce the glass by blowing is very rapid. It's a very easy technique once you've mastered it. So you can produce many, many vessels from a small amount of glass, and this is why these have such thin walls as well - because they're blown.
Richard - How do you know they recycled the glass?
Harriet - We've known for a long time that the Romans recycled glass. We've got evidence from a late 1st century AD source that talks about broken glass in Rome being collected up in exchange for items of small worth. There's archaeological evidence for glass recycling. So we find the equivalent of bottle banks.
Richard - So what did you do then?
Caroline - We took a lot of material from about 20 sites in Britain and analysed them chemically to see if we could see anything within their compositions which might give us an indication that recycling was taking place. And the results of that were quite surprising.
Harriet - They were surprising because of the amount of recycling that we found that was going on. A significant proportion of the assemblage that we looked at which was over 500 samples suggested to us that the glass was being recycled in quite large amounts and that was interesting because it throws out more questions about, what does that mean about the supply of glass to the province at that time? Was glass being recycled because there weren't fresh supplies available or is it something else?
Richard - Have you got any ideas why this might be going on?
Caroline - By the late 3rd and 4th century, the Roman Empire is starting to change. The large industrial complexes that you see are starting to decline, trade is starting to contract. Now, we don't know exactly what was happening with supplies of glass because we don't have any archaeological evidence at this point that is published, where we know glass was being produced. So we can't say this factory stopped and maybe that factory started, but we can say that it looks as though that supply of raw glass is starting to decline, because there is a greater proportion of recycled glass that you can see in the chemical fingerprint. We've got elements in there which can only come from mixing that can't have come from the raw materials. That's not just for the common forms and the common colours, even this more high status colourless glass, which is always thought of as something which is reserved perhaps for more specialised vessels, even that is showing evidence of large scale recycling. So it's going on throughout the glass industry.
Richard - Okay, so you're learning about the glass, but you can also, by using the glass, tell a lot about how the society was changing as well.
Caroline - Well that's archaeology. That's what we do. The glass is a really interesting material, but only interesting in what it tells us about the people who were making it and consuming it. In the same way as we look at any archaeological material, it's there for a purpose. It's there to tell us about people, the individual, and society.
Kat - That was Caroline Jackson and Harriet Foster, talking to there to Planet Earth podcast presenter Richard Hollingham. You can hear the latest edition of the Planet Earth podcast as well as links to other Planet Earth Online resources at thenakedscientists.com/planetearth.

22:31 - Strengthening your Skeleton
Strengthening your Skeleton
with Professor Tim Skerry, Sheffield University
Kat - There are 206 bones in the average adult human although Dr. Chris is clearly lacking in his funny bone and has one less! But how do our bones grow and develop and what controls how strong they become? We're joined today by Professor Tim Skerry and he's from the Mellanby Centre for Bone Research at the University of Sheffield where he's looking at the impacts of exercise on bone density and how bones respond to stress. So hi, Tim.
Tim - Hello, Kat.
Kat - Now let's start by finding out what bones are. When I think of bones, I just think of a skeleton, these hard 'twigs' that are inside us, but it's not like that. What are bones made of? How do they work?
 Tim - Well it's certainly very easy to fall into that sort of trap because you see bones in museums and think "dinosaurs", and they're just sort of lumps of concrete really, but that's not the case at all. I think you only have to think about a baby being born, and the speed with which babies grow into children and then adults, to realise the skeleton is quite an unusual material for something that's apparently sort of hard and rock-like. It's made of two main components, a very strong tough protein, rather flexible, called collagen, and a mineral which is a calcium phosphate called hydroxyapatite. These combine to make a thing called a composite material like fibreglass and carbon fibre composites which are extraordinarily strong and very light. So it's a smart material arranged in a smart way and it can grow and repair itself during life.
Tim - Well it's certainly very easy to fall into that sort of trap because you see bones in museums and think "dinosaurs", and they're just sort of lumps of concrete really, but that's not the case at all. I think you only have to think about a baby being born, and the speed with which babies grow into children and then adults, to realise the skeleton is quite an unusual material for something that's apparently sort of hard and rock-like. It's made of two main components, a very strong tough protein, rather flexible, called collagen, and a mineral which is a calcium phosphate called hydroxyapatite. These combine to make a thing called a composite material like fibreglass and carbon fibre composites which are extraordinarily strong and very light. So it's a smart material arranged in a smart way and it can grow and repair itself during life.
Kat - So the idea is that the bones are really kind of dynamic living things. They're not just sticks in our body, they're constantly growing and changing, repairing themselves.
Tim - Yes, that's right and the way you should think about this is really to start with some very simple fundamentals. If you think that when we're conceived and born, no one knows what our lives are going to be. Some of us will be weight lifters, some of us will be more sedentary people, and the sorts of activities we're going to do in our life will vary hugely. Now, if the bones we had were purely determined by some sort of genetic thing, then we'd all have to have extremely strong skeletons for the very small percentage who were going to do extreme activities. Nature doesn't really do that. It doesn't like to grow, maintain and carry around vast amounts of material that for most of us, wouldn't be used. So, very much like our muscles, our bones respond to what we do, they get strong if we use them, they get weaker if we don't use them, so that we're carrying around just enough to do the things we do with our daily life. Plus a certain amount of safety factor to cover for those occasional high impacts.
Kat - When you accidentally fall over or something like that. What sort of things can actually affect the density of your bones? How can we make our bones stronger?
Tim - Well, exercise is the most important thing that a single person can do. Everyone talks about genetics but you can't change that. You can certainly change the exercise you do and there's no question that if you exercise, certainly, while you're growing up, it's an extraordinarily potent stimulus to make a strong skeleton. And if you keep exercising through life then you'll end up having a strong skeleton and being more unlikely to get osteoporosis when you're older.
Kat - But what do we mean when you talk about exercise? I mean, exercise to me could be anything from Pilates to bench pressing, or running a marathon. What sort of exercise really builds your bones?
Tim - Well the way we can determine this is by measuring what we call strain or deformation. If you think about an eraser that you squeeze between your fingers, then if you squeeze it, it gets shorter. Our bones are exactly the same and they deform. So if I'm sitting down as I am now, my bones are longer than if I stood up. If I stood up, they'd be shorter because they were squashed down. So, the strains that we impose by our activities are the things that determine whether the exercise has an effect. And so, big loads that deform our bones a lot have potent effects on the skeleton. But also, exercises that impose those loads very quickly, so impacts, high impacts are more potent as a bone building stimulus than slow repetitive things. And we can really take this and work out what exercises are good for your skeleton. And they're the things which are doing a wide range of different things usually quite quickly, not things like marathon running, cycling or swimming. Now it does not say those aren't good for you, but they're just not particularly good for your bones, and if you measure the bone mass of people who do those activities at high levels, they're very little different from age-matched people in the population. So, fast impacts are very good and if you want a simple rule of thumb, if you do an exercise which makes your muscles bigger then you'll have big bones. If you do an exercise which makes your muscles trained for endurance, but they don't get bigger, then it generally doesn't affect your skeleton very much.
Kat - So, high impact would be something like squash or jumping up and down. Would something like weightlifting count?
 Tim - Weightlifting is good, it's a very interesting question because weightlifting is good because they're obviously high loads, but if you think about someone lifting weights, quite often, they lift the weight very slowly, but then they might drop it very fast. And in fact, there are experiments which have shown that the direction in which you applied these strains doesn't matter nearly as much as the speed with which they change. So it may well be that weightlifters who lift things slowly and drop them fast are getting more exercise for their skeleton by the dropping than by the lifting.
Tim - Weightlifting is good, it's a very interesting question because weightlifting is good because they're obviously high loads, but if you think about someone lifting weights, quite often, they lift the weight very slowly, but then they might drop it very fast. And in fact, there are experiments which have shown that the direction in which you applied these strains doesn't matter nearly as much as the speed with which they change. So it may well be that weightlifters who lift things slowly and drop them fast are getting more exercise for their skeleton by the dropping than by the lifting.
Kat - How is this actually happening? Obviously, there must be some kind of chemical signals from bone bending that's making the bones grow. How does this actually work?
Tim - Well, the bone is full of cells. If you take a cross-section of a piece of bone, the actual hard material has lots of cells all the way through it and these cells have processes which touch each other, and they communicate with each other. These cells, which are called osteocytes, are thought to be the cells which can sense the mechanical environment of the skeleton. So, when the bone bends, fluids flow through the bone and the cells are actually stretched and twisted and compressed. The combination of those two things means that the cells are able to perceive whether they're being loaded by more than they should be, or less than they should be, and therefore do something about that. If they see this sort of amount of loading that they're expecting, they'll send signals which keep the bone in the same size. If you do less, if you lie on a hospital bed or you go up into orbit in weightlessness as an astronaut, then that would be seen as low levels of signal, and that would lead to your bones being reabsorbed and taken away because you weren't using them very much. That certainly happens in astronauts, but generally from their legs, not their arms because they're pushing satellites around and doing stuff, but they're not doing very much with their legs. So they lose bone from the parts that aren't being used very much.
Kat - Now we're going to be talking later to Ken Poole who's going to talk about osteoporosis, the thinning of the bones, but you've mentioned that people who are in hospital or incapacitated in some way would lose their bone density. Are we trying to find out what these chemical signals are and is there any way of mimicking them? Maybe we could come up with some kind of drug that could make someone's bones mimic the effects of impact exercise without actually having to go to the gym?
Tim - I'm smiling at your question because that's exactly what I thought 25 years ago when I started doing my PhD and I thought after three years, I might have some of the answers. I have a very small number of them. It's certainly the key question, I think. We understand much more than we did about the signalling that goes on, but I think there are lots of things we don't know yet and one of the most interesting things, to me certainly, is that the skeleton can respond to incredibly brief periods of exercise. You certainly don't need to do it for very long and there are other things that suggests that the skeleton can actually remember for probably up to 24 or maybe 48 hours, things that have happened to it, and those exercises will potentiate the effect of subsequent exercise. So, to me, that's a very important thing because if we knew more about that, we might be able to make drugs that could either increase the length of that persistence of memory, or maybe make it more sensitive. As you say, potentiating the effect of a walk down to the shops, into what the skeleton might perceive as a vigorous trip in the gym.
Kat - Fantastic stuff! Thanks, Tim. That's Professor Tim Skerry from the University of Sheffield.
30:55 - Scanning for Osteoporosis
Scanning for Osteoporosis
with Dr Ken Poole, Cambridge University
Chris - Every year, about 75,000 people suffer a hip fracture in the UK and a majority of those will be down to the condition osteoporosis, that's the underlying cause of their bones being too weak. This is basically bone thinning as we age. Dr. Ken Poole is with us now. He's a rheumatologist at the University of Cambridge where he's studying how bones weaken and change with age, and then some new ways that we can try to use to combat the problem. Hello, Ken.
Ken - Hello, Chris.
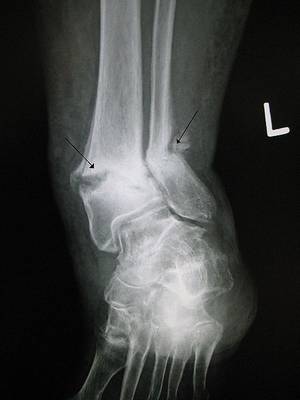 Chris - Thanks for joining us on the Naked Scientists. First of all, just fill us in, what actually is osteoporosis?
Chris - Thanks for joining us on the Naked Scientists. First of all, just fill us in, what actually is osteoporosis?
Ken - Well, exactly as you say. They're porous thin bones that fracture when they shouldn't. So, a lady walking along, falling over from a standing height shouldn't break a wrist, but in osteoporosis, you might, bending to tie the shoelaces fracturing a vertebra. So the little old lady, why is she little? Because of vertebral fractures, the bones collapsing. In the case of hip fracture, a trip or a stumble, or a fall, enough to break a hip.
Chris - If you were to take a piece of bone from someone who has got osteoporosis, how would it compare if you looked at it both macroscopically, just by eye, and then microscopically down a microscope with someone who's got normal bone?
Ken - Well, we do things like this in the clinic. So, when we take a piece of bone normally from people's pelvis, the bone has a thick outer shell and an inner honeycomb which is a mesh full of struts. In a young person, the outer shell is nice and thick and the honeycomb mesh is also thick, providing lots of support, like the inside of a Crunchy bar. If you have an osteoporotic patient, then the outer shell is very much thinner and the mesh of struts may not even be there at all or where they're there, they're thinned and they don't connect properly.
Chris - Do we actually know why this happens? I mean, are some people pre-destined to get this because of family reasons? Is there a genetic cause or is it purely just down to how much exercise you have or haven't had, or what your diet's been like earlier in your life?
Ken - Well those are all good points and fractures are multifactorial. So you have the low bone strength from the osteoporosis, but you also have falls, you also have genetic factors. One interesting factor is that if your parents have had a hip fracture, a mother or father, then your own risk of a hip fracture is higher as well. And coming into that, things like reflexes as well, so your ability to deal with the fall which gets worse as you get older.
Chris - What can we do about it? So someone is diagnosed with osteoporosis. They have a fracture of a wrist or something, they slip on the ice, what can we do once you've picked it up?
Ken - Part of that job is greater awareness and things like the National Osteoporosis Society is great at doing that. A risk fracture in an older woman is not something that you can leave, and you can ask some questions of that person and find out whether they've got very low bone density that can be improved by drugs. And we've got plenty of drugs now that actually help and as we've discussed earlier, the exercise will help too. And it's also dietary and even sunlight, things that can help with your bone strength.
Chris - Okay. Well, talk us through then, when you put someone on therapy, how are you monitoring them and taking this forward in the research domain, what are you actually doing to try to work out how to optimise therapy for this?
Ken - So, one of the things that we use at the moment is DEXA scanning. A lot of people have heard of this bone density scanning. That is where we put people onto a bed, look at their spines and hips, and work out how dense the bone is and compare it to a normal range. But we also know that half of people with hip fractures have a normal bone density result or one that doesn't come up as the osteoporosis threshold. So, one of the things we need is better ways of picking people up, and also, monitoring their treatments.
Chris - And how are you doing that?
| Osteoclast: |
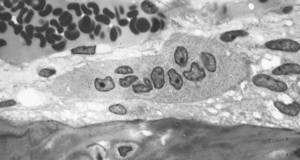 |
| Osteoblasts: |
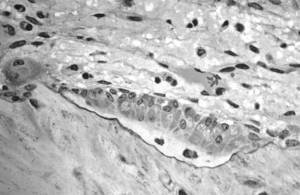 |
Ken - Together with some of the engineers at the University of Cambridge, we've developed a technique which is
based on the CT scans that Ruth dealt with earlier. It was actually interesting to hear that because we're using CT which is computer tomography scans of patients. We can now map the outer shell of femurs, the femoral bone, and produce a colour map of how thick the bones are, and it's very interesting to see that in older people, they're often as thin as an eggshell. My wife says that when you get old, your hips go pink because we use pink for less than 0.3 mm which is eggshell thin. We've done a recent study that shows that one of the commonly used osteoporosis drugs called teriparatide which actually makes bone grow, that's actually able to put bone in places, just like Tim described where you have peak strain in the femur which is a good thing because that's where they often tend to break.
Chris - Because that's where you want the bone to grow.
Ken - That's right.
Chris - Yeah. So, I mean, that was going to be my next point, which is - people who've got osteoporosis have lost bone mass, have actually lost bone substance. You want to put it back. How do drugs and doctors attempt to make people who've got thin bones grow new bone, and why don't people just do that anyway?
Ken - Well, there's drugs called bisphosphonates which are a lot of people are on, which are once weekly drugs usually, and they actually don't make new bone grow. They're the most commonly used drugs, but then you actually stop it being broken down.
As Tim said earlier, there's a process going on all the time of cells called osteoclasts, digging pits in bone, and osteoblasts, filling those pits back up. In osteoporosis, that's out of sync and we called drugs called bisphosphonates which actually stop the osteoclasts from digging pits, and allows the bone that's there to harden off, and that actually prevents fracture, and they're very effective. But right down on the other end of the spectrum, the osteoblasts that cause the bone to grow, there's a drug called teriparatide which is given to patients who are really in a bad way with osteoporosis at the moment. So they've got multiple fractures and our studies showed that when you give this teriparatide, a daily injection, it causes bone to grow in these particular sites of interest.
Chris - One of the stories I mentioned at the beginning was making hearts regrow and scientists are trying to make a heart that's in an adult, think it's in an embryo again because then it will regrow lost tissue. Could we do the same thing for bones and fool bones into thinking that they're back in a baby so they begin to grow new bone, and thereby, get rid of the osteoporosis problem?
Ken - Well, Tim said earlier about the vast amounts of material that might be unnecessary to carry around if you had, say a gene; And in fact, there is a gene, the sclerostin protein which is a funny name, but it's from a disease called sclerosteosis, and by examining that disease, scientists have been able to find just that, the off switch for forming bone. By giving people a monoclonal antibody against this protein, you can actually make bone form at amazing rates in all sorts of places, and that seems to be the most likely approach to make new bone in all sorts of situations where it's needed.
Chris - But that sounds a bit dangerous because you could end up growing bone where you don't want to grow bone, as in where you don't need it.
Ken - And that is actually one of the features of the condition, sclerosteosis, so bone is beautifully regulated and held back from doing silly things, and one of the silly things that it can do is, if it closes off the inside holes called foramen inside the skull, that's a bad place to have bone. The patients who have this disease actually close off things like their facial nerve cavity, their ear canals, and even the very large foramen magnum that carries the nerves, and they can die from this condition. So, it's all things in moderation and a lot of work has to be done to make those safe.
Chris - And just to finish off briefly, Ken. I suppose if you've got systems now that are enabling us to track where new bone is being laid down, if you could work out what the signals are that are active in that site, and then tell this gene which is the bone growth off switch to activate just in that site, then you'd have a way of thickening up the bone where it needs to be thicker, and leaving other sites like the sensitive ones you mentioned unharmed.
Ken - Working out those processes is absolutely key.
Chris - Ken, thank you very much. That was Ken Poole. He is an arthritis research UK clinician scientist at the NIHR Biomedical Research Centre in Cambridge.
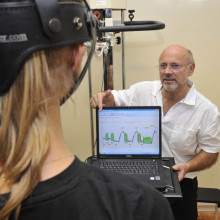
39:24 - Naked Engineering - Measuring Physiotherapy Success
Naked Engineering - Measuring Physiotherapy Success
with Don Gatherer, The Gatherer Partnership; Thea Maxfield
Kat - When muscles are injured or weakened, patients are usually referred to a physiotherapist for help, regaining their strength or improving their range of movement. But it can be difficult for the physio to accurately gauge the work load the patient can safely tolerate and the level of improvement that they've made. So now, physiotherapist Don Gatherer, who's previously worked with the England Rugby Team and the British Olympic Squad, has come up with an elegantly simple solution. And for this week's Naked Engineering, Dave and Meera have been along to meet him to find out how it works.
Don - Well the challenge is literally, objective data. A lot of our work is subjective. In other words, you can say, "Hey, how are you feeling? Is it feeling better? Are you stronger?" but we have no measurement for that.
Dave - I guess the other problem is that the human body is incredibly good at adapting to a problem and so, if you've got a problem in one place, it'll do all sorts of strange adaptations to compensate for it.
Don - Yes. That's exactly the case. The body is a really clever piece of engineering.
 Meera - The reason we've come along here today is because you've designed an objective way to calculate just what's going on in our muscles?
Meera - The reason we've come along here today is because you've designed an objective way to calculate just what's going on in our muscles?
Don - Yes. There are instruments out there but they're all fairly big and cumbersome and not portable, so my biggest problem was, how could I design a system that is very easy, portable, and will analyse all the actions very quickly.
Meera - Well you have one of your devices here, so it consists of a harness and a handle with a cylinder attached to it.
Don - Inside that cylinder, we have a load cell and these load cells were developed in F1 and so, I applied that technology to medicine because of the high quality of the load cell that's produced.
Dave - So a load cell is effectively a means of measuring force. It's the sort of thing you get in a set of electronic scales. I guess this is measuring a bit quicker and a bit more accurately.
Meera - And it's measuring the forces of our muscles.
Don - It will measure both the compression and traction forces. So it's a bidirectional load cell.
Meera - To see your device in action, we've also got Thea Maxfield here with us who is a horse rider, but also, Thea, you're a patient of Don's.
Thea - Yes. I came to see Don a year ago after I had an accident where I fell off my horse and I broke the C2 and C3 vertebra in my neck which is commonly called a hangman's fracture.
Meera - So Don, from a physiotherapist perspective then, what was Thea's case when she first came to you?
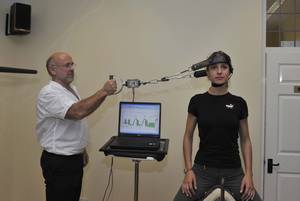 Don - Thea had obviously been given the all clear from the surgeon to start looking at exercise programs. So what we needed to do is to find out the objective data analysis of her neck. How strong it was, whether the muscles had wasted, peripheral profile, is there any weaknesses in her arms? So the idea of this is once we know how much force the neck can take, it means that we can then work below that force and apply a load to her neck to strengthen it, but without any risk of injury to the underlying fractures of the spine.
Don - Thea had obviously been given the all clear from the surgeon to start looking at exercise programs. So what we needed to do is to find out the objective data analysis of her neck. How strong it was, whether the muscles had wasted, peripheral profile, is there any weaknesses in her arms? So the idea of this is once we know how much force the neck can take, it means that we can then work below that force and apply a load to her neck to strengthen it, but without any risk of injury to the underlying fractures of the spine.
Meera - So, this is where your device comes in, measuring the forces that the muscles can take before they fatigue.
Don - Well that's it. We measure two types. We measure how strong they are and then we can measure how quickly they can fatigue.
Meera - Well to see how this is possible, Thea is primed and ready. She is sat behind one of your examination couches here. So how would you measure these forces?
Don - Yes. What we're going to do is connect the harness up to the load cell and then we're going to apply the harness to Thea's neck.
Meera - So her head's really encased in this harness and then there are two strings coming out to which the load cell is attached and you're holding the handle of this load cell.
Don - Yes. The two strings are really important because they're connected through a pulley and what this does is equalise the force on either side of the head so we don't get any shear or rotational forces during our analysis. What we do is position Thea into the correct position of the neck and then we apply a controlled force, and she'll hold that force. The test will continue until she loses her position. The moment she loses her position, we stop the test.
Thea - He's trying to pull my head forwards and what my job is, is to keep resisting against it and keep my neck and my head upright, and I can only do that so long as my muscles can manage it, and then I give, and I lean forwards.
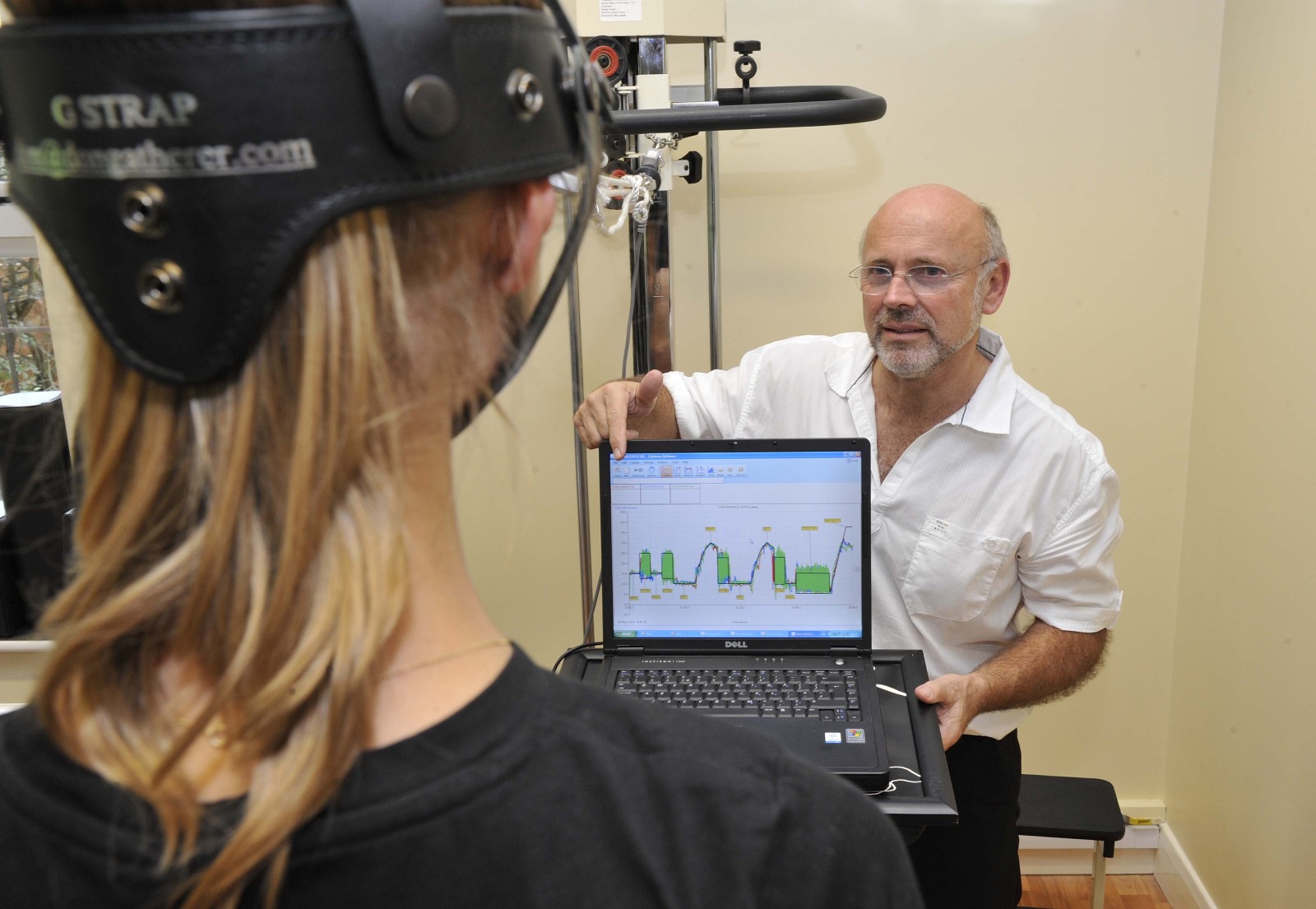 Don - Okay. Here we go. Right, hold, hold, good girl. And there we go. That's fine.
Don - Okay. Here we go. Right, hold, hold, good girl. And there we go. That's fine.
Meera - Thea, you looked quite strained there.
Thea - Yeah, it is quite physically demanding.
Meera - Don, how is this translating then? We've got a big screen behind us and you've selected for the muscles in Thea's neck to be analysed. So what are we looking at on this graph here?
Don - We have the graph for neck extension, so what's happening is that we tare or zero the load cell, so we have a zero rating, and then as we apply the force, you'll see the readout appear, and it'll measure the force as it's being developed. The moment she loses her position then we stop the force, we can then measure the peak and the area below that force.
Meera - I'm assuming you can test other areas in other muscle groups.
Don - We can test her whole body- shoulder, elbow, hand, fingers, knees, foot, toes - everything, hamstrings...
Meera - How is this all then put together to give Thea and other patients a treatment program?
Don - We'll now have a profile of the actions that were tested which means that we can then apply a safe load, whether that would be a stimulatory load for strength or endurance, and we can achieve our physiological goals because we know that we can apply that particular force but in a safe way. We won't fatigue the muscle, we won't then cause undue shearing forces like across her spine, which could irritate underlying structures like the spinal nerve, and so on.
Meera - So how would you summarise the real benefit of this system and this way of measurement?
Don - It is down in simple terms to objective analysis. We take the readings, we can use those readings and then from there, you can database, you can design training programs, but you can also look to see at your clinical effectiveness, if they are improving, if they're not improving, and then you can redesign your testing program. It all bases down to objective analysis.
Kat - That was Don Gatherer from the Gatherer Partnership and his patient, The very willing Thea Maxfield, talking with Meera Senthilingam and Dave Ansell about his new device to help physiotherapist to identify particular muscle groups in a patient that might be weaker. Particularly maybe on one side or the other so they can get special attention, workout what load they need, and workout if they've made any improvement.
What diet & nutritional factors are important for bone strength?
We put this to Dr Ken Poole and Professor Tim Skerry... Ken - Diet and nutrition is very important, and the one that people think about most is vitamin D. Calcium is also very important. We have not enough vitamin D from sunlight at these sorts of northern latitudes, so those are important. In order to work out how important, you can look at the body mass index and a low body mass index is a risk factor for fracture. That's one of the things that we plug-in to the World Health Organisation Frax tool which is a thing that predicts what your own fracture risk is. You can run this on an Apple, on an iPhone, or even just on the web from the Frax website and that will tell you what your own risk is over the next 10 years. So yeah, nutrients and diet are very important.
Chris - Sounds good. Anything to add to that, Tim?
Tim - I don't think so. I think generally, western diets have reasonably good levels of calcium, but yes, as Ken says, vitamin D could be something that people need to worry about.

47:21 - Are healed bones stronger than never-broken bones?
Are healed bones stronger than never-broken bones?
We put this to Professor Tim Skerry and Dr Ken Poole...
Tim - Well yes, actually. As bones heal, a sort of cuff of new bone forms around the fracture. So when that's healed and stuck together, the clinical union, which is a few weeks after it's broken, there's actually a big thick load of bone where the fracture was, so that would be stronger than the original bone. But over time, that thick piece will remodel away. It will be resorbed away by those osteoclasts that Ken was talking about. If you have a fracture as a child, you'll find that within a couple of years, you wouldn't be able to tell that the bone had ever been fractured. So eventually, the strength will go back to what it was.
Chris - Do you see this on scan, Ken?
Ken - Can we see it on a scan? Yeah, it's not one of the bones that we normally see with our DEXA scans which are normally, hip and spine. If there are forearm scans, we'd normally do the one that wasn't broken because we ask that question before people go in.
Why are steroid inhalers bad for bones?
We put this to Dr Ken Poole...
Ken - Yes. The steroids are one of the classic "bad for bone" drugs. It's normally steroid tablets that are worse than the inhalers, but there is a smaller effect of the inhalers. It's important that if someone is on a steroid inhaler like that, that they do all the lifestyle things like the diet and the exercise, and nutrition to ensure that they get the most out of their bones.
Why doesn't all cartilage become bone?
We put this to Professor Tim Skerry...
Tim - Well this is a question that goes to even more fundamental biology than about the skeleton particularly, because it's really the issue which is called patterning - how cells in your fingertip know they're fingertip cells, different from liver cells, different from eye cells. Cartilage cells which are cartilage that's going to become bone have some sort of positional information in their set of genetic information which tells them what to do. Whereas cartilage cells that are going to be at the surface of a joint have a different set of information. So, it's a patterning issue rather than just - they're not - all cartilage cells aren't the same.
Chris - There's a set of instructions written into them that tell them what to become basically.
Tim - Yes and knowledge about where they are.
How much force does it take to snap the femur?
We put this to Dr Ken Poole...
Ken - Normally, it's thought of in terms of the amount of energy available in a fall; and it's a surprisingly little amount of the energy: about a fifth to a tenth of the amount of energy in a fall is enough to break that femur.
With a hip fracture, there's such a thing as the perfect force. If you are unlucky and fall backwards and to the side - even if you're young - you can snap your femur.
Can we strengthen our skulls?
We put this to Professor Tim Skerry...
Tim - Well this is a really interesting question and it's a question that we're actively involved in researching at the moment because in fact, you don't need to strengthen your skull. It's much stronger than it needs to be for the amount you use it during the day and the whole concept of this is an interesting one because if you think about the safety factors of different bones, your long bones have a safety factor of about 4. That means if you bend them four times as much as they bend normally, you may break them. With your skull, it's about 40 and what that really translates into is that if you exercise and do things with your long bones, you bend them by about 0.2% With your skull, no matter what you do, you bend them no more than about 0.02% a 10th as much and we know this because we put strain gauges onto the skull and long bones of one of my PhD students a few years ago, so we know this is absolutely right. What it tells us is that the cells in the skull are either much more sensitive to load than cells in long bones or much less sensitive to the lack of load, and either of those would be a great thing to be able to understand. So we're comparing cells from different sites to try and understand the mechanisms behind that so that we might be able to find a drug treatment for helping people with osteoporosis.
Chris - To what extent does this reflect the embryology though because long bones form in a different way, a cartilage model gets replaced by bones, compared with skull which is formed by sheets of bone growing into tissue as the skull forms in development? So, is that a clue?
Tim - You're quite right. That's a really important question. It's certainly the first thing we thought of. That the axial skeleton, the skull, the spine, and ribs and bits of the pelvis, and the clavicles are formed from the same sort of thing. In fact, it doesn't really fit with that. It's not just embryological origin. Certainly, when you get those bones and you take the cells from them, they look very similar in adult humans and animals. And really, that doesn't seem to be the difference. There's something fundamental about the way the cells are embedded in the material and the way that they do the mechanical sensing that seems to be different, and that does seem to be a key for the skull, not just any old part of the axial skeleton, because the vertebrae are commonly affected by osteoporosis.
Chris - But you haven't flushed out exactly what yet?
Tim - Like all science, every question you answer raises another five that you need to do things with and we're in that position.
Is bone tissue replaced by fat as we age?
We put this to Dr Ken Poole...
Ken - Yes, it is true. The fat cells are part of the same lineage as the bone cells. So when we get older, we start making more fat cells and less bone forming cells, and that's an inevitable consequence within the bones.
Do bones stop taking calcium after 35?
We put this to Professor Tim Skerry...
Tim - They don't actually stop or they don't lose the ability to do that because actually, in our skeletons, there's about 2 million places in each of us, all the time, there's a bit of bone removal and bone formation going on. So at one of those little tiny sites, you can get new bone laid down, new incorporation of calcium. What happens is that the balance between the digging up and the in-filling shifts to be more digging up and less in-filling, but the ability to form bone and to incorporate calcium is still there throughout life.
Is there any difference between the bones in people from different races?
We put this to Professor Tim Skerry and Dr Ken Poole...
Tim - Well there's certainly some basis for that. Black people do have stronger bones than white people but I think there's more to it than that and I think Ken, probably, as a clinician has some additional thoughts about that.
Chris - Ken?
Ken - Well that's very interesting. The MRC Human Nutrition Team in Cambridge often work in the Gambia and the bones of people there may look like they're osteoporotic on the scan, but actually they're stronger, and that's one of the paradoxes. But it's one of those things that when using the Frax tool, which is a new way of finding out people's risk of a fracture, you have to make sure you're in the right ethnicity group because it's calibrated by a different nation.
Chris - That's interesting. So if you don't make an account or take it into account where someone comes from, you could end up with the scan, giving you a misleading result for the very reason Tim just mentioned.
Ken - That's absolutely right and there are numbers available for each of the countries that they've got data available at the moment.
55:10 - What causes muscle cramps, and are there any cures?
What causes muscle cramps, and are there any cures?
We put this to Dr. Steven Huan, Ashley Montagu Fellow in the faculty of Education and Social Work at the University of Sydney...
Steven - A muscle cramp is an involuntary and forcibly contracted muscle that simply does not relax. When you use your muscles, you can control them voluntarily, in your legs and arms for example, and they can contract alternately, they can relax and then contract. But when you lose control of this, this is the muscle cramp. Now, about 95% of humans have muscle cramps, have experienced them at least sometimes, some more than others and it happens particularly in old age, but even children can have muscle cramps.
Now what do we do to prevent them? Well you have to be very careful about warming up before exercise, you have to be very careful that you don't become dehydrated. That's why, when you see for example, tennis players getting leg cramps, that's because they're losing a lot of fluid when they're on the tennis court, especially in very, very hot weather. So you have to remain hydrated.
You also have to have a high level of potassium in your body because if you have low potassium, sometimes this can cause the muscle cramps, but also, low potassium can be associated with muscle weakness as well.
Diana - Sometimes, muscles over-shorten themselves and this can be caused by low sodium, low potassium, dehydration, hypoglycaemia, and even hypocalcaemia, which is a lack of calcium. So, maybe try having a drink and something to replace whatever nutrients you might be missing. While sometimes it happens because the joint has been flexed too much and you just have to straighten it out or walk it off.










Comments
Add a comment