On this week's Naked Scientists, we tackle your questions. We find out what creates a 'Moonbow', how much water there was on Earth over one million years ago and what happens to milk in the freezer. Also, how butterflies could remember what caterpillars learn, why electric cars may stress stretched water supplies and how the 'smell' of a coral reef attracts fish from miles around. Plus, we speak to Marc Abrahams, creator of the Ig Nobel awards for science that makes you laugh, then makes you think! And in Kitchen Science we try to strike a balance between two balloons!
In this episode

02:03 - Memories Survive Metamorphosis
Memories Survive Metamorphosis
Despite dramatic changes in every aspect of their biology, it seems that moths and butterflies can remember things they learned as caterpillars.
Caterpillars can be thought of as eating machines - as they do very little but eat to store up they energy they will need when they form a chrysalis. During metamorphosis, as they become a butterfly or moth, they undergo a radical change in body plan as well as lifestyle and diet. The change is so dramatic that it had been assumed that memories or associations learned during the larval, or caterpillar period would be lost to the adult stage.
Writing in the March issue of PLoS One, Doug Blackiston and colleagues at Georgetown university trained tobacco hornworm caterpillars to avoid certain odours by associating the smell with an electric shock. After metamorphosis, the adult moths also avoided the odours, showing they had retained the association despite huge changes in physiology.
However, the ability to retain these memories depended on when they were learned - Caterpillars younger that three weeks old could learn to avoid the odour, but did not retain the memory into adulthood. In those caterpillars conditioned shortly before pupation, the memories survived the pupal stage and were demonstrated in the adult.
Retaining memories from before metamorphosis could allow female butterflies to lay her eggs on the same type of host plant that she fed on during her larval stage. This 'habitat selection' could lead to the development of new sub species of butterfly.

Mysterious Forces
For the last 10 years physicists have been puzzled by a mysterious force acting on space probes. There have been various slight anomalies detected in how spacecraft move which don't seem to be moving quite as we would predict them to.
One of the first anomalies was noticed on the pioneer spacecraft that visited the outer solar system in the 1970s and are still heading off into space. They seem to be slowing down slightly faster than we would predict using our present understanding of physics. This effect is minute, about a ten billionth as strong as the force of gravity on the surface of the earth but it is measurable. This effect could just be due to some physics we already understand, like a gas leak or we don't understand how the probe is radiating heat.
 Now a team from the jet propulsion laboratory in California have found another anomaly which seems to appear when some spacecraft make a flyby of earth. For examble when the NEAR spacecraft flew past the earth on its way to visit an asteroid, it left moving at 13mm/s slower than it should have done, and the Galileo probe was speeded up by 4mm/s these are tiny effects as the probes are moving more than a million times faster than this, but it is well within our ability to measure their speeds.
Now a team from the jet propulsion laboratory in California have found another anomaly which seems to appear when some spacecraft make a flyby of earth. For examble when the NEAR spacecraft flew past the earth on its way to visit an asteroid, it left moving at 13mm/s slower than it should have done, and the Galileo probe was speeded up by 4mm/s these are tiny effects as the probes are moving more than a million times faster than this, but it is well within our ability to measure their speeds.
Some probes have this effect and others don't, Frank Jordan who is part of the team which was looking at this effect and trying formulae to try and model these results and found one that works nicely. It seems to suggest that probes which approach and leave the earth at a similar angle to the equator won't feel an effect, but probes which enter and leave at different angles feel this strange extra force.
This doesn't say anything about what could be causing the force, but the fact that the equator is important could indicate that the earth's rotation is affecting it somehow. Einstein's theory of general relativity suggests some effects related to rotating massive objects, but they should be much smaller than this, so it is just possible that these may be some hints at some interesting new physics.

Water-Guzzlers
Running vehicles on alternatives to fossil fuels could stress scarce water resources, US scientists have warned.
 Michael Webber and Carey King, from the University of Texas at Austin, suggest that powering America's cars with electricity, rather than gasoline (petrol), could triple the nation's water consumption. And biofuels, derived from crops requiring irrigation, are even more water-intensive, according to a further unpublished analysis by the team.
Michael Webber and Carey King, from the University of Texas at Austin, suggest that powering America's cars with electricity, rather than gasoline (petrol), could triple the nation's water consumption. And biofuels, derived from crops requiring irrigation, are even more water-intensive, according to a further unpublished analysis by the team.
'This doesn't mean that we shouldn't try to switch to alternative fuels - but if we're going to do it, we need to be aware of a potential downside,' Webber explains.
Increased evaporation
Water is the main coolant in the coal, gas, or nuclear power plants that largely supply vehicles with electricity. Webber and Carey calculate that when switching from 'gasoline miles' to 'electric miles', approximately three times more water is evaporated into the atmosphere, and seventeen times more water withdrawn for cycling around the plant before being returned to a river or lake.
While parts of the US are water-rich, limited water availability in some drought-stricken areas (such as the south-east) might hinder the growth of power plant systems that would be needed to cope with the greater demand by electric vehicles, says Webber.
He recommends switching to less water-intensive methods of generating energy: for example, solar or wind power; using non-irrigated biofuels; or using reclaimed or salt water to cool power plants.
Neglected link
Peter Gleick, president of the environmental think-tank the Pacific Institute in Oakland, California, thinks that the added water demands Webber identifies may not be very significant. 'In regions where water is scarce, the key will be to produce fuels, or electricity, where water is less of a constraint,' he said.
But both Webber and Gleick feel that the link between water and energy has been neglected amid understandable concerns about security and carbon-costs of energy supplies. 'We've typically considered water and energy separately, and it is no longer appropriate to do so. We can no longer push for energy policies that ignore our water challenges, just as we can't push for water policies that ignore energy costs and benefits,' Gleick said.
Richard Van Noorden

The Smell of the Reef
Phytoplankton and algae living on coral reefs release a chemical which attracts fish from miles around, and could play a key role in reef ecology.
 Ecosystems are all about balance, and the phytoplankton have an ingenious way to keep things in check. Phytoplankton are similar to plants, they harvest energy from sunlight using chlorophyll, and are a vital part of the ocean ecosystem. Zooplankton also play a vital role, including feeding on the phytoplankton. Jennifer DeBose and colleagues at the University of California at Davis have shown that in response to being grazed by zooplankton, phytoplankton release a chemical, Dimethylsulfoniopropionate (DMSP), which attracts fish which feed on zooplankton.
Ecosystems are all about balance, and the phytoplankton have an ingenious way to keep things in check. Phytoplankton are similar to plants, they harvest energy from sunlight using chlorophyll, and are a vital part of the ocean ecosystem. Zooplankton also play a vital role, including feeding on the phytoplankton. Jennifer DeBose and colleagues at the University of California at Davis have shown that in response to being grazed by zooplankton, phytoplankton release a chemical, Dimethylsulfoniopropionate (DMSP), which attracts fish which feed on zooplankton.
DeBose and team tested the hypothesis at different reefs by releasing either DMSP or distilled water from special tanks and found over four times as many fish attracted to the DMSP plumes.
Their findings suggest that planktivorous fish, those that eat plankton, have developed a way to 'eavesdrop' on the interactions between phyto- and zooplankton to help find good food resources.
Can you get moonbows?
Dave: Was the moon behind you or in front of you?Sam: It was in front of me and I was going into the rain.Dave: I think that is a straight moonbow. It's exactly the same as a rainbow caused by the sun so what happens is, if you shine light on a droplet of water the light will bounce around inside it and when it comes out different colours will come out in different directions. So if you look at different places you'll see a rainbow. The moon is a source of essentially white light so you'll get exactly the same effect. Because it's so dim your eyes won't be sensitive to the colour so you just see a bow of brightness rather than a colour. If you took a very long exposure photograph of it or you had an image intensifier which worked in colours then you would be able to see the colours and it would look just like a normal rainbow.Sam: I was looking them up afterwards to see if I was imagining things. It seems they're quite rare.Dave: Yes, proper rainbows are. Normally people just see a ring around the moon itself, maybe 20-30 degrees outside the moon which is normally created by little ice particles. It isn't a true rainbow, it's a similar sort of effect.

Why does milk change colour when it freezes?
The reason for this is that milk isn't just a straightforward solution, it's not like dissolving salt in water. Milk is what's called an emulsion. It's a mixture of various things which are suspended in water. Milk is about 5% fat and that fat is dispersed through the water as tiny globules. They're cells and surrounded by little proteins. They have a fat-loving part of the protein and a water-loving end. The fat loving part of the protein sticks into the fat and the water-loving bit sticks into the water. This helps to solubalise or keep suspended the fat molecules. Also in the milk are these things called pepsinogens or proteins because milk contains a lot of protein.
The idea of milk is to nourish a growing animal and protein is what you need to grow more muscle and get bigger. When you put your milk in the freezer all these things which are normally well-suspended and kept separate begin to become very close together. The freezing process means that the water in the milk, 95% forms big chunky ice crystals and they don't want to have the proteins and the fat in them. The proteins and the fat get squeezed out of the mixture. They tend to form around this central core of ice. So because you see all the fat in one place it looks yellow. If you look where butter comes from, butter comes from milk and it's yellow. That's where the yellow colour comes from. It's all conglomerated in one place and you can actually, physically see it.

Has the Earth gained or lost water?
Chris: In that sort of timeframe, I reckon the answer is [Earth's water has remained] roughly the same. It does increase by a small amount - I think the stated geological figure is about 1 inch every 20,000 years or so - but that's extrapolated over the lifetime of the Earth.
Most of the water we have came in the form of comets and asteroids hitting the Earth. When the Earth first formed, of course, there was a disc of stellar debris which was basically the material left over from when the sun formed. Out of that we condensed planets, as planetesimals. They slowly aggregated more material and formed bigger planets. The material left over was comets and other asteroids and other bodies, which were out there in orbit.
Comets are viewed as "dirty ice balls," - basically lots of water with some other stuff chucked into them. Occasionally one of them's going to cross the orbit of another planet, get drawn in by gravity and crash land. Most of the water on Earth, we think, comes from comets, originally. Given that they're not actually that common these days but over the millions of years time scale I'd say the amount of water on Earth hasn't changed a huge amount. I would add that it probably is increasing very, very slightly. What do you think, Dave?
Dave: There is also the mechanism whereby the Earth loses water. What happens is, in the upper atmosphere you've got a little bit of water vapour high up in the atmosphere and it gets hit by ultraviolet light from the sun. That can split apart into hydrogen and oxygen. This light hydrogen will tend to float up really high and then get blown away by the solar wind. It's a very, very slow process but we are losing hydrogen from the water all the time. The oxygen will stay on the Earth because it is much heavier.
Chris: The same thing happened to Mars, didn't it? About 4 billion years ago, when Mars was about 400 million years old, it lost its magnetic field because the planet got too cold to have a liquid iron core. Because that's how the planet generates its magnetic field, it couldn't therefore have a magnetic field. That meant it was vulnerable to the solar wind which was just plucking all of the gas, the atmosphere and the water from the planet and it dried out.
Dave: And also to Venus. Venus is very similar to the Earth but has a much weaker magnetic field which tends to protect it from the solar wind. We think that Venus at the moment is about 500 degrees centigrade on the surface and the difference between Venus and Earth is that the Earth has a magnetic field which stops it losing water. Venus has lost all its water.

What's the difference between salt and fresh water?
Spot on, Alan. They are, as you say, the same chemical. H2O is water.
The water cycle on Earth is why we have fresh water.
What happens is the energy from the Sun hits the Earth's surface and the oceans' surface. Every square metre of the Earth gets about one kilowatt of energy coming from the Sun.
That's like having a one bar electric fire shining on it. That heat evaporates the water from the surface of the ocean.
Water can leave quite easily, but the heavier elements and ions which are dissolved in the water find it much more difficult, so they stay behind.
What evaporates is essentially fresh water. This goes up and forms clouds. These then drift across the ocean's surface until they're forced to rise over, say, a mountain range or something.
As the clouds are forced to rise they find it much harder to hold on to water which is condensing inside them, clouds are just massive bodies of tiny droplets of water.
Water rains down on Earth and falls on the ground as fresh water. As it percolates through rocks and rivers it absorbs small amounts of salts on the way through becoming slightly salty in the process but not perceptibly salty.
As it slowly drains into the ocean it carries those salts with it to the sea, which picks up those salts.
What leaves the sea is fresh water again, so the sea is continuously becoming slightly more salty.
Why is the sea not becoming more and more salty? The answer is it's become about as salty as it's ever going to because it's now reached sort of equilibrium position where if you add more salt to the sea chemical reactions kick in and take it out again.
Should you walk or run through rain?
If the wind isn't blowing and the rain's falling straight down vertically, as you travel through the rain then your front will always hit the same amount of rain no matter how fast you run. If you go slowly you'll get more that falls on top of your head, so running is slightly better than walking.
But don't fall over because if you fall over you'll get very wet indeed!

How much water does a dripping tap waste?
We've done a couple of rough calculations on this. If you've got a drip, say, 1 tenth of a millilitre and it drips every ten seconds, it's going to lose you about 36 millilitres an hour. If you multiply it out for the whole year it's about 315 litres a year, one or two baths full, depending on how big your bath and your drip is!
So if you have a dripping tap it's really worth fixing because one drip every ten seconds is really quite slow for a tap but if you're talking several a second then you're losing quite a lot of water.
Why does the windscreen mist up faster when it rains?
There's two things to think about here. Number one, when it's raining the air's pretty moist. Lots of water in the air even if you're sitting inside the car the airs going to have a lot of water vapour in it. It's going to condense onto the inside of your windscreen. The second point to think about is that when it's raining outside you've got cold water hitting the outside of your windscreen. Previously you'd just have some air going over the windscreen. The cold water's just going to take the heat away much better than the air does. That's really encouraging any water on the inside of the car to condense on the inside of the screen. It'll mist up your windscreen very quickly.
Why does frozen milk turn back into milk?
Well, it's all about energetics. In other words, when you bring the temperature back up you've got molecules of water bumping into each other and the consequence of that is that the particles of milk reform into these little cells which then spread out evenly through the milk. That's the most energetically stable way for them to organise themselves.

27:11 - Artificial Enzymes, Sea Cucumber Skins and Safer Chips
Artificial Enzymes, Sea Cucumber Skins and Safer Chips
with Mark Peplow, Chemistry World
Chris - Mark Peplow is the editor of Chemistry World. Mark hello, welcome to the programme.
Mark - Hello Chris.
Chris - So tell us about this first item.
Mark - Scientists in America have managed to design and build a working artificial enzyme from scratch. Now enzymes are nature's catalysts, they speed up chemical reactions in our bodies by tens of millions of times. We wouldn't work as living beings without enzymes. They're blobby proteins with a cavity in them perfect to fit chemicals that are involved in reactions. It brings them together in the perfect way to beat them up. You can hijack these to do useful chemistry. For example, an enzyme in yeast turns sugar into alcohol.
Chris - That is very handy!
Mark - But what about non-natural reactions? What happens if that reaction you want to catalyse doesn't exist in nature. Normally chemists use things like metals or metal compounds to do that but it would be much better to be able to persuade bacteria to do these reactions for us but to do that you need to make your own enzymes. Now Ling Jiang and colleagues at the University of Washington have done just that. They have made a computer model of a particular reaction called a retro-aldol and they designed an enzyme to fit around it. Then they actually made 72 similar versions of this enzyme in the lab. They found that 32 of them worked and the best one speeded it up by about ten thousand times. That's still a thousand times worse than what you see in nature but it proves that you can actually make these things from scratch. It's exactly the sort of thing that if Craig Venter gets his hands on it and puts it in one of his artificial life forms that he's promising to unveil next year: one of these artificial bacteria. This is exactly the sort of thing that you could put into that system.
Chris - It just goes to show that if you borrow from biology nature spent millions of years getting things right. If we apply the same concepts nature does and do it slightly differently you can actually come up with some pretty elegant solutions.
Mark - That's right.
Chris - In the same vein, sea cucumbers are providing a whole raft of solutions.
 Mark - This is a lovely little story that came out this week. Sea cucumbers are a relative of a star fish in that they graze on the ocean floor for carrion. When they're startled they can become rigid in seconds, basically because they can control the collagen fibres that are in heir skin. Now scientists at Case Western University in Cleveland, Ohio, have used this as inspiration to make their own material which does exactly the same thing. They made nano-fibres of cellulose, these are thousands of times thinner than a human hair and they embedded them within a sort of rubbery polymer substance. What they found that normally when dry the material's quite stiff. The nano-fibres are really glued to each other. As soon as you introduce water it separates the nano-fibres and the properties of the polymer start to dominate the composites so it's soft and flexible. Now, they think that these sorts of materials could be used, for example, in the micro-electrodes that are used in brain implants to help Parkinson's sufferers. Normally you'd assemble the components in dry conditions so they're always stiff, easy to work with but as soon as they get into the wet brains they become soft and flexible. That's one way that you could potentially reduce the scarring that you see in these sorts of implants.
Mark - This is a lovely little story that came out this week. Sea cucumbers are a relative of a star fish in that they graze on the ocean floor for carrion. When they're startled they can become rigid in seconds, basically because they can control the collagen fibres that are in heir skin. Now scientists at Case Western University in Cleveland, Ohio, have used this as inspiration to make their own material which does exactly the same thing. They made nano-fibres of cellulose, these are thousands of times thinner than a human hair and they embedded them within a sort of rubbery polymer substance. What they found that normally when dry the material's quite stiff. The nano-fibres are really glued to each other. As soon as you introduce water it separates the nano-fibres and the properties of the polymer start to dominate the composites so it's soft and flexible. Now, they think that these sorts of materials could be used, for example, in the micro-electrodes that are used in brain implants to help Parkinson's sufferers. Normally you'd assemble the components in dry conditions so they're always stiff, easy to work with but as soon as they get into the wet brains they become soft and flexible. That's one way that you could potentially reduce the scarring that you see in these sorts of implants.
Chris - Exactly how do they envisage this being done because they've actually had to go to these animals and get the fibres?
Mark - No, this is the thing. This is the inspiration that they're using exactly the same mechanism. They're making their own nano-fibres of cellulose. It's basically the same arrangement of thin threads packed into a rubbery, soft material. Depending on the conditions, the whole composite can behave like a rubber or like a stiff CD case, in fact.
Chris - Just brain implants or other things as well?
Mark - Well, this is the thing. Potentially, we spoke to a few scientists about this. A guy called Craig Hawker at the University of Santa Barbara in California was so excited about this, he was gushing about it. He was saying, why not think further? Why not do exactly what the sea cucumber uses this for? You could make a defensive suit which, under certain conditions, becomes impenetrable.
Chris - Sounds handy on the streets of Cambridge. Now what about this last one which is going to have people who are into their chips, that's French fries for our American friends; why should soaking them make them safer?
 Mark - Yeah. Back in 2002 people were quite surprised when a group of Swedish researchers said that a chemical called acrylamide, which is thought to be a cancer-causing agent, was found really widely in baked and fried food. This happens because when you heat food like that up there's a reaction that goes on among the amino acids in the food and the sugars in the food. The trouble is, this is part of the browning process that generates flavour.
Mark - Yeah. Back in 2002 people were quite surprised when a group of Swedish researchers said that a chemical called acrylamide, which is thought to be a cancer-causing agent, was found really widely in baked and fried food. This happens because when you heat food like that up there's a reaction that goes on among the amino acids in the food and the sugars in the food. The trouble is, this is part of the browning process that generates flavour.
Chris - But it makes it taste great!
Mark - It tastes great so the problem is how do you get rid of it? There's an enzyme that you can use to destroy the amino acid that's involved in this and indeed, some scientists based in Denmark have now proved that you can do this to cut out a huge amount of the acrylamide that you normally get in this. This is for the sort of manufacturers that make ready-made oven chips. Scientists in Britain have also shown, just last week, that simply soaking chips in water before you cook them can halve the acrylamide levels. There is a drawback though. We talked to the UK's leading expert on acrylamides, Bronek Wedzicha at the of the University of Leeds, he said, "you know, this is great but the trouble is it's going to give you pale chips that lack flavour." The flavour is so intrinsically tied to the reaction that makes this chemical acrylamide.
Chris - I'm sure they can find something suitably carcinogenic to flavour them with afterwards.
Mark - I'm sure.

33:27 - The Ig Nobel Prizes
The Ig Nobel Prizes
with Marc Abrahams
Chris - The Ig Nobel prizes honour achievements that make people first laugh and then think. The prizes are meant to celebrate the unusual, honour the imaginative and also to spur people's interest in science, medicine and technology. As one major journal has put it, they come with little cash but much cache. The guy who sorted it all out and got it going is Marc Abrahams. So Marc, why did you start this?
Marc - Let me correct something first, "they come with little cash." They come with no cash.
Chris - Ok. Fair enough. Why did you get it going? What was the idea?
 Marc - I had become the editor of a science journal and I had discovered that when you edit a science journal, I'm sure that Mark [Peplow] has the same thing, suddenly all kinds of people get in touch with you. They tell you about the wonderful things they've done. Some of those things are wonderful, some aren't. You know that most of these people, no matter what they do will never ever get any recognition from anybody. I kept running into people who had done things that were funny and interesting. It seemed a shame that nobody would ever know about it. That, more or less, is why we started the Ig Nobels.
Marc - I had become the editor of a science journal and I had discovered that when you edit a science journal, I'm sure that Mark [Peplow] has the same thing, suddenly all kinds of people get in touch with you. They tell you about the wonderful things they've done. Some of those things are wonderful, some aren't. You know that most of these people, no matter what they do will never ever get any recognition from anybody. I kept running into people who had done things that were funny and interesting. It seemed a shame that nobody would ever know about it. That, more or less, is why we started the Ig Nobels.
Chris - The point about the Naked Scientists is we try and have fun with science. Because at the end of the day if people are impressed as well as amused they'll tend to remember and also listen harder, I think.
Marc - Yeah, I don't think there's too much reason to try to bore people. I've never heard of people arguing that.
Chris - Let's look at some of the things you've got on your tour and past prizes. I love the one about country music.
Marc - Yeah, we'll be doing some shows in London, a couple of shows in London this week and then in Newcastle on Friday. There is a professor Marc A Gundlack from Alabama who, with a colleague, did a study about the effects of country music on suicide.
Chris - And?
Marc - He found there may be some effects of country music on suicide!
Chris - Does it prevent or does it encourage suicide?
Marc - Well, the numbers are about ten years old so as to what would happen right now which I think is what you're really getting at no one can say. What he found was that in cities that played a lot of country music on the radio the suicide rates were a little bit higher.
Chris - By all accounts, there is a very popular country show on this radio station and it certainly does very well. It's thriving and gets lots of calls so the audience are certainly not dead.
Marc - No one's tried to sue you yet ..
Chris - Not me personally! Is it people who are listening to the show who are writing themselves out a death wish or is it people who incidentally bump into country music and aren't habituated?
Marc - There is an effect, it's on people listening to music. It's not on people who don't listen to music.
Chris - There's an interesting thing in Question of the Week, this week. If you cut hair does it grow more? There's this idea that if you cut hair, and my wife worries about this, that if she shaves her legs they'll get more hairy. Are people worried about pubic lice?
Marc - Of course people are worried about pubic lice. As soon as you mention it people get worried. One of the people of note is Kees Moeliker from the Netherlands. He's the guy who won the Ig Nobel prize a few years ago. He's an ornithologist. He won his prize because he documented the first known case of homosexual necrophilia in the Mallard duck. That's not why he's here this year. This year he's going to talk about a study done at Leeds by two doctors who said that in the past ten years the incidences of pubic lice in Britain have just fallen. It's almost in viro. The question is why? They have a theory. Their theory is that this is exactly the same time period when we saw a rise in popularity of that hairstyle known as the Brazilian.
Chris - So it's just the absence of a suitable environment.
Marc - Yes, that's well-put.
Chris - So you can see the correlate of that in Nature all the time. If you cut trees down do you have any bears?
Marc - This has yet to be proven and Kees is touring Britain partly because he's te curator of a Natural History Museum. Here we have a report of an imminently endangered species and he's asking that anybody who has a sample, a living sample of human lice, please come to the show as he would love to collect it and take it to the museum.
Chris - The problem is, how are you going to conserve them because they only really like living on us?
Marc - One step at a time here. First you get the specimens while they're still alive.
Chris - Then you look for suitable hosts?
Marc - Could be irritant.
Chris - One person we had on the programme about a year or so ago was Clare Rind. She's a researcher at Newcastle. She came to you guys' attention, the Ig Nobel prizes because she showed locusts Star Wars and monitored their brain activity.
Marc - This is true. She and her colleague, Peter Simmons, monitored the activity of one brain cell of a locust while they had that locust strapped down and watching selected scenes from Star Wars. The scenes they were watching were in the original Star Wars movie if you remember the fighters diving to the trough inside the Death Star. What they were wondering about was locusts typically are in swarm s: tens of thousands, hundreds of thousands, maybe millions but they seem not to collide very often. They were wondering what's going on inside the brain of a locust that helps prevent these collisions.
Chris - They did actually get quite a lot of interesting answers out, didn't they? What they're now saying is that they could apply the neurological version of that to a robot.
Marc - I believe they also got some funding from one of the large automobile manufacturers, too, who are also interested in preventing collisions. Although, they don't care that much about locusts.
Chris - Could come in handy on the roads in the UK which are hopelessly overcrowded. Marc Abrahams, thank you very much. Just tell us so that people can come along if they want to; where are the next set of shows and how do they find out more if they want to come and see your shows?
Marc - The schedule is on www.improbable.com. The shows are Tuesday night at Imperial College, Wednesday night at The Guardian's museum on Faringdon Rd. Then on Friday night in the Life Centre in Newcastle.
Chris - And they'll basically get yourself..?
Marc - Oh, a whole bunch of people. Ourselves and the Ig Nobel prize winners and there are free events too.
Chris - If someone wants to nominate someone?
Marc - Oh, please do! We're always collecting nominations. That's one of the best parts of the tour is people come up afterwards with things they've discovered or people they know.
Chris - Marc, thank you very much.
40:22 - Skygazing
Skygazing
with Rick Fienberg, Sky and Telescope Magazine
Dave - Now it's time to hear from Rick Fienberg of Sky and Telescope Magazine. Rick spoke to Meera earlier this week about the latest developments this week in space. First up, GLOBEatnight: a project to bring together both professional and amateur astronomers with the public and get them all skygazing.
 Rick - One of the newest things that's happened already is a worldwide effort on the part of professional astronomers and amateur astronomers and even among ordinary citizens to measure the darkness or brightness of the night sky to try to figure out what the impact of light pollution is. When astronomers talk about light pollution we're talking about artificial lighting which is wastefully being shot up into the sky, causing faint stars to disappear. Most people live in cities now and they rarely get to see the night sky in all its glory.
Rick - One of the newest things that's happened already is a worldwide effort on the part of professional astronomers and amateur astronomers and even among ordinary citizens to measure the darkness or brightness of the night sky to try to figure out what the impact of light pollution is. When astronomers talk about light pollution we're talking about artificial lighting which is wastefully being shot up into the sky, causing faint stars to disappear. Most people live in cities now and they rarely get to see the night sky in all its glory.
Meera - What's the project actually getting people to do?
Rick - The GLOBE project is very simple, they ask people to go out at night in late February and early March and look at the constellation Orion which is very familiar to most stargazers and visible to most points of the Earth at this time of year. They count how many stars they can see in the constellation and they compare their view of the constellation with the templates that are on the GLOBEatnight website and determine whether they're seeing stars that are at a third magnitude or a fainter fourth magnitude or even fainter fifth magnitude. That's how astronomers measure star brightness. That enables the GLOBEatnight project to do a worldwide map of sky brightness.
Meera - Has anything been revealed so far?
Rick - Well, this is the third year. In 2006, 2007 they had something like 10-20,000 participants doing measurements and they've been able to determine that measurements done from the ground by people track very consistently with measurements done by satellites in orbit which is useful. If the information didn't match up we might begin to suspect that your satellites weren't operating properly. As you might expect, most of the light pollution is located in populous cities. One of the interesting things is that it's possible, if you have a large number of people doing measurements, in any particular city to find little enclaves where the night sky is a bit darker than surrounding areas. These are places where amateur astronomers are going to want to sit their telescopes because they'll be able to see more.
Meera - That would be quite useful for people just to know their local best spot.
Rick - Right and the GLOBEatnight project is planning to be much larger in 2009 because next year is the International Year of Astronomy that celebrates 400 years of telescope. Galileo first used the telescope to look at the stars in 1609. The plan is to have a lot more people than have been participating in the GLOBEatnight so far rejoin so that awareness of light pollution will be raised around the world.
Meera - Well, hopefully all will go well with the project and the number of astronomers out there will increase. Moving from the stars to the planets it turns out that the liquid water that was thought to have existed on Mars wasn't there at all.
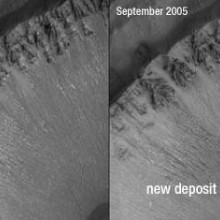
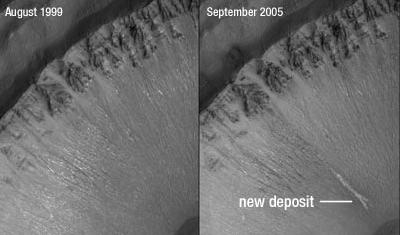 Rick - NASA's plan for studying Mars is to follow the water. That's how they put it. If you follow the water, you follow lines of evidence that might ultimately point toward life on other worlds. Astronomers have seen lots of evidence that liquid water flowed on Mars in the distant past. We have huge canyons on Mars, we have things that look exactly like river deltas so there's very little argument over the probability that liquid water flowed on the planet many, many years ago: we're talking millions or even billions of years ago. The question is, is there liquid water on Mars today? In late 2006, using images from the Mars Global Surveyor, several planetary scientists saw evidence that liquid water can actually flow on Mars even now. What they saw was a couple of gullies that were cut into the sides of craters. These gullies had suddenly fresh, light-coloured deposits in them. The thought was that between the time when the images were taken there were no deposits and then there were deposits liquid water had somehow erupted out of the gullies, probably from underground and slowed down and dropped some rocks into these gullies, causing them to look different than they had a few years earlier.
Rick - NASA's plan for studying Mars is to follow the water. That's how they put it. If you follow the water, you follow lines of evidence that might ultimately point toward life on other worlds. Astronomers have seen lots of evidence that liquid water flowed on Mars in the distant past. We have huge canyons on Mars, we have things that look exactly like river deltas so there's very little argument over the probability that liquid water flowed on the planet many, many years ago: we're talking millions or even billions of years ago. The question is, is there liquid water on Mars today? In late 2006, using images from the Mars Global Surveyor, several planetary scientists saw evidence that liquid water can actually flow on Mars even now. What they saw was a couple of gullies that were cut into the sides of craters. These gullies had suddenly fresh, light-coloured deposits in them. The thought was that between the time when the images were taken there were no deposits and then there were deposits liquid water had somehow erupted out of the gullies, probably from underground and slowed down and dropped some rocks into these gullies, causing them to look different than they had a few years earlier.
Meera - What's happened to make them not think this any more?
Rick - Now there's a much higher resolution camera in orbit on Mars (the Mars Reconnaissance Orbiter) and those craters that had the gullies in them were retargeted. Much higher resolution images enabled planetary scientists to build a computer model that tries to explain how these gulley deposits were laid down. What they did was they now had really good three-dimensional maps of the gullies. They simulated the creation of these light, streaky deposits through liquid flow, through a mud flow or a lava flow or through something more like a landslide: as if a hill had collapsed and all kinds of particles and rocks and dust had flown down. What they found was that they could reproduce the appearance of these deposits in the gullies much more precisely using a dry flow, something like a landslide than they could using a wet flow as if there had been a flash-flood in these gullies. Suddenly, the compelling evidence that liquid water had flowed on Mars as recently as the last few years was cast into doubt.

45:52 - Clean Cut Hair Grows Quicker?
Clean Cut Hair Grows Quicker?
Professor Des Tobin, University of Bradford:
Unlike most other mammals, we have a very specific personal care and hygiene regime. Washing doesn't affect the hair growth itself except for some effect you may get from massaging the scalp as you are washing the hair by ensuring optimal blood circulation in the scalp, which may have some positive benefits for the hair follicle itself.
Shaving does not impact on the quality of the actual hair fibre produced in that the hair is a dead entity above the surface of the skin. There is a perception of thickened hair re-growth because if you were to cut it with a sharp instrument like a razor you would end up getting a sharpened end of the hair at its thickest point rather than that more fine, tapered end.
Hair growth is very important for the success of the mammal and nature has provided enormous back-up systems to ensure that the hair continues to grow.
In the wild you can imagine the loss of a coat would really be disastrous for a mammal in terms of thermoregulation or camouflage etcetera. As a result the skin has invested enormous power into maintaining the hair follicle.
That's why it's hard to grow when you want it to grow or stop growing when you want it to stop growing. That's because the principal driver for hair growth is hormonal, especially in those areas of the body with changed hair pattern after puberty.
There's a lot of clinical evidence to suggest that if you have abnormalities of the endocrine system or the hormone system you can have altered patterns of hair growth: either too much or too little.

Where does dust come from?
Chris: I'm afraid actually, it is Nicholas, it's you and all the other people who have lived and visited your house...
The stats really stack up and they are really quite scary. The average human loses about 30 or 40 thousand dead skin cells every single minute. If you were to add them all up and put them in a giant bag, over the course of a lifetime that would weigh about 1-and-a-half stone in dead skin alone.
Most of that debris you see lying around your house is dead bits of you floating around! You're breathing that in, you're breathing in bits of your partner, your family, your visitors, your friends. It's just bits of yourself.
Nicholas - Well, I'll just have to accept that, but even so, 1.5 stone over the period of a lifetime doesn't seem like that much.
Chris: Yes, but a skin cell is very, very tiny. The weight of a cell is measured with about 9 zeros in front of it (10-9 kgs). It's tiny!
Nicholas: But a particle of dust is not one skin cell, is it?
Chris: No, these things are clusters. They get stuck together, and other fats and materials that are present stick them together. As a result you end up with something that forms a blob of dust.
Do fizzy drinks contribute to global warming?
The CO2 does escape from the water, but you shouldn't worry about it because of the source of the CO2. It comes from the waste streams of power plants, so it's essentially a by-product that would have made it's way into the atmosphere anyway. But, any consumer product takes manufacturing, shipping etc which does use up fossil fuels and release CO2 into the atmosphere. So giving up fizzy drinks could help reduce climate change, but not inthe way you thought!
Could we engineer a microbe to absorb carbon-dioxide?
Dave: I don't know about an airborne version, but we do already have microbes which do this - algae and plants. In the presence of sunlight, they feed on CO2 and give out oxygen. At the moment, they're not consuming the CO2 as quickly as we add it to the environment.Chris: Those algae are largely in the sea, and the sea accounts for over a third of the absorbtion of the CO2 that we put into the atmosphere.Dave: But even if more algae grows, something will come along and eat the algae, and then metabolise the sugars and carbohydrates and produce even more CO2.











Comments
Add a comment