This week, how rogue antibodies turned one woman's existence into a living nightmare of delusions, hallucinations and paranoia, we examine the evidence that ME - or chronic fatigue syndrome (CFS) - might be an autoimmune disease, and why the blues might be down to a hostile immune response. Plus, how tracking eye movements can be used to influence decisions, why remembering causes you to forget, a new 3d-printer inspired by Hollywood's Terminator, and the genetic map of the UK: apparently the Romans didn't fancy breeding with us very much...
In this episode

01:17 - Eye tracking can alter decision-making
Eye tracking can alter decision-making
with Daniel Richardson, UCL
Killing people is bad, right? And surely lying is wrong? We all like to think that we make good judgements based on our inner moral beliefs, and that these are strongly held and unshakeable. But it turns out that our morals may be more wobbly than we'd like to think, according to new research from Daniel Richardson at UCL, where he's been using eye-tracking devices to manipulate peoples' choices, as he explains to Kat Arney...
Daniel - We were interested in a process that people go through when they're making difficult choices. In this case, we looked at moral decisions because they are things that we think are permanent about ourselves. There a permanent things about our values, and our principles, and our ethics, and even our religious beliefs. We wanted to see if you make those decisions in the same way that you make a simple choice like whether or not to have a salad or a hamburger for lunch.
Kat - How do you go about figuring what's going on when someone's making a decision like this?
Daniel - Well, what we did is we looked first at some of the previous research on eye movements and show that if we asked people - if I show you two pictures of people and say, "Who's the more attractive one? Who would you like to date?" what we find is that people look back and forth between those two alternatives. But their gaze will centre around the one that they're going to eventually decide. We can look at people's eye movements and that gives us a little window into the process of their decision. So, before they actually decide, we can tell which one they're leaning towards.
Kat - Their eyes are giving it away, maybe before they've even really figured it out.
Daniel - Yeah. The eye movements are a window into this cognitive process. Well, if we have this window into people's decision making process, can we use that information to change their minds? Can we use it as a tool to persuasion, given that we've got this magic insight into what people are thinking?
Kat - So, how do you go about doing that then using this information about where people look to even manipulate what they're going to say, what they're going to think?
Daniel - So, what we do is, a phrase pops up on a computer, our moral dilemma. Something like, "Is lying always wrong?" And on screen there are two options. You can only have two choices, yes or no or sometimes or always. And then we eye-track people as they look back and forth between these two choices. But what we did was randomly pick one of those beforehand that we're going to try and bias people towards. So, say I wanted you to say, "Yes, murder sometimes is justifiable." What the eye-tracker does is as you're looking back and forth between these two, it waits until you're looking at the one that we want to bias you towards. So, the system waits until you're looking at the word 'yes' and then suddenly, a thing pops up in the screen saying, "Decide now!" What we found is if you make that decision at the moment that we tell you to, at the moment that you're currently considering that choice, you're more likely to pick that choice.
Kat - How do you know that you are actually influencing someone's choices? They're not just picking the thing that they would've picked anyway, that you can actually change what someone's thinking.
Daniel - Well, that's a very good question and sometimes of course, maybe you do have a very strong belief about this particular moral question. So, what we do is we randomly choose these targets. We're going to bias you towards a yes, or a no for every person. We do this with lots of people on lots of different trials. So sometimes we try and bias you towards saying, "Yes, murder is justifiable." Sometimes we bias you towards saying, "No." For any one person, perhaps they did have a strong decision one way or another. When we look over lots of people we average, we find that on average, we get these increases on people picking the target that we bias them towards. So, we know that we're having this cause and effect on people's decisions because we can change them one way or the other, and we can do it at the toss of a coin.
05:10 - Remembering makes you forget
Remembering makes you forget
with Maria Wimber, Birmingham University
The process of recalling memories also involves actively forgetting things. Researchers at Birmingham University asked a group of volunteers to memorise pairs of things, but then to recall just one of the pair,  forcing their brains to suppress the memory of the other item, which ultimately led to it being forgotten, as Maria Wimber explains to Chris Smith...
forcing their brains to suppress the memory of the other item, which ultimately led to it being forgotten, as Maria Wimber explains to Chris Smith...
Maria - Imagine you're standing in front of a cash machine and you just got a new bank account. So, you're desperately trying to remember your new pin code but the old one that you've used for a couple of years keeps popping up back in your mind. You can probably imagine that in those situations, it's quite useful to have a mechanism that helps us forget the old pin code in order to better remember the new one.
Chris - So, what is the mechanism that's at play to solve that so that I can remember my pin code despite having lost my card and had it replaced - God knows how many times.
Maria - What we show in the study is that the brain has a mechanism of actively getting rid of information that constantly blocks our access to the information that we're really trying to recall. Like in the example of the bank account or the bank pin, it's trying to get rid of the old pin in order to make it easy in the future to retrieve the new pin.
Chris - Are you saying then that by remembering one thing, I am actively forgetting another?
Maria - Absolutely, yeah. I think it's important to mention that it's an active process that is happening. So, it's not just that we passively forget the information that we don't use, but remembering actively shapes which information we retain in long term memory and which one gets lost.
Chris - Is that an irreversible loss?
Maria - In the long term, it might be, yeah.
Chris - Gosh! That sounds a bit worrying. How did you find this?
Maria - New developments in brain imaging have made it possible recently to decode just based on somebody's brain activity what picture for example somebody is looking on right now.
Chris - And this is because you have shown those people those pictures before and looked at what their brain did when they were looking at those pictures. So, you can, in that person, say, with that pattern of brain activity correspond to them viewing a picture of Einstein or Marilyn Monroe.
Maria - Absolutely. We show people the same pictures over and over again. So, we can sample something like the prototypical fingerprint that Albert Einstein leaves in the brain. The challenge for us was then to ask, well, using these neurofingerprints, can we even try to decode what somebody is thinking of, or what somebody is recalling from memory at a given time? And the answer is yes. We were able to do that and the studies that we were able to track individual memories as they come back in the brain when people are remembering something. So, we trained people to create certain links in their memory, links between words and different kinds of pictures. So, we knew that each of the keywords that we were using would elicit two different pictures in their brains. So, the word 'sand' for example might have been linked to Barack Obama, but also, to a picture of a rope. So, when we ask people to remember only the face that had been linked to this word, we could actually watch whether their brain was able to bring up the face of Barack Obama and at the same time, to suppress the picture of the rope that is interfering or distracting them at this time.
Chris - I see. So, in other words, when you're saying to them, "We only want the face" but because the word you're cuing them to remember the face with is also linked to another memory, the brain needs to be able to suppress the rope so that it doesn't interfere with them thinking about Barack Obama and you can actually see that happening.
Maria - Exactly.
Chris - How do you know that the brain is suppressing the image of the rope?
Maria - So, it's not just the neuro-fingerprint of the rope is more and more fading out. But the picture of the rope was being suppressed for example below the picture of a hat that wouldn't interfere during the task.
Chris - The brain is actively deactivating that fingerprint that would correspond to the picture of the rope in the minds of these people. So, you see a very strong representation of Barack Obama, but the representation that would normally correspond to the rope is being more and more eroded.
Maria - Absolutely, yes.
Chris - Goodness and so, that suggests that the brain has actually actively unwritten the memory of that association.
Maria - It sounds a bit scary but yes, that's probably the essence of it.
Chris - What would be the implication of this then in everyday life, apart from actively, when you learn something new and you overwrite a memory, what other implications might there be?
Maria - Most people tend to think of forgetting something that is like a failure of a memory system. But I think in most situations, forgetting is actually something incredibly useful. To give you a real life example, this forgetting might become really relevant when you're studying for exams. It's often recommended that you test your own memory often enough. So, you read a chapter in the book and then you use for example flashcards to test your own memory. This is actually a very good idea. Testing your own memory makes your memory better and more stable in the long term. But it can also make you prone to this negative side effect of remembering which is forgetting related information. So in simple words, when you study for an exam and you use flashcards to test yourself, try to do this comprehensively. So, test yourself on everything you have just learned and don't leave out any information because by testing yourself on some pieces of information, you might actually lose other pieces of information.

11:04 - Fast 3D Printer inspired by Terminator
Fast 3D Printer inspired by Terminator
with Professor Joseph DeSimone, University of North Carolina & Carbon3D
But first, a revolutionary form of 3D printer inspired by the Hollywood blockbuster "Terminator 2" has been developed by researchers at the University of North Carolina and the company Carbon3D. Rather than painstakingly adding details a layer at a time, which is the conventional approach,  the new system, called CLIP, uses UV light to project a movie into a bath of liquid material; the UV causes the material to solidify rapidly into the desired shape. Georgia Mills spoke with co-inventor Joseph DeSimone...
the new system, called CLIP, uses UV light to project a movie into a bath of liquid material; the UV causes the material to solidify rapidly into the desired shape. Georgia Mills spoke with co-inventor Joseph DeSimone...
Joseph - We were inspired by the scene out of the Terminator 2 movie with a T-1000 arising out of a liquid puddle. And so, we thought, "Why couldn't we get a 3D printer to operate in a manner suggestive of what Hollywood inspired us to think about?" and so, the idea of, could we have a complex 3-dimensional object arise out of the puddle - continuously - and do it rapidly with essentially no waste to make a great object?
Georgia - You mentioned Terminator 2. I watched the video of this printer. It's fantastic, but the film it made me think of was Mary Poppins when she brings these giant things out of a tiny bag. The layer just rises up slowly and this giant object way bigger comes out of it. It's amazing! So, how does this work?
Joseph - What we're basically doing is we're harnessing both light and oxygen to grow the parts. We have a reservoir that maintains a puddle of liquid resin and underneath that is a special window that's permeable to oxygen and transparent to light. And then we lower a stage into the puddle. It gets very close to the window and then we start playing basically a movie. Underneath the window, we project it and it's done in the ultraviolet region of the spectrum. We'll go through very quickly a range of images, the speed of which is coordinated with the elevator that lifts the object out of the puddle. We basically grow these objects very, very quickly with no layers and that way, we can have some parts that have amazing mechanical properties - they look like injection moulded parts. And so having a part come out of a 3D printer that has the properties to be a final part and do at a game changing speeds really opens up 3D manufacture. Our printing speeds go 25 to 100 times faster than traditional 3D printing. So literally, minutes instead of hours and tens of hours is what's possible with this breakthrough.
Georgia - Is this going to scale up? Can you make very large objects? What about the resolution? Can you make really tiny ones as well with the same method?
Joseph - Yes. So, we think we can. Our first machine is going to be in a sort of intermediate size range and 4 inches by 7 inches. Ultimately, we can make that 4 times bigger. But there's a lot of opportunity when one actually goes smaller. And the feature sizes that are relevant smaller are pretty cool. Because we don't have to do layers and because of the gentleness of our process, it's a really powerful tool for fabricating things from tens of microns to thousands of microns with complex geometries. And so, we think this opens up new sorts of sensor technologies. Think about the accelerometers and our health bands, and our automobiles, all sorts of lab on a chip applications and even new drug delivery technologies that we're very excited about.
Georgia - So, you were turning a liquid into a solid essentially. Is there any way it could be turned back?
Joseph - You've been watching too many Hollywood movies. Yeah, that would be very cool. Right now, our process is only the production of the T-1000. We don't have the ability to have them go away yet. Let's work on that.
15:37 - Reprogramming leukaemia
Reprogramming leukaemia
with Ravi Majeti, Stanford University
Leukaemias are cancers of the white blood cells that we rely upon to fight infection. One of the most common forms of childhood leukaemia is called ALL - acute lymphoblastic leukaemia. Normally, doctors treat the disease by giving drugs to destroy the stem cells  that are producing the leukaemia cells, and sometimes this is followed by a transplant with healthy bone marrow cells to restore the patient's immune system. But this is a complicated and risky procedure. Now scientists at Stanford University, in the US, have discovered that, by administering a certain cocktail of growth factors, they can trigger cultured leukaemia cells to switch from cancer cells into healthy immune cells called macrophages, as Ginny Smith heard from the study author, Ravi Majeti...
that are producing the leukaemia cells, and sometimes this is followed by a transplant with healthy bone marrow cells to restore the patient's immune system. But this is a complicated and risky procedure. Now scientists at Stanford University, in the US, have discovered that, by administering a certain cocktail of growth factors, they can trigger cultured leukaemia cells to switch from cancer cells into healthy immune cells called macrophages, as Ginny Smith heard from the study author, Ravi Majeti...
Ravi - When we took leukaemia cells, we were able to turn them into macrophages. Most importantly, those macrophages no longer caused leukaemia when we tested for that in the laboratory. It was a very surprising and unexpected finding because we didn't anticipate that the cells could change from one to the other.
Ginny - So, how have they done that?
Ravi - The details of that biology are things that we're still working out, but we were able to do that by using growth factors.
Ginny - So, these are chemicals that are found in the bone marrow normally, that affect normal development of cells.
Ravi - That's correct.
Ginny - And you were applying these to the leukaemia cells and managing to make them turn into something else.
Ravi - Exactly. We were trying to apply them to the leukaemia cells to see what would happen, essentially.
Ginny - Effectively, leukaemia cells are cells that have never grown up, that have kind of got stuck in childhood and you're now managing to make them grow up. But not just grow up and become the cells they would've become but actually, become a different type of cell. Why is it beneficial to have them turn into these macrophages? What do they do?
Ravi - Well, macrophages are important cells of the immune system. Their name actually comes from the Greek origin. So, the "macro" means big and the "phage" means eater. They're big eaters. They're very large cells. Their normal function is to ingest and digest bacteria or other foreign particles that shouldn't be in the body. But they also have a second function besides just eliminating things. The second function is then to simulate the T-cells and the other lymphoid cells to develop immune responses against those bacteria. So, not only do these macrophages not have the leukemic properties of cancer, but they may also be able to stimulate the immune system to fight the cancer directly.
Ginny - So, you would not only be getting rid of the cancer but perhaps giving the patient the means to fight back even better.
Ravi - Well, that's what we hope. It could be a two-pronged approach to fighting the cancer.
Ginny - Where are you in the sort of research stage? Are we looking at possible drugs in the near future?
Ravi - I would say that we are at the very preliminary stages of this type of investigation. This is the first record on this mechanism of turning cancer from one cell lineage into another and then potentially, stimulating the immune system to respond. We have only been able to accomplish these in the Petri dish in the laboratory. We're working now on two primary fronts. Number one is to try to identify drugs that might be able to cause this cell transition in an animal and then hopefully then in a human. But we're also working to try to develop methods in which we can make this transition happen, not just in the Petri dish but in an actual animal model.
Ginny - So, that's your next step to try and do this actually in an animal and cure it of leukaemia.
Ravi - That's exactly right. Now, we may use a variety of different methods including drug screening and other types of animal studies. But that's the real next step of translating this into a finding that it could have some impact on patients.
Ginny - Are there any risks with this type of therapy? Could you end up making their immune system overactive and perhaps giving an autoimmune disease?
Ravi - That is a very interesting question and I think it's a possibility because when you're trying to stimulate immune responses, you can both get responses against things that shouldn't be there as well as responses against the normal host. But we won't know that until we move into animal studies and that's definitely the type of safety in animal testing that will be necessary prior to thinking about any approaches to the patients.

20:10 - The genetics of the UK population
The genetics of the UK population
with Garrett Hellenthal, UCL
Who do you think you are? Tracing your family history is becoming more and more popular, but as well as digging through dusty parish records, our genes can tell us a lot about where we've come from. Now a major new study has used genetic analysis to look at the ancestry of British people,  showing that different parts of the UK seem to harbour their own distinctive genetic flavour and helping to reveal where some of these groups might have originated from, hundreds of years ago. UCL's Garrett Hellenthal, who was part of the study, explained how he and his colleagues traced our country's family tree.
showing that different parts of the UK seem to harbour their own distinctive genetic flavour and helping to reveal where some of these groups might have originated from, hundreds of years ago. UCL's Garrett Hellenthal, who was part of the study, explained how he and his colleagues traced our country's family tree.
Garrett - We sampled some 2,000 individuals from across the United Kingdom. There was a requirement for each individual; all 4 of their grandparents are to be born within 80km from one another. So, trying to get individuals that have been living in a region for a while so we can get a snapshot of older history and void recent migration. And the aim of the study - so, two major things that we did - one was to see if we can cluster individuals based solely on looking at their genetics and seeing that for example, individuals that were sampled from one part of the United Kingdom, are they more similar to each other than they are to individuals sampled from another part of the United Kingdom. And how does this correlate with kind of known boundaries in history. The second thing was to take the DNA of these same individuals and compare them to some 6,000 individuals from across continental Europe and identify if different parts of the UK, share matching DNA patterns with different parts of Europe. And if so, do those correlate with known migrations from Europe and of all the migrations from Europe into the UK which have kind of had the biggest genetic impact in the people in the UK today?
Kat - Tell me some stories about us. What have you found?
Garrett - Just by comparing DNA of individuals in the UK to each other, there was kind of a striking correlation - it turns out - with genetics and geography, to the point which we can tell whether an individual came from Cornwall which is in the southwest of England versus whether they were sampled from one county over in Devon. Genetically, we can tell them apart which is quite surprising because geographically, those are quite nearby regions. In fact, the difference between the two groups seems to be separated largely along modern day county borders which is another sort of interesting finding. But then at the same time, there's some parts of the UK and for example, central south and eastern England, individual sample from across very large swath of those areas, all were genetically very homogenous. We couldn't really tell them apart, suggesting that there had been quite a bit of movement and intermixing along those regions. And so then when we try to take the DNA of these individuals and compare them to Europe to learn about why we're observing these sorts of pockets of genetics across the United Kingdom, we found that one of the bigger stories in UK history appears to be the Anglo-Saxon migrations. So, UK has a very strong history, lots of migrations. For example, the Romans occupied big parts of England for about 400 years until about the 5th century. And yet despite that they've built lots of walls and baths and other things that you can see across England today. But in spite of that, there seems to be very little genetic impact at all related to the Roman Empire as far as we can tell.
Kat - They didn't fancy us basically.
Garrett - Yeah. It is believed that they didn't really migrate here in large numbers. Perhaps it was difficult to convince people to leave the Mediterranean and come up to the UK. In contrast to that, after the fall of the Roman Empire, that kick started what were known as the Anglo-Saxon migration's in the 5th and 6th centuries. And for those migrations, there's large scale migrations from places in modern-day Denmark and northwest Germany who settled into a big area within southeast England. It was unknown amongst archaeologists as to whether the Anglo-Saxons completely displaced the people that were there so that if you looked at an Englishman today, basically, they'd be entirely Anglo-Saxon in heritage or whether they intermixed with the inhabitants that were there. And so, what our study threw up was that they appeared to have intermixed. If you look at an Englishman today on average in these regions, this area of the southeast England, they have about 10% to 40% of their DNA that seems to trace to these Anglo-Saxon migrations. And the rest seems to be similar to other areas of the UK, which we think of as the kind of pre-Saxon inhabitants.
Kat - There's quite a lot in the news about immigration coming into the British Isles. Do you feel that when you look at the genetic history of the people who live here that this has been going on for many, many years?
Garrett - Yeah, that's right. One of the things that I've found in my studies is that every group appears to be mixtures of other groups. So we basically all descend from this sort of passing or mixing events. Human populations, clearly, once we expanded across the globe, we didn't just stay put. We went back out again and intermixed. And so, you see these signals everywhere. It's a constant interaction amongst different groups and that's still continuing today and will for a long time.
25:19 - Immune attack on the brain
Immune attack on the brain
with Belinda Lennox, Oxford University, Susannah Cahalan, writer.
It's becoming increasingly clear that the immune system plays a role in a number of nervous system disorders, including schizophrenia, depression and Chronic Fatigue Syndrome. Belinda Lennox, from Oxford University, 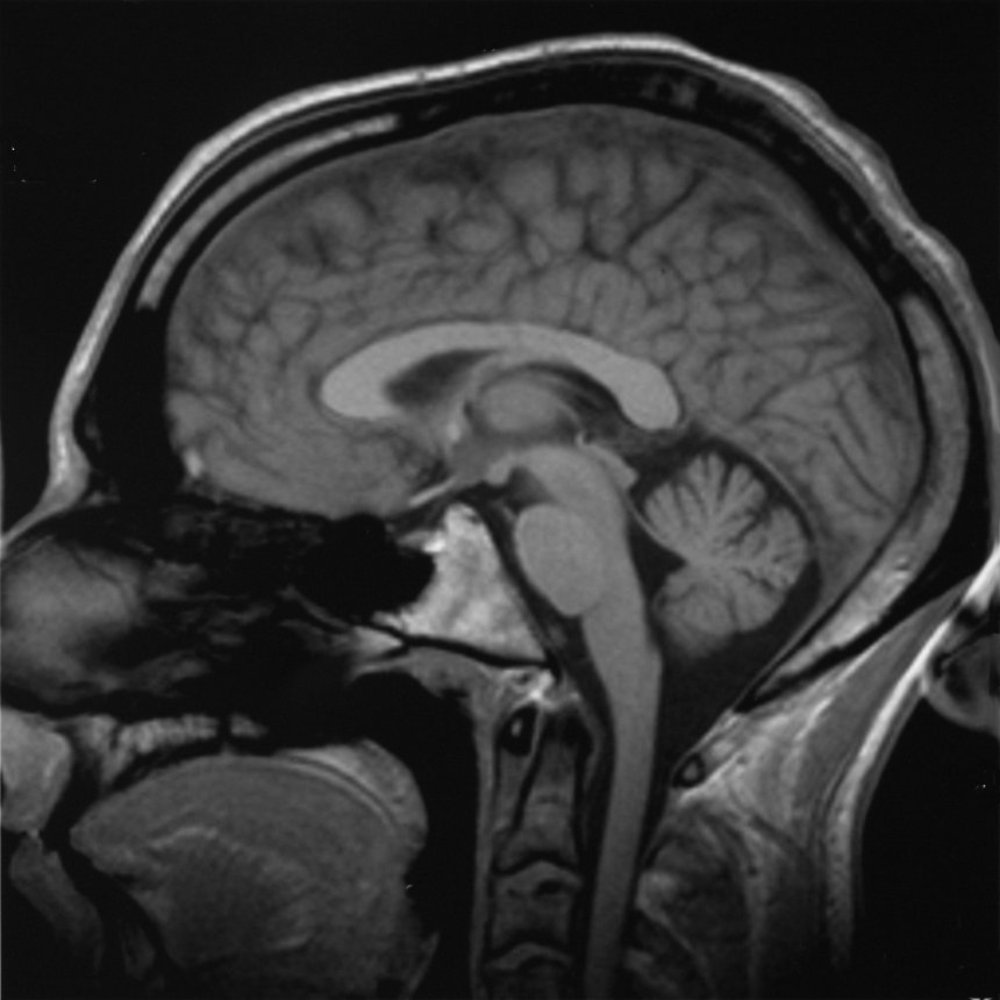 has been studying patients who present with psychotic symptoms caused by an immune attack on the brain. Kat Arney hears from her what causes episodes like the one suffered by New York journalist Susannah Cahalan, heard here speaking to Hannah Critchlow...
has been studying patients who present with psychotic symptoms caused by an immune attack on the brain. Kat Arney hears from her what causes episodes like the one suffered by New York journalist Susannah Cahalan, heard here speaking to Hannah Critchlow...
Susannah - This was 2009 that my symptoms started and I was 24. A lot in my life was very new. I had just started a full-time job as a news reporter, new apartment, new boyfriend, kind of living on my own in New York City for the first time. I started to feel not like myself. At first, I just kind of chalked it up to the fact that I'm in this new environment. I have this new life. Of course, I don't feel like myself. So, I felt very lethargic, I couldn't really concentrate at work. I was kind of moody. I was sleeping a lot. I became kind of obsessive about certain things, for example, I thought that I had bed bugs. An exterminator came in and said I didn't but I was obsessed with it anyway. I became convinced that my boyfriend was cheating on me and kind of went through all his belongings. I just thought, you know, I'm just not feeling like myself because I'm in the middle of a new life and that's what I chalked it up to in the beginning at least. But things quickly escalated from there. So, from just a month of not feeling of myself, all of a sudden, I was not myself in an extreme way. Pretty much overnight, I stopped sleeping, I stopped eating. My emotions which were already kind of influx went wildly out of control. One moment, I would be crying and then the next moment, I'd be laughing, I'd be on top of the world like the best I've ever felt. It was very confusing for me.
Hannah - And then you were almost sectioned to a psychiatric hospital.
Susannah - What happened was, I had my first seizure. I emerged from that extremely psychotic. Paranoia, I believed everyone was out to get me and then I began to hallucinate shortly after. So, I was actually seeing and hearing things that were not there. I believed that my mum had hired actors. When we returned to the doctor's office when I was released from the hospital, I believed she had hired everyone there as actors because she wanted to play a trick on me. She wanted to teach me a lesson. At one point, I was at my father's house, the room was kind of coming alive and the paintings were moving. The room almost had this energy like it was breathing. I remember as clear as day, hearing my dad hitting my stepmother. And again, this is all in my head, but this was as real as anything in my life. I thought he was going to come and get me next. I remember looking out the window. He's about 3 floors up and I looked at the window and thought, I can jump and escape. My stepmother keeps a Buddha statue in the bathroom and it smiled at me. So, I did not jump. After that night, my parents were very concerned about my safety. The doctor was worried that I was suffering from partying too much. My parents were adamant. They said, "No. there's something seriously wrong here, she needs to go to a hospital" and they won.
Hannah - Sounds absolutely terrifying - the whole experience - for yourself and also, your parents. So, what happened then? Did they reach a diagnosis quickly?
Susannah - I was in the hospital for a month, about 3 and a half weeks before we got a diagnosis. My clinical picture changed starkly. So, it started with extreme psychosis and I got even more psychotic when I went to the hospital. I saw myself on the news, I was trying to escape, I was ripping out EEG wires from my head, and IVs from my arm, and I had to be restrained and heavily sedated. My symptoms began to change and this actually scared my parents more than the psychosis because I started to become catatonic. I stop really speaking, my tongue hung out of my mouth. I just wasn't really there. At this point, what I'm relating to now is all from my parents and my medical records. I don't have any memory of this. But every single test that was conducted, they all came back negative. So, on paper, I'm a healthy person but they still did not have a cause. They couldn't figure out what was wrong with me. And then about 3 weeks in, a man named Souhel Najjar was called on the case.
Hannah - You had one little test I believe to help with your diagnosis.
Susannah - Yeah. It's incredible to think about it. He asked me to draw a clock, very simple. He kind of came up with this idea on the spot while he watched me do it. I had trouble forming the circle, I had trouble with the numbers and I would write them several times. And then when he looked down at my finished result, the whole left side of the clock was completely blank. That showed him the right side of my brain was very impaired.
Kat - So, that's quite a distressing picture that we've heard from Susannah about her symptoms and the clock test she was asked to do sounds like almost something off the TV show "House". So, how would people be diagnosed in this kind of situation if everything is coming up not right? What's going on here?
Belinda - Well, it was almost by chance really that Souhel Najjar thought that Susannah had something atypical because to all intents and purposes, her story was typical from my perspective of a psychotic illness, of a severe Schizophreniform, kind of acute psychosis. There was nothing in there to really raise alarm bells that there was anything different about her presentation. The test of the clock face is just a test of visuospatial processing. It's a bedside cognitive test. It's not very specific, but it does, I mean, he's right; it indicates that there's something going on in the brain if you want to be as crude as that. But he took that to mean that she needed further investigation and he looked further.
Kat - So then what was going on in her brain and how was it diagnosed?
Belinda - So, she had NMDA receptor antibody encephalitis and she was...
Hannah - Wow! That sounds complicated.
Belinda - It's a long name, but it's an antibody against a very common brain protein, the NMDA receptor that is fundamental for a whole range of brain functions. The antibody was only first described a year before Susannah started in her illness in 2008. So, she was incredibly lucky that Dr. Najjar had heard of it and actually, it was only after extensive investigations and a brain biopsy that they thought of testing for this antibody because all other investigations were completely normal. That's what we see in patients with this disorder; all other investigations are negative usually. And patients usually receive the diagnosis of Schizophrenia or a psychotic illness prior to having a positive blood test for this antibody.
Kat - So, let's unpick this a little bit. You're talking about an antibody. What is this antibody? It's something made by the immune system, so what's going on?
Belinda - Well, antibodies are produced by everybody. They're normal response usually to infection or to tumours or to other insults to the body. But sometimes they're produced against our own organs just as what we term an autoimmune disorder. This disorder was originally described in association with benign tumours - teratomas usually and the antibody was described in young women usually, with their ovarian teratomas. That was thought to precipitate the production of these antibodies. But since then the sort of the description of the disorder has broadened so that usually, we don't find a teratoma and we're assuming that this is an autoimmune disorder. The body is producing these antibodies against itself.
Kat - Now, this presents a bit of a problem because as with any autoimmune disease, your body is doing this against your own tissues. How do you go about treating something like this?
Belinda - Well very crudely, we suppress the immune system. So, we stop the production of antibodies. We give steroids and we also flush out the antibodies that are already in the system using treatments such as intravenous immunoglobulins or plasma exchange which is...
Kat - So, you kind of wash someone's blood basically.
Belinda - Absolutely. You flush out the antibodies that are there already and you replace them with plasma that doesn't have antibodies.
Kat - Well, Susannah had a treatment. So, let's find out from her how she faired on it.
Susanna - It took me anywhere from about a year and a half to 2 years to kind of return to base level of who I was before. In terms of the way I see the world now, I mean, it's changed everything about my life. You know, a big part of my reason for existing now is to kind of talk about this disease and others like it. I'm very interested in mental health issues and the idea, and the stigmatisation of mental illness. I mean, all these questions have emerged in my life that did not exist prior to having this illness. I think you can't really go through something this traumatic - this frankly interesting - I mean, it's a fascinating disease - and not have a change in your world view...
Kat - It is certainly is absolutely fascinating. So, Belinda, Susannah said things have started to go back to normal, it took a long time. But given what we know about how the immune system is a bit of an unknown quantity, could there be a problem with this disease returning?
Belinda - Absolutely. So in the cases where we don't find a teratoma or an obvious cause for the antibody being produced, there is certainly a risk of recurrence. Although we've only been seeing patients with this disorder for the last 7 years or so, we are seeing in about a third to a half of cases, we see recurrences and relapses. So, we need to suppress the immune system for a good couple of years. That's our current thinking anyway to keep people well.
Kat - When people think about psychosis, I mean, you do think about Schizophrenia and there are other psychotic conditions. But do you think that this kind of thing could be underlying maybe other types of psychosis or other sort of very severe mental disorders?
Belinda - Absolutely and even Susannah's story, she had a very prominent effective component, a mood component to her illness. Alongside her psychotic symptoms, she describes a wildly fluctuating mood state as well. And so certainly, from the cases that we've seen, there's a mixture of symptoms. So, it's not restricted to Schizophrenia-type presentation at all.
Kat - We'll be going on to look at some other mental health conditions that might be linked to the immune system. But what it wanted to pick up on briefly with how Susannah says, she wants to de-stigmatise the disease and I think with physical illness, you can kind of say, "Okay, that's wrong with you. You've got a cancer or you've broken your leg" but are we starting to now understand some of the actual, the physical things that are going wrong to cause mental health symptoms and that we should say, these are illnesses just like physical illnesses?
Belinda - Absolutely and some of the work that we've been doing has described that about 5% to 10% of people presenting with a first episode of a psychotic illness presenting to psychiatric services have these antibodies against the NMDA receptor and other neuronal cell surface proteins. So, it's vitally important that we look for these antibodies because it could result in a dramatically different treatment approach for these patients.
Kat - And this is one particular antibody. Where next? Are you looking for others that might be involved?
Belinda - Yeah. So, there's a whole range of different antibodies against different cell surface proteins that have been described. So, the voltage gated potassium channel antibody, GABA receptor antibody, glycine receptor antibody - I could go on.
37:29 - Is ME an autoimmune disease?
Is ME an autoimmune disease?
with Øystein Fluge and Olav Mella, Haukeland University Hospital, in Bergen
Chronic Fatigue Syndrome (CFS), also known as ME, affects about 1 person in 500. Sufferers describe symptoms of profound tiredness and lethargy that doesn't improve with sleep or rest. There's currently no consensus on what might be causing the condition, there are no tests to confirm the diagnosis, and nor is there a cure. But now we may be closer to understanding what causes at least some of the cases of the condition thanks to research carried out in Norway, as Øystein Fluge and Olav Mella from Haukeland University Hospital, in Bergen, explain to Chris Smith...
Øystein - We are oncologists that work mainly on lymphomas and brain tumours. But in 2004, we observed a patient with longstanding ME who got lymphoma and she experienced a totally unexpected and very marked recovery of ME symptoms after she received lymphoma treatment with cancer chemotherapy. We speculated on this case and when we met new ME patients, it was striking to learn how similar the patients were in symptoms and how they described to be previously completely healthy and often, with an abrupt start of ME after infections. So, we reasoned that B-cells could be important in a subgroup of ME patients.
Chris - When you say, B-cells, these are the white blood cells, the lymphocytes that make say, antibodies and have a memory against infections we've seen before, aren't they?
Øystein - Yes, but we did a small pilot case series with a single infusion of the drug rituximab which targets these B-cells to 3 ME patients and they all had a marked but transient clinical response. We published this case series in 2009.
Chris - So, you were giving these people in the course of treating their blood cancer, their lymphoma, the drug rituximab which hits and destroys B-cells in the body which are in these patients, the cancer cells. And as a side effect almost of that treatment, these people who had previously said they had disabling symptoms of ME, chronic fatigue syndrome, they got better.
Øystein - That's correct.
Chris - Olav, I was going to bring you in and say, what actually happened to these people when you did this?
Olav - The 3 pilot patients all had response to treatment and one thing we observed in these 3 patients is that we have a pattern of responses and relapses after the rituximab treatment with a lag time of several months from initial and rapid B-cell depletion until they start getting clinical responses. Such patients are also seen in established autoimmune diseases after rituximab treatment. We believe that this fits with B-cells not being produced for a period after rituximab which allows a natural degradation of autoantibodies - that is antibodies that have adverse effects on bodily functions, with the symptom improving when the antibody level drops. We really think this points at ME at least in a large subportion is an autoimmune disease. It's also spotted that 70% of the patients with ME, they get it immediately after an infection and there's, like in other autoimmune diseases, 3 to 4 times as many women as men who get the disease. And also, a big study from the US has shown a moderate and highly significant risk of B-cell lymphoma in elderly ME CSF patients, showing that the patients may have chronically activated B-cell system. We see the same lymphoma risk also in established autoimmune diseases like rheumatoid arthritis, lupus and Sjogren's disease.
Chris - So, putting all of this together, you get these patients who, they happen to have a lymphoma - a cancer of the blood system - but they also have chronic fatigue syndrome. You treat their lymphoma with a drug which takes down their B-cells which are the cause of their cancer. They get this pattern of, their disease recovers when the B-cells go away but with the time lag corresponding to the time it takes the B-cells to go and the antibodies to go. And then when the B-cells come back in these patients, then the symptoms come back which looks like it's tying the two things together, doesn't it? So, what do you think that the B-cells are doing in these people to actually make, Olav, the symptoms of chronic fatigue syndrome in those patients?
Olav - Well, we think that it is a kind of an immune response and it obviously affects some very central function in the body. We're not at all convinced that ME is kind of inflammatory condition in the brain. Probably, more important, I think what is happening in blood vessels, we have indications that the blood vessel do not function as they should, given the dynamic flow to different parts of the body when it is needed.
Øystein - It's like the patients have a problem in the fine-tuning or regulation of blood flow according to the demands of the tissues for oxygen and nutrients. The patients often describe they feel like running a marathon when they have done a limited exertion and they get brain fog from exertion and so on. So, our hypothesis is that the immune system somehow disturbs the fine-tune regulation of blood flow and tissues including in the brain.
Chris - Øystein, what are you now doing to try to firm this up because your initial results as you published, while very interesting are on very small numbers of patients? Obviously, we'd like to see big numbers of patients in order to make sure that this is not a statistical blip, it's not happening due to chance, it's real.
Øystein - To try to convince ourselves, we first did, as you say, a small randomised study which was published in 2011 with 15 patients given two infusions of rituximab and 15 patients given placebo, turn out that the 15 that got rituximab had a clinical response while 2 of the 15 in the placebo group. So, that was some kind of sign of clinical activity to us. So then we did an open label study with no placebo group, further rituximab infusion, prolonging the period with low B-cells. We gave 6 infusions of rituximab up to 15 months and then followed the patient for 3 years. So, this study is now submitted for publication but we can say that again, 2/3 had a clinical response and the response durations were much prolonged when we gave maintenance treatment. But these two studies are not designed to give a definite answer to whether rituximab works in ME. So therefore, we are now performing a larger phase III study in Multicentre in Norway with 152 patients. Half of them will receive 6 rituximab infusions during the first year and half of them will receive placebo. And then we will follow the patients for 2 years. And we hope that this study will either verify or refute if rituximab may give benefit to ME patients in a significant proportion.

44:53 - Does the immune system cause depression?
Does the immune system cause depression?
with Declan Jones, Johnson and Johnson
Can the immune system cause more common complaints, like depression? We all go through spells of feeling down, but for ten percent of people the symptoms persist for weeks or months.  What role might immune responses play in this? Kat Arney spoke with Declan Jones, the neuroscience lead at the pharmaceutical company Johnson and Johnson...
What role might immune responses play in this? Kat Arney spoke with Declan Jones, the neuroscience lead at the pharmaceutical company Johnson and Johnson...
Declan - The link between the immune system and depression is not a new idea. It's been around for many years now, but it's still very exciting. There are several lines of evidence to suggest a role of the immune system in depression. First off, there's a significant increased risk of depression in patients with autoimmune diseases such as rheumatoid arthritis and psoriasis. I think some very direct evidence is that drugs which enhance the immune system such as interferon alpha which is used for the treatment of hepatitis C can cause depression-like symptoms as side effects in almost a third of patients. For me, that's very compelling data.
Kat - With cases like arthritis and things like that, is that not just because someone is in constant pain that they then become depressed?
Declan - Well, it's a good question. It's very difficult to disentangle the symptoms. We actually have a piece of work ongoing which is funded by the Medical Research Council and it's a consortium between ourselves and GlaxoSmithKline and a number of academic groups led by Ed Bullmore here in Cambridge. One of the things we're trying to do is, can we take some mood data from our immunology trials to really understand, can we distinguish the effects on mood from the effects upon say, the joint damage. At the moment, it's very hard to disentangle but there seems to be evidence that you can actually have effects on mood without actually having effects on the physical symptoms and vice versa. So for us, that's a really interesting piece that we're trying to follow up on.
Kat - So, for people where the drugs don't necessarily work for the disease you're trying to treat them for, actually, they either feel a lot more depressed or less depressed.
Declan - Less depressed, with some treatment for rheumatoid arthritis. There's been anecdotal cases by physicians and it's something that was obvious to physicians who weren't psychiatrists. It's also clear. I mean, patients with depression have elevated levels of some of the immune molecules in the blood such as cytokines IL-6, IL-1 beta and TNF-alpha.
Kat - These are all chemicals produced by white blood cells, produced by immune cells basically.
Declan - Yes, exactly and can act as messengers for the immune system to actually directly impact upon the brain function.
Kat - So, what's actually going on here? How is this alteration in the immune system causing depression or causing this very low mood?
Declan - There's increasing understanding, but it's still not entirely clear. There is some evidence to suggest that the inflammatory markers, the cytokines that I mentioned before can actually have direct effects upon - on enzymes in our brain which alter the level of neurochemicals such as serotonin, noradrenalin or dopamine. As you know, these are neurochemicals we know are associated with mood function.
Kat - They're kind of the sort of happy chemicals when your cells talk to each other in the brain, they go, "Yeah! Things are good."
Declan - That's exactly right and effects upon synaptic plasticity, actually, the way that the synapses function. And maybe, you know, neurogenesis, so production of new neurons. There's increasing evidence that immune mediators can actually have a direct effect upon brain function.
Kat - You mentioned this is not necessarily new, the idea that depression can be linked to immune function, but the idea that there is quite so much immune meddling in the brain, is this quite a new field that's opening up?
Declan - It's a field that's been active for quite a few years now, but I think the reason why we're getting excited by this is because I think there's been significant advances in the field of immunology and novel treatments for autoimmune diseases. So the excitement might be that you could take those treatments, actually use them now in depression. So, I think there's compelling evidence and there's actually converging evidence from different groups, from clinicians, from pre-clinical scientists. And what we're trying to do is say, can we actually jump on the back of the improvements in immunology?
Kat - So that's what I want to ask really. There's two prongs to this. It's the idea that maybe you could have a blood test, if someone comes to the doctor and says, "I think I'm depressed. I'm really not feeling good about life." Could you have a blood test to look for this kind of immune-related depression? And then obviously, the treatments for depression as we know are not necessarily as great as they could be so, more immune-based drugs in the future.
Declan - We're part of the consortium with the Wellcome Trust and this is led by Ed Bullmore in Cambridge and we're working with our colleagues at Lundbeck and another pharmaceutical companies and a host of excellent academic partners. But one of the pieces of work that we really want to understand is, what is the role of immune system? How can we demonstrate perhaps that there really are robust signals of immune changes in depressed patients? But also, can we distinguish between depressed patients who respond well to antidepressant drugs and those who, perhaps who don't? So, can we actually identify blood markers that would be able to help us to identify the right patients to put into a clinical trial and then as a separate piece of work, we're trying to understand what are the best molecules to test so that at the end, we may be able to do a therapeutic proof of concept trial in the right patients using the right molecule.
Kat - And the disease that I know a lot about is cancer and it's much more moving towards doing say, a genetic test on someone saying, "Okay, you have this type of tumour with this gene fault. You need X, Y, Z drug." In terms of looking at maybe someone's genome and looking at the way that their genome interacts with their immune system and affects levels of immune markers, is this the way that things might go in the future?
Declan - Potentially. I think we're a long way from that. Part of the work that we're doing in this consortium is to try and understand the genetic link, the genetic risk factors and how they may link with the immune system and depression. But I think at the moment, we're quite some distance away from that.
Kat - And very briefly, we heard from Susannah right at the beginning that she was very keen to de-stigmatise the disease that she'd had and we know that there's still a huge amount of stigma around mental illness, even very common ones like depression. Do you think more and more trying to talk about it as an illness caused by organic molecules in our body that have gone wrong? Is that going to be helpful for sufferers from this condition?
Declan - I believe so. I think the more we talk about it, the more research we do into this, I think the better this will be for patients that we may be able to choose the right therapy that might not even be drug therapy. It could be talking therapy or even nervous stimulation. But I think the more research that goes into it, the more people get involved in research and help the scientists, and I think the better that will be.

Can cheese give you nightmares?
Upon being visited by three ghosts in one night, it was Ebenezer Scrooge who famously blamed a crumb of cheese for his ghoulish encounters. But Scrooge and indeed his creator Dickens are not the only believers in the relationship between cheese and nightmares.
The supposed link between late night cheese binges and bad dreams had stood the test of time. but is there any science to back up the stories?
One promising theory comes from an active ingredient in cheese known as tryptophan - an essential amino acid, tryptophan is used by our own bodies to make the chemical neurotransmitter serotonin which is being proven to play a role in mood, memory, and you guessed it, sleep. But whilst cheese may provide us with the tryptophan needed to produce more serotonin which in turn may alter sleep patterns, the specific role it plays in nightmares is less clear.
Interestingly though, poultry such as turkey also has very high levels of tryptophan which may explain grandad getting a bit sleepy after Christmas lunch. While we might not be able to directly link cheese to nightmares, is there any other explanation for the food we eat affecting our dreams?
Apart from tryptophan, another major constituent in cheese is fat. Apart from expanding your waistline, fat is also particularly hard for the body to digest. Because of this, consuming high levels of fat less than 3 hours before bedtime can lead to periods of indigestion and ultimately, interrupted sleep.
While a restless night doesn't cause nightmares, what it can lead to is better dream recollection.
A recent neurobiological study from the Neuroscience Research Centre in Lyon examined why some people are naturally more inclined to recall dreams and some are not. Aside from slight differences in people's brains, they found that those who are better at remembering dreams had more periods of wakefulness. Now, they theorise that this might have something to do with the encoding of these dreams in subconscious memory. So, it would appear that late-night eating of high fat foods such as cheese can lead to more periods of wakefulness and this in turn can lead to better encoding and recall of dreams or drum roll please. nightmares. Thus, leading poor old Ebonezer Scrooge to believe that it was indeed a crumb of cheese that brought about the ghost of Christmas past, present, and future.










Comments
Add a comment